Abstract
Goals: Evaluate the root cause of periodontal disease, the predominance of bacteria and the total bacterial load before and after. Materials and Methods: Microbiological Tests: it consists in taking the crevicular fluid colonized by bacteria and containing epithelial cells of the person being examined, using a sterile paper cone with a diameter of 60/80 in the periodontal or peri-implant pocket for at least 30 seconds (so that it is soaked in liquid crevicular possibly without blood); it is placed inside the test tube; the procedure is repeated to obtain from a minimum of two to a maximum of four paper cones. The test was performed on 420 patients aged 30-60 years, 236 women, 184 men, 4-11mm pockets moderate-severe chronic periodontitis, for a total of 498 samples, in 78 patients a second sampling was carried out after treatment. Results: The study highlighted that the main cause of periodontal disease are parafunctions (Bruxism, clenching, bad habits, atypical swallowing, mouth breathing), dental misalignment, pre-contacts and incongruous prosthetic products. Therefore, as "direct and triggering" local etiological factors and no longer "indirect and predisposing" as has always been claimed. In the background by bacteria.
Keywords
Periodontal disease, Inflammation, Plaque, Tartar, Bacteria, Microbiological Analysis, Parafunctions
Introduction
The periodontal disease: The term periodontal disease (periodontitis) indicates a set of inflammatory pathologies, of an infectious nature, which are characterized at a clinical level by the pathological involvement of all the tissue components of the periodontal organ (gingiva, periodontal ligament, alveolar bone and root cementum) [1-3]. “Direct and triggering” local etiological factors: Bacterial (bacterial plaque, tartar, “alba” (“dawn”) material, food residues.
“Indirect and aggravating local” factors: Functional (occlusal trauma, parafunctions, bad habits, oral respiration, atypical swallowing, hypofunction).
"Indirect and predisposing" factors: Mechanical (wrong oral hygiene, presence of food). Anatomical (dental malposition, dental shape, shape of periodontal tissues).
"Indirect, predisposing and aggravating": Iatrogenic (overflowing fillings, incorrect prosthetic margins, oversized prosthetic crowns and orthodontic devices) [4,5].
The initiation and progression of periodontal disease are commonly attributed to pathogenic bacteria of the oral microbiota, mainly part of the red/orange complexes:
Aggregatibacter actinomycetemcomitans
Porphyromonas gingivalis
Tannerella forsythia
Treponema denticola
Fusobacterium nucleatum
Campylobacter rectus
Other factors including bad habits, anatomical and latrogenic, genetic and hereditary factors.
The aim of this study is to evaluate the main cause of periodontal disease, the predominant presence of bacteria in percentage and the total bacterial load before and after non-surgical therapy [6,7].
Materials and Methods
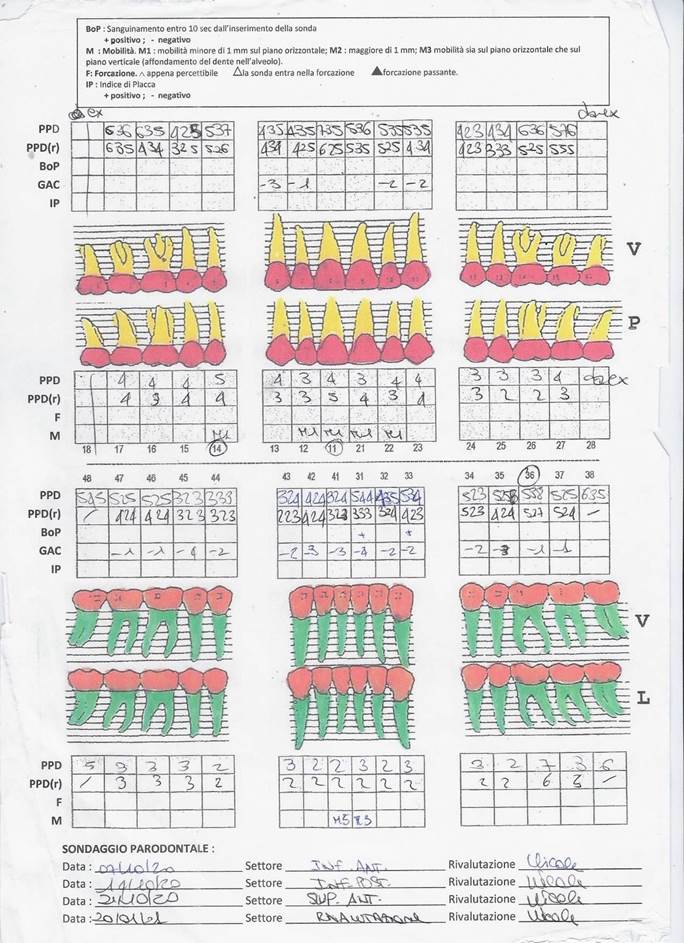
Microbiological Tests: it consists in taking the crevicular fluid colonized by bacteria and containing epithelial cells of the person being examined, using a sterile paper cone with a diameter of 60/80 in the periodontal or peri-implant pocket for at least 30 seconds (so that it is soaked in liquid crevicular possibly without blood); it is placed inside the test tube; the procedure is repeated to obtain from a minimum of two to a maximum of four paper cones. The test was performed on 420 patients, aged 30-60 years, 236 women, 184 men, 4-11mm pockets moderate-severe chronic periodontitis, for a total of 498 samples, in 78 patients a second sampling was carried out after treatment [6-9].
The patients had received no antibiotics or periodontal treatment during the past 6 months, no systemic disease. It follows the methodology, but differs primarily in the choice of patients, with specific characteristics (age, smokers 10/20 cigarettes a day, moderate- severe periodontitis, 4/11mm periodontal pockets) more responsive to ordinary people. Plaque index (IP), bleeding index (BoP), pocket depth (PPD), mobility (M), recession (GAC) was evaluated. All patients were provided with information about the study, having signed the informed consent [10-14].
Clinical Procedures: Microbiological testing was used for the study. The mesial area of the upper right first molar (1.6) and the mesial area of the lower left first molar (3.6) were selected. A group of 78 patients underwent a second sampling after treatment. (Scaling and root planning).
Microbiological Analysis Results
The following sample was analyzed: 498 withdrawals for a total of 420 patients.
In 78 patients a second sampling was carried out after the treatment, for a consideration of 15,7% of the total samples and 18,6% of the patients.
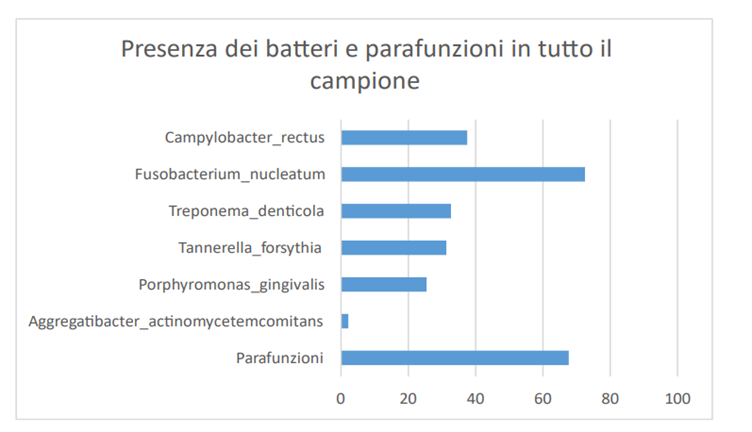
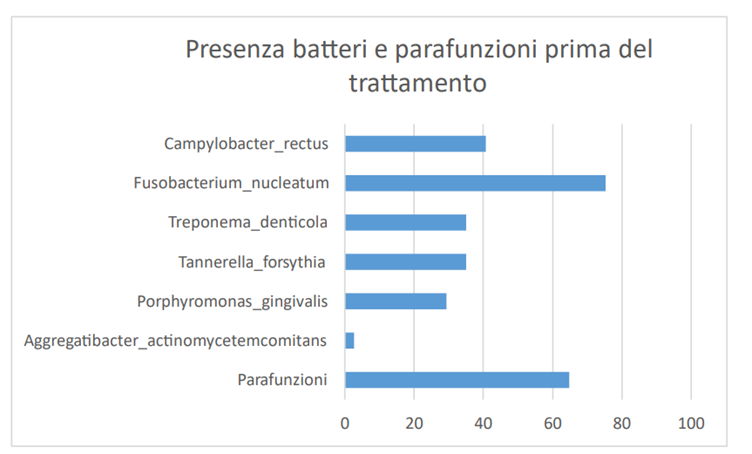
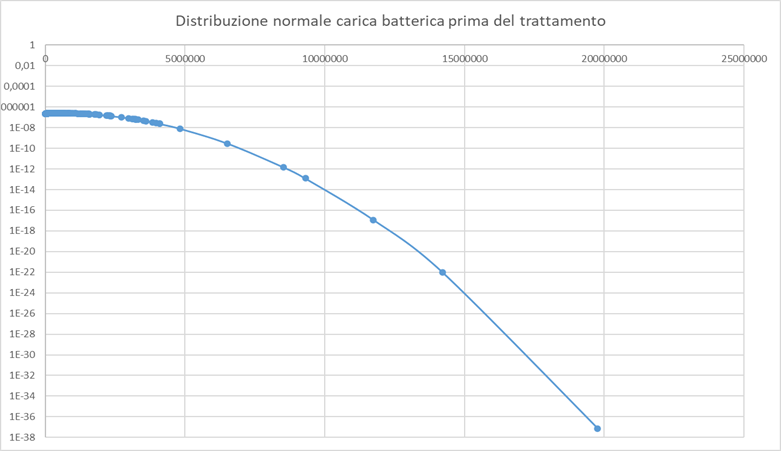
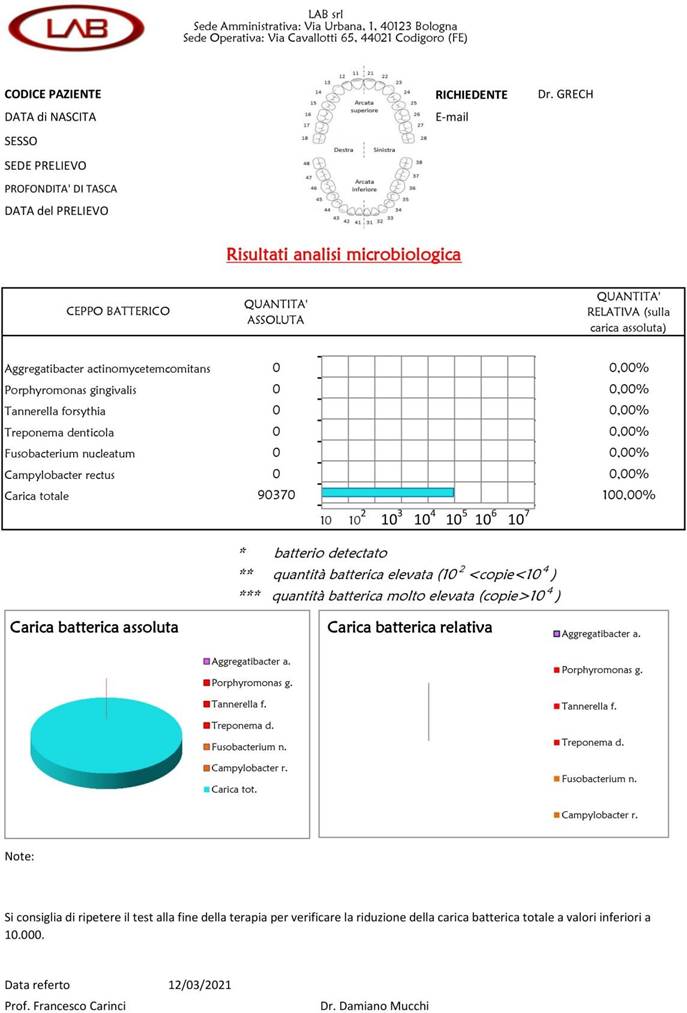
The bacterium Aggregatibacter actinomycetemcomitans is present in 2.2% of the samplings (11 cases out of 420) with values ranging from 0,0006% to 1,7875% of the total bacterial load, with an average value of 0,4995% before treatment (Graph 1).
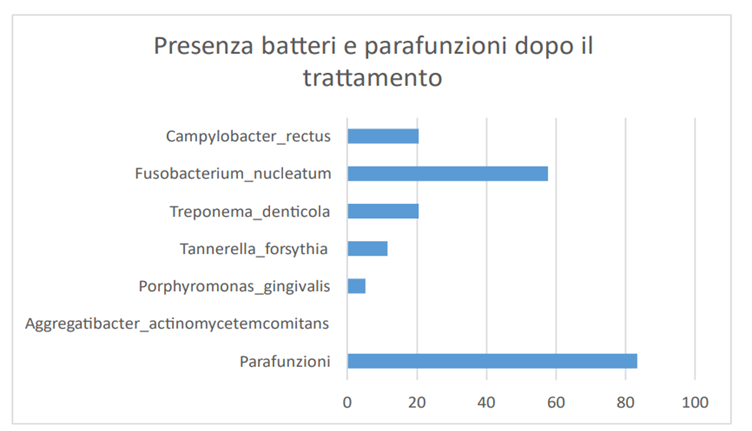
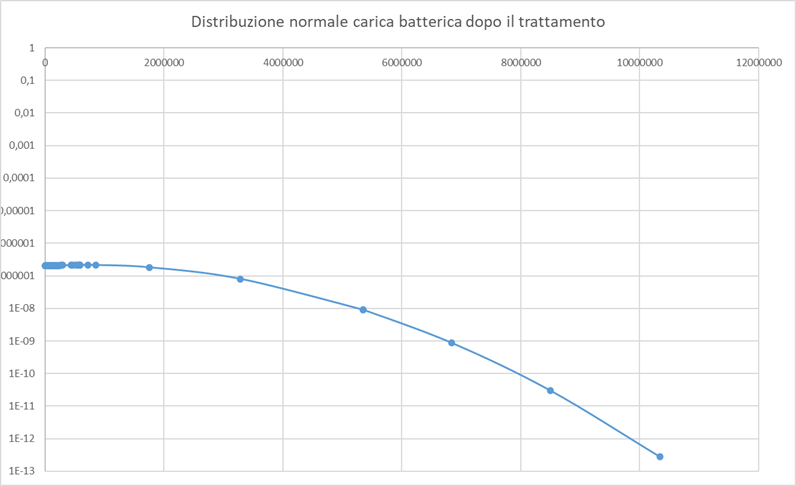
The bacterium Porphyromonas gingivalis is present in 25,5% of the samplings (127 cases out of 420) with values ranging from 0,0003% to 48,5433%, with an average value of 2,7917 on the total bacterial load. In the case of patients in which the measurement was made before treatment, the average percentage value is 2,8689, the average value after treatment drops to 0,4178% (Graph 2).
The Tannerella forsythia bacterium is present in 31,3% of the samplings (156 cases out of 420) with values ranging from 0,0004% to 15,1197% with an average percentage value of 1.1961 on the total bacterial load. In the case of patients in which the measurement was made before treatment, the average percentage value is 1,2331, the average value after treatment is 0,5912% (Graph 3).
The Treponema denticola bacterium is present in 32,7% of the samplings (163 cases out of 420) with values ranging from 0,0010% to 52,4201%, with an average percentage value of 1,5862 on the total bacterial load. In the case of patients in which the measurement was made before treatment, the average percentage value is 1,6668, the average value after treatment is 0,846% (Graph 4).
The Fusobacterium nucleatum bacterium is present in 72,5% of the samplings (361 cases out of 420) with values ranging from 0,0035% to 86,3505%, with an average value of 6,4628% of the total bacterial load. In the case of patients in which the measurement was made before treatment, the average percentage value is 7,0148, the average value after treatment is 2,5865 (Graph 5).
The Campylobacter rectus bacterium is present in 37,5% of the samplings (187 out of 420 cases) with values ranging from 0,0006% to 29,5476%, with an average value of 1,5387% of the total bacterial load. In the case of patients in which the measurement was made before treatment, the average percentage value is 1,4527, the average value after treatment rises to 2,4589 (Graph 6).
Discussion
The graphs have highlighted that after the treatment (Scaling and root planning) there is a drastic reduction (elimination of some bacteria) of the bacteria and of the total bacterial load. Before treatment the presence of Aggregatibacter actinomycetemcomitans, Tannerella forsythia, the percentage is very low and in many cases their absence, on the other hand we have a very high percentage of the bacterium Fusobacterium nucleatum also present in the intestine and causes colorectal cancer (studies have demonstrated a correlation between the bacterium present in the oral cavity and colorectal cancer).
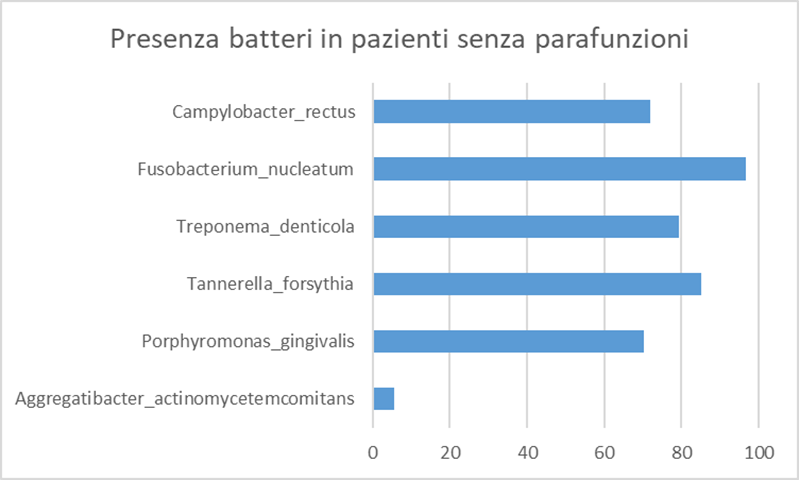
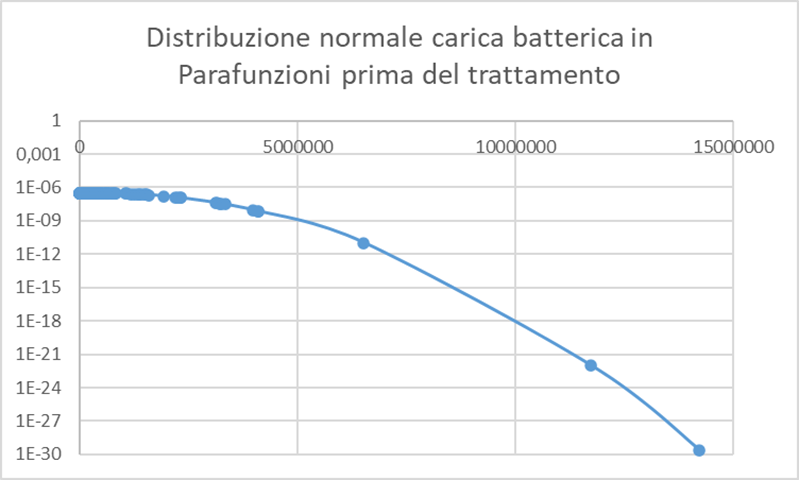
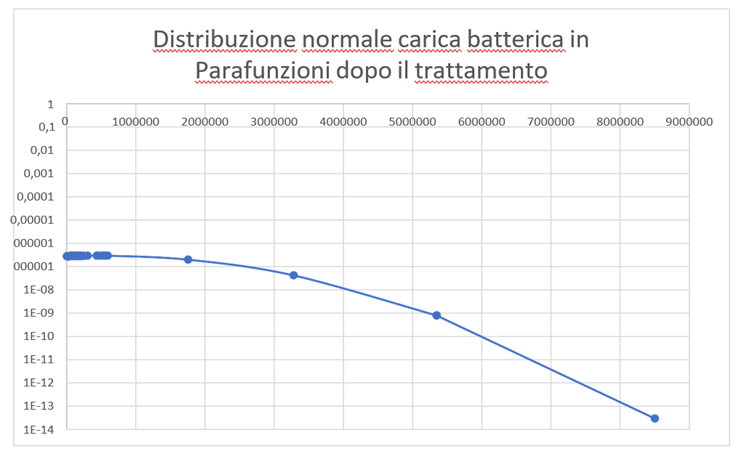
In graph 6, the Parafunctions are 70% (bruxism, clenching, atypical swallowing, precontacts, bad habits, dental misalignment, oral respirator and incongruous prosthetic products), with high bacterial load in some cases without main bacteria in other cases the almost constant presence of the bacterium Fusobacterium nucleatum, out of 420 patients 263 are with parafunctions 157 periodontal disease is attributed to bacteria.
Of these 263 patients (Chart 15)
N 60 are without bacteria
N 86 are without 5 bacteria
N 112 are without the first 4 bacteria
N 2 only have Aggregatibacter actinomycetemcomitans
N 13 have only Porphyromonas gingivalis
N 173 have Fusobacterium nucleatum
Results
The study highlighted that the main cause of periodontal disease are parafunctions (bruxism, clenching, bad habits, atypical swallowing, mouth breathing), dental misalignment, pre-contacts and incongruous prosthetic products. Therefore as "direct and triggering" local etiological factors and no longer "indirect and predisposing" as has always been claimed. In the background by bacteria. Before treatment the presence of Aggregatibacter actinomycetemcomitans, Porphyromonas gingivalis, Tannerella forsythia, the percentage is very low and in many cases their absence, on the other hand we have a very high percentage of the bacterium Fusobacterium nucleatum, also present in the intestine and causing colorectal cancer (studies have shown a correlation of the bacterium present in the oral cavity and colorectal cancer). After the treatment (Scaling and root planning) there is a drastic reduction (elimination of some bacteria) of the bacteria and of the total bacterial load [15-26].
Out of 420 patients 263 are with parafunctions 157 periodontal disease is attributed to bacteria.
Of these 263 patients (Chart 15)
N 60 are without bacteria
N 86 are without 5 bacteria
N 112 are without the first 4 bacteria
N 2 only have Aggregatibacter actinomycetemcomitans
N 13 have only Porphyromonas gingivalis
N 173 have Fusobacterium nucleatum
Out of 78 patients where a second sampling was carried out after treatment, 40% (31 patients) of the patients had Fusobacterium nucleatum always present.
Acknowledgement: I
thank Dr. Nicole Savioli, Dr. Laura Giuliani, the ASO manager Manuela Fuganti and all the staff of the "Dr Giuseppe Grech" team for the collaboration. I thank Dr. Caterina Giovinazzo for the technical-scientific support.
References
- American Academy of Periodontology (1989) Supportive treatment. In: Proceedings of the World Workshop in Clinical Periodontics: Chicago. Am Acad Periodontol 24.
- American Academy of Periodontology (1998) Position Paper. Supportive periodontal therapy (SPT). J Periodontol 69: 502-506. [Crossref]
- The American Academy of Periodontology, Position Paper (2003) Periodontal Maintenance. J Periodontol 74: 1395-1401.
- Löe H, Theilade E, Jensen SB (1965) Experimental gingivitis in man. J Periodontol 36: 177-187. [Crossref]
- Löe H, Anerud A, Boysen H, Morrison E (1986) Natural history of periodontal disease in man. Rapid, moderate, and no loss of attachment in Sri Lankan laborers 14 to 46 years of age. J Clin Periodontol 13: 431-445. [Crossref]
- Aass AM, Preus HR, Zambon JJ, Gjermo P (1994) Microbiologic tests in epidemiologic studies: are they reproducible? Scand J Dent Res 102: 355-360. [Crossref]
- Abrahamson M, Wikström M, Potempa J, Renvert S, Hall A (1997) Modification of cystatin C activity by bacterial proteinases and neutrophil elastase in periodontitis. Mol Pathol 50: 291-297. [Crossref]
- Abu-Amer Y, Ross FP, Edwards J, Teitelbaum SL (1997) Lipopolysaccharide-stimulated osteoclastogenesis is mediated by tumor necrosis factor via its P55 receptor. J Clin Invest 100: 1557-1565. [Crossref]
- Addy M, Langeroudi M, Hassan H (1985) The development and clinical use of acrylic strips containing anti-microbial agents in the management of chronic periodontitis. Int Dent J 35: 124-132. [Crossref]
- Adriaens PA (1989) Bacterial invasion in periodontitis, is it important in periodontal treatment? Rev Belge Med Dent 44: 9-30. [Crossref]
- Amsterdam M, Rossman SR (1960) Technique of hemisection of multirooted teeth. Alpha Omegan 54: 4.
- Amsterdam M (1974) Periodontal Prosthesis: Twenty-five years in retrospect. Alpha Omegan. [Crossref]
- Amsterdam M (1980) Periodontal Prosthesis. In Goldman H.M. Cohen, D.W. Periodontal Therapy.
- Beaudreau DE (1965) The role of the posterior fixed bridge in occlusion. Dent Clin North Am 13:24. [Crossref]
- Beaudreau DE (1980) Periodontal considerations in restorative dentistry. In Goldman H.M., Cohen D.W., Periodontal therapy ed. 6 St. Louis: Mosby 1116.
- Bower RC (1979) Furcation morphology relative to periodontal treatment. J Periodontol 50: 23-27. [Crossref]
- Box HK (1935) Experimental traumatogenic occlusion in sheep. Oral Health 25: 9.
- Carnevale G, Di Febo G (1981) Greter New York Academy of Prosthodontics. Personal Commun.
- Carnevale G, Freni SS (1982) Ist. International Congress of the Italian Society of Periodontology. Personal Commun.
- Carnevale G, Freni SS, Di Febo G (1983) Soft and hard tissue wound healing following tooth preparation to the alveolar crest. Int J Periodont Rest Dent 6: 36-53. [Crossref]
- McIlvanna E, Linden GJ, Craig SG, Lundy FT, James JA (2021) Fusobacterium nucleatum and oral cancer: a critical review. BMC Cancer 21: 1212. [Crossref]
- Gallimidi AB, Fischman S, Revach B, Bulvik R, Maliutina A, et al. (2015) The periodontal pathogens Porphyromonas gingivalis and Fusobacterium nucleatum promote tumor progression in an oral specific chemical carcinogenesis model. Oncotarget 6: 22613-22623. [Crossref]
- Fujiwara N, Kitamura N, Yoshida K, Yamamoto T, Ozaki K, et al. (2020) Involvement of Fusobacterium species in oral cancer progression: a literature review including other cancer types. Int J Mol Sci 21: 6207. [Crossref]
- Komiya Y, Shimomura Y, Higurashi T, Sugi Y, Arimoto J, et al. (2019) Colorectal cancer patients have identical strains of Fusobacterium nucleatum in colorectal cancer and oral cavity. Gut 68: 1335-1337. [Crossref]
- Abed J, Maalouf N, Manson AL, Earl AM, Parhi L, et al. (2020) Colon cancer - Associated Fusobacterium nucleatum can originate from the oral cavity and reach colon cancers via the circulatory system. Front Cell Infect Microbiol 10: 400. [Crossref]
- Tefiku U, Popovska M, Cana A, Zendeli-Bedxeti L, Recica B, et al. (2020) Determination of the role of Fusobacterium Nucleatum in the pathogenesis inside and outside the mouth. Pril 41: 87-99.