Thanatophoric skeletal dysplasia is the most lethal, rare, sporadic birth defect due to de novo mutation in the fibroblast growth factor receptor-3. Clinically this is characterized by shortening of the limbs (micromelia), small conical thorax, flat vertebral bodies and macrocephaly at birth. We encountered a similar case with ultrasonographic findings suggestive of Thanatophoric skeletal dysplasia which resulted into death of the baby within an hour of birth. Almost all cases of this condition have been reported to have died interuterinally or few days after birth.
birth defect; micromelia; skeletal dysplasia; thanatophoric skeletal dysplasia
Skeletal dysplasia refers to heterogeneous group of heritable disorders characterized by abnormalities of cartilage and bone growth, resulting in abnormal shape and size of skeleton and disproportion of long bones, spine and head.1 Thanatophoric skeletal dysplasia (TSD) is the most lethal, rare, sporadic birth defect due to de novo mutation in the fibroblast growth factor receptor-3 (FGFR3). 2-7 We encountered a newborn baby with shortening of the limbs (micromelia), small conical thorax, flat vertebral bodies and macrocephaly and ultrasonographic findings suggestive of TSD. Because of very high mortality and no case report from Nepal, we aimed to report this case to make health professionals more aware about this rare condition.
A 22 years old non- smoker, non- alcoholic primigravida who had normal regular antenatal check-ups and no any chronic illnesses or family history of congenital abnormality came to Patan Hospital for her routine checkup at 39 weeks of gestation(WOG). USG was done revealing average age of fetus as 39 plus weeks and femoral length of 17 weeks of gestation (2.3 cm) only, biparietal diameter 9.9 cm (corresponding to 40 WOG), head circumference was 35.2 cm (corresponding to 41 WOG), Arm circumference 34.1 cm (corresponding to 38 WOG). All four limbs were small to corresponding gestation (micromelia) and amniotic fluid index (AFI) 30 cm which is suggestive of thanatophoric skeletal dysplasia (Figure 1).
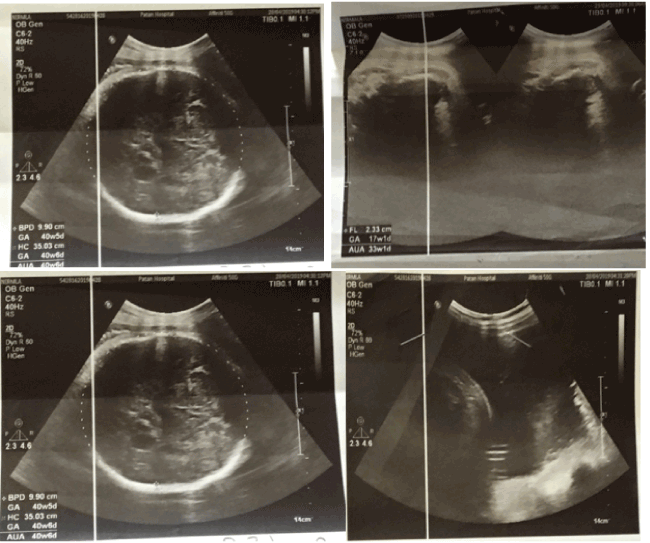
Figure 1. USG showing features suggestive of TSD
Emergency lower segment cesarean section (LSCS) was done at 39+6 WOG for fetal bradycardia and macrocephaly. A single, live, male with birth weight of 3270 grams was born. The Apgar score was 4/10 in first minute and 3/10 in fifth minute. The baby did not cry immediately after birth despite rigorous stimulation and suctioning. We started bag and mask ventilation and still there was no cry and heart rate were less than 60 beats per minutes which was gradually decreasing. Meanwhile, need for ventilator support and NICU care was counselled to patient’s family but they denied to do any lifesaving interventions and the baby died after an hour of birth.
Examination revealed length of 36 cm (<5th centile for newborn boy), macrocephaly (OFC: 38cm, in-between 90th and 95th percentile), frontal bossing, short neck, central and peripheral cyanosis, saddle nose, low set ears. The sutures were not separated. The thorax was narrow, cone shaped with small rib cage and abdomen was protuberant with the abdominal girth of 33 cm. All the limbs were small with deformed attitude (Lower segment: 14cm, Upper segment 22 cm and ratio was 1.57). Thighs and legs were bowed. Deep creases were present in all limbs (Figure 2,3).
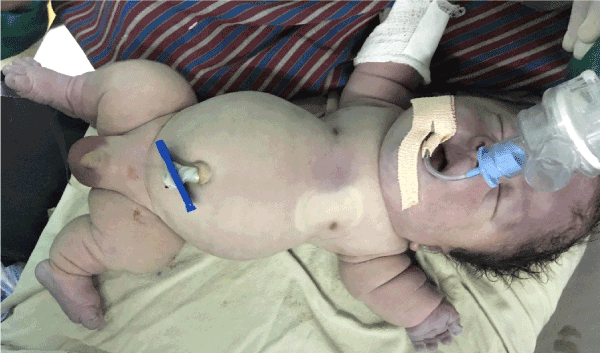
Figure 2. Showing micromelia, macrocephaly, narrow thorax and protruded abdomen
There was small pit/dimpling over the sacral region, cryptorchidism and flat back and spine (Figure 3). However there were no murmurs, passed meconium immediately after birth and umbilical cord has 2 arteries and one vein.
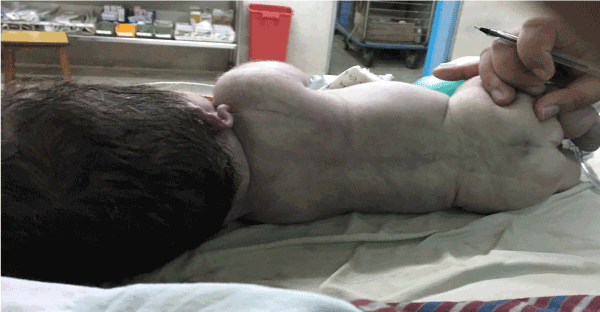
Figure 3. Showing flat back and spine with pit/dimpling over sacral region
TSD (also known as Thanatophoric Dysplasia) is most lethal, rare, sporadic birth defect due to de novo mutation in the FGFR3 gene [1-4] FGFR3 gene is located in chromosome 4p16.3, responsible for giving instructions for making a protein that is involved in the development and maintenance of bone and brain tissue [4]. The malformations due to bone growth seen in TSD is due to over activation of FGFR3 gene.5 The examination findings in this case reveals macrocephaly, narrow thorax associated with polyhydroaminos, micromelia, bowed thigh, frontal bossing, saddle nose, low set ears, protruding abdomen and flattening of spine. We found similar findings in different reported case of type I TSD [2-7]. The radiological and morphological features revealed in our report (Figure 1-4) confirms the diagnosis of Type I TSD [2-7]. Phenotypically TSD is of two clinically defined subtypes: Type I and Type II, the former being the more frequent (80%) [4].
The inheritance pattern of TSD is autosomal dominant but virtually all cases of TSD occur in people with no family history of TSD [6]. The reason being, no affected individuals are known to have had children; therefore, the disorder has not been passed to the next generation. As presented in our case, TSD usually leads to death in-utero or shortly after birth [5-10]. The reason for mortality in TSD is either due to reduced thoracic capacity, hypoplastic lung or brainstem compression [6-8].
Usually the diagnosis of TSD is made by USG during second trimester and further specific type is distinguished on later scans during third trimester with the help of fetal skeletal morphology.10 Further Diagnosis can be confirmed with autopsy and histopathology but unfortunately could not be done in present case as consent was not given by the parents [2-10].
The radiologic and morphologic features described in this report were compatible with TSD Type I. These findings helped us to correlate them with their pathogenesis and realize the reason why this disease usually has a poor prognosis. Proper counselling to the patient’s family is crucial for management and should be advised to undergo anomalies screening in subsequent pregnancies.
JNMA case report form was signed by the patient’s father and the original article is attached with the patient’s chart.
None.
- Warman ML, Cormier-Daire V, Hall C (2011) Nosology and classification of genetic skeletal disorders: 2010 revision. Am J Med Genet A 155: 943-968.
- Samsudeen MF, Maggonage CG, Wedisha IG, Thuvaratheepan R, Kaluarachchi A (2017) Fetal thanatophoric dysplasia. Sri Lanka journal of obstetrics and gynaecology.
- Manish S, Jyoti S, Rekha J, Devendra R (2015) Thanatophoric dysplasia: A case report. J Clin Diagn Res.
- Wilcox WR, Tavormina PL, Krakow D (1998) Molecular, radiologic, and histopathologic correlations in thanatophoric dysplasia. Am J Med Genetics 78: 274-281.
- Yuvaraj MF, Sankaran PK, Raghunath G, Begum Z, Kumaresan M (2017) Thanatophoric dysplasia; A rare case report on a congenital anomaly. Intern J of Pediat 5: 4227-4231.
- Naveen NS, Murlimanju BV, Kumar V, Pulakunta T, Jeeyar H (2011) Thanatophoric dysplasia: a rare entity. Oman Med J 26: 196-197.
- Jahan U, Sharma A, Gupta N, Gupta S, Usmani F, et al. (2019) Thanatophoric dysplasia: a case report. Int J Reprod Contracept Obstet Gynecol 8: 758-761.
- Faye-Peterson OM, Knisely AS (1991) Neural arch stenosis and spinal cord injury in thanatophoric dysplasia. Am J Dis Child 145: 87-89.
- Baker KM, Olson DS, Harding CO, Pauling RM (1997) Long term survival in typical thanatophoric dysplasia Type 1. Am J Med Genetics 70: 427-436.
- Wong HS, Kidd A, Zuccollo J (2008) A case of thanatophoric dysplasia: the early prenatal 2D and 3D sonographic findings and molecular confirmation of diagnosis. Fetal Diag Ther 24: 71-73.



