A Review of current research has revealed that all the diseases in human beings arise from one common cause which is Stress. Stress is the fundamental response of the body to various stressors. Chronic stress initiates stress response by activating innate and adaptive immunity. A cascade of events that comes into action requires energy. As a result cell metabolism increases just like when an acute stress develops, body increases its metabolism and utilizes, the nutrients present in the body by mobilizing macro- nutrients i.e., glucose, fat and protein, it also utilizes micro- nutrients, i.e., minerals and vitamins to facilitate fundamental reactions necessary for cell metabolism to occur.
This article reviews the consequences of chronic stress on health and role of energy and metabolism in the development of various diseases.
stress, oxidative stress, macro- nutrients, micro- nutrients, energy, cell metabolism
Abbreviations
HPA: Hypothalamus-pituitary-adrenal axis, SAM: Sympathetic-adrenal–medullary axis, ATP: Adenosine phosphate, P Cr: Phospho Creatine, FFA: Free Fatty Acids, TDEE: Total daily energy expenditure, BMR: Basal metabolic rate, REE: Resting energy expenditure, TCA: Tricarboxylic acid cycle, G3 P: Glyceraldehyde 3- phosphate, (1-3 BGP): 1-3 biphosphoglycerate, ADP: Adenine diphosphate, PEP: Phosphoenol pyruvate, FAD: Flavine adenine dinucleotide, NAD: Nicotinamide adenine dinucleotide, FADH: Flavine adenine dinucleotide hydroxide, NADH: Nicotinamide adenine dinucleotide hydroxide, ETS: Electron transport system, GTP: Gaunisone-5 triphosphate, NADPH: Nicotinamide adenine dinucleotide phosphate hydroxide, PARPS: Poly-ADP-ribose polymerases, DNA: Deoxy ribonucleic acid, RNA: Ribonucleic acid, PLP: Pyridoxal 5 phosphate, PPO: Pyridoxine 5 phosphate oxide, MTHFR: Methylene tetrahydrofolate reductase, TTP: Thianepyrophosphate, BCKDH: Alpha keto acid dehydrogenase, BCAA: Branch chain amino Acids, GABA: Gamma amino butyric acid, L Dopa: Dihydroxy phenyl alanine, RBP: Retinol binding protein, ROS: Reactive oxygen species, VDR: Vitamin D receptor, LDLs: Low density lipoproteins, MKS: Menaquinones, VKDPS: Vitamin K dependent proteins, Ca2+: Calcium ions, GRP: Gla rich protein, Gas 6: Gene 6 protein, PTH: Parathyroid hormone, Cr3: Trivalent Chromium O2: oxygen, H2O: Water, MCO: Multi copper oxidase, Fe2+: Ferrous ions, Fe3+: Ferric ions, Mg: Magnesium, cAMP: Cyclic adenosine phosphate, Mn: Manganese, Mn SOD: Manganese superoxide dismutase, PEPCK: Phosphoenol pyruvate carboxykinase, Mo Co: Molybedenum cofactor, mARC: Mitochondrial amodoxime reducing component, SEPP: Selenoprotein P, TRxR: Thiredoxin reductase, PDI: Protein disulfide isomerase, vitamin C: Ascorbic acid, vitamin E: alpha tocopherol, DIOs: Deiodonases, T4: Thyroxine, Cu Zn SOD: Copper Zinc superoxide dismutase, AGEs: Advanced glycation products, DRI: Dietary Reference Intake.
Human beings are being afflicted with medical illnesses at a rapid pace and various illnesses develop more often as multi morbidities. As described in my paper [1], all the diseases i.e., diabetes mellitus, hypertension, cardiovascular diseases, non alcoholic fatty liver disease, chronic kidney disease, depression, arthritis, chronic obstructive lung disease, or inflammatory bowel disease result from inflammation caused by chronic stress. Clinical presentation varies with timing and severity from one individual to another as exposure to stressors varies from person to person.
While there is continuous ongoing research to treat these illnesses, until now it has remained as management and symptomatic relief, and it is not always possible to achieve cure.
It is important to identify the etiology of the diseases, in order to prevent the occurrence and reduce the morbidity and mortality.
Therefore a thorough knowledge of human physiology is important to track when and how physiological processes in health begin to turn into pathological phenomenon resulting in disease.
I will review current literature on stress, human energy, metabolism and detailed role of micronutrients i.e., vitamins and minerals in next section.
In 1936, Hans Selye defined the term “Stress” as ‘the non-specific response of the body to any demand for change “and then redefined it as “rate of wear and tear on the body” [2]. To summarize the term stress used in Selyean tradition refers to response, whereas its original usage within the science of physics, it referred to stimulus and the term Strain referred to response. Using the term stress to denote a response, left Selye without a term to describe the stimulus that engenders a stress response. Selye chose the term stressor to denote any stimulus that serves to engender the stress response.
The author of The Anatomy and Physiology of the human stress response defines it as follows, Working definition of stress response is described as,“Stress is a physiological response that serves as a mechanism of mediation linking any stress to its target-organ-effect”. Term stressor is the stimulator for the stress response”. Stressor events fall in one of the two categories.
1. Psychosocial stressors are either real or imagined environmental events that set the stage for elicitation of the stress response. They can not directly cause the stress response but must work through cognitive appraisal mechanism. Most stressors are indeed psychosocial stressors.
2. Biogenic stressors however actually cause elicitation of the stress response. Such stimuli bypass the higher cognitive appraisal mechanisms and work directly on affective and neurological triggering nuclei. Thus by virtue of their biochemical properties they directly initiate the stress response without the usual requisite cognitive-affective processing. Examples of such stressors are, Ginseng, Ginko bioloba, Amphetamine, Caffeine, Nicotine, certain physical factors such as pain evoking stimuli, extreme heat and extreme cold [3].
Main focus therefore has to be on Stress. Chronic stress due to various stressors i.e., psychosocial, or biogenic (pathogens, toxins i.e., tobacco, alcohol, any molecule recognized as non self e.g. preservatives in foods, environmental toxins, pollen radiation) invokes stress response.
Stress response activates Hypothalamus –pituitary –adrenal (HPA) axis and Sympathetic–adrenal –medullary (SAM) axis, which activate innate immunity followed by adaptive immunity in chronic stress. In order to mount an effective response body requires energy, which is obtained by increasing the rate of cell metabolism. At this point I would review, the process of Human Energy and Cell Metabolism in health and discuss how it can play a role in development of disease.
Energy is the essence of life. Human being is able to function as a result of body’s ability to produce energy. Body is able to perform its physiological function due to availability of energy and regulates it according to changing conditions, i.e., less energy in basal conditions or more during physical activity and exercise. It responds to pathological conditions requiring more energy in similar manner.
How does body obtain energy?
The food we eat must be converted into energy form that body can use. Thus human body is equipped with a number of metabolic systems to produce and regulate for its diverse needs such as synthesis of tissues, movement of substances between tissues and muscular contraction.
The body uses the food we eat to provide energy, to build and repair tissues and to regulate metabolism. Of these three functions the human body ranks energy production first and will use food for this purpose at the expense of the other two functions at the time of need. A discussion of the role of nutrition as a means to help provide and control human energy is important from several stand points.
First inadequate supply of necessary energy nutrients such as muscle glycogen or blood glucose may cause fatigue. Fatigue may also be caused by the inability of the energy systems to function optimally because of a deficiency of other nutrients such as selected vitamins and minerals. In addition the human body is capable of storing energy reserves in a variety of body forms, including body fat and muscle tissue.
Just as energy is essence of life. Metabolism is life as it is through metabolism, the human being is able to convert stored energy from food into chemical energy and utilize it to sustain life. Human metabolism represents the sum total of all physical and chemical changes that take place within the body.
The transformation of food to energy, the formation of new compounds such as hormones and enzymes, the growth of bone and muscles tissue, the destruction of body tissues and a host of other physiological processes are part of the metabolic process.
What is energy ?
Energy represents the capacity to do work.. Work is one form of energy, often called mechanical energy.
Our body possesses stores of chemical energy that can be used to produce electrical energy for creation of electrical nerve impulses to produce heat to help keep our body temperature at 37c0 (98.6F0) even on cold days and to produce mechanical work through muscle shortening so that we may move about.
When humans consume plant or animal products, the carbohydrates, fat and proteins (all forms of stored chemical energy) they undergo a series of chemical changes and are utilized to develop body structure, to regulate body processes or to produce storage form of chemical energy. The optimal intake and output of energy is important to all individuals but especially for the active person. To perform to capacity, body energy stores must be used in the most efficient manner possible.
One of the basic purposes of body cells is to transform the chemical energy of these simple components into forms that may be available for immediate use or other forms that may be used in future.
The foods possess stored energy. When we consume these foods, the digestive processes break them down into simple compounds that are absorbed into the body and transported to various cells.
Energy in the body is available for immediate use in the form of adenosine triphosphate (ATP). It is a complex molecule constructed with high energy bonds, which when slit by enzyme action can release energy rapidly for a number of body processes including muscle contraction. ATP is classified as high energy compound and is stored in the tissues in small amounts. It is important to note that ATP is the immediate source of energy for all body functions and the other energy stores are used to replenish ATP at varying rates.
ATP may be formed from either carbohydrates, fat or protein, after these nutrients have undergone some complex biochemical changes in the body. Phospho Creatine (P Cr) is derived from excess ATP. It is another related high energy phosphate compound which is also found in the tissues in small amounts. Although it can not be used as an immediate source of energy, it can replenish ATP. Because ATP and P Cr are found in very small amounts in the body, they can be used up in matter of seconds, it is important to have adequate energy stores as a back up system. The body stores of carbohydrates, fat and protein can provide an individual with ample amounts of ATP enough to last for many weeks, even on a starvation diet.
It is important to note that parts of each energy nutrient may be converted to the other two nutrients in the body under certain circumstances. For example protein may be converted into carbohydrates during prolonged exercise, where as excess dietary carbohydrates may be converted into fat in the body during rest.
Carbohydrate is stored in limited amounts as blood glucose, liver glycogen and muscle glycogen. The largest amount of energy is stored in the body as fats. Fats are stored as triglycerides in both muscle tissue and adipose tissue. Triglycerides and free fatty acids (FFA) in the body are a limited supply. The protein of the body tissues is a large reservoir of energy but is not used under normal circumstances. The role of each of these macro nutrients during exercise is an important consideration The body stores energy in a variety of ways, ATP, P Cr, muscle glycogen and so on. In order for this energy to be used in basal conditions, or physical activity it must undergo certain biochemical reactions. These biochemical reactions are termed Metabolism.
What is metabolism ?
Metabolism is life. Metabolism involves two fundamental processes, anabolism and catabolism.
Anabolism is a building up process or constructive metabolism. Complex body components are synthesized from the basic nutrients. Energy is needed for anabolism to occur.
Catabolism is needed for anabolism to occur. Catabolism is the tearing down process. This involves the disintegration of body compounds into their simpler components. The breakdown of muscle glycogen to glucose and eventually carbon dioxide, water and energy is an example of catabolism process. The energy released from some catabolic processes is used to support the energy need of anabolism.
Metabolism represents human energy. The metabolic rate reflects how rapidly the body is using its energy stores and this rate can vary tremendously depending upon a number of factors.
For all practical purposes the total daily energy expenditure (TDEE) may be accounted for three factors.
Basal energy expenditure increases due to eating a meal and physical activity. Basal energy expenditure accounts for the largest component of TDEE, where as physical activity is the most variable. What factors account for the amount of energy expenditure during rest.
The body is continuously using energy to build up and tear down substances within the cells. Certain automatic body functions such as contraction of the heart, breathing, secretion of hormones and the constant activity of nervous system also are consuming energy. Basal metabolic rate (BMR), represents the energy requirements of the many different cellular and tissue processes that are necessary to continuing physiological activities in a resting post absorptive state, through out most of the day, other than sleeping, it is the lowest rate of energy expenditure. Although some of the energy released during oxidative processes at rest supports physiological functions such as pumping activity of the heart muscle, the majority of the energy is released as heat, a thermal effect that keeps our body temperature at about 98.6 F° (37°C), eating a meal and exercise are two other factors that induce a thermal effect.
Energy sources used during rest
The vast majority of the energy consumed during a resting situation is used to drive the automatic physiological processes in the body. Because the muscles expend little energy during rest, there is no need to produce ATP rapidly. Hence the Oxygen system is able to provide the necessary ATP for resting physiological processes. The oxygen system can use carbohydrates, fats and proteins as energy sources.
Protein is not used as a major energy source under normal dietary conditions. Carbohydrates and fats, when combined with oxygen in the cells are the major energy substrates during rest. Several factors may influence which of the two nutrients is predominantly used. In general though on a mixed diet of carbohydrates, fat and protein about 40% of the REE is derived from carbohydrates and about 60% comes from fat. However eating a diet rich in carbohydrates or fat will increase the percent of the REE derived respectively from carbohydrate and fat. Also when carbohydrate levels are low such as after an overnight fast, the percentage of the REE derived from fat increases.
Human energy metabolism during exercise
Any physical activity will raise metabolic activity above REE (Resting energy expenditure) and thus increase energy expenditure. Exercise is a stressor to the body and almost all body systems respond.
What nutrients are necessary for the operation of human energy system?
Although energy for formation of ATP is derived from the energy stores in carbohydrates, fat and some protein, this energy transformation and utilization would not occur without the participation of the other major nutrients –water,vitamins and minerals.
These three classes of nutrients function very closely with protein in the structure and function of numerous enzymes, many of which are active in the muscle-cell energy process.
Water is used to help keep up and transform such energy compounds by a process known as hydrolysis.
Several vitamins are needed for energy to be released from the cell sources. For example niacin serves an important function in glycolysis, thiamine is needed to convert glycolytic end products to acetyl Co A for entrance into Kreb cycle and riboflavin is essential to forming ATP through Kreb cycle and electron transport system. A number of other B vitamins are also involved in facets of energy transformation within the cell.
Minerals too are essential for cellular energy process. Iron is one of the more critical compounds. Aside from helping hemoglobin deliver oxygen to the muscle cell, it is also a component of myoglobin and the cytochrome part of the electron transport system. It is needed for proper utilization of oxygen within the cell itself.
Other minerals such as zinc, magnesium, potassium sodium and calcium are involved in a variety of ways, either as parts of active enzymes in energy storage or in the muscle contraction process.
Proper utilization of body energy stores requires attention not only to the major energy nutrients but also to the regulatory nutrients - water, vitamins and minerals [4].
Pathways for nutrient degradation that converge onto the TCA cycle.
Glycolysis
Glycolysis is the pathway in which one glucose molecule is degraded into two pyruvate molecules. Interestingly during the initial phase energy is consumed because two ATP molecules are used up, to activate glucose and fructose 6 phosphate. Part of the energy derived from the breakdown of phospho anhydride bond of ATP is conserved in the formation of phosphate ester bonds in glucose 6 phosphate and fructose 1, 6- phosphate. In the second part of glycolysis the majority of free energy obtained from the oxidation of the aldehyde group of glyceraldehyde 3- phosphate (G3P) is conserved in the acetyl phosphate group of 1-3 biphospho glycerate (1-3 BPG) which contains high free energy. Then part of potential energy of 1-3 BGP released during its conversion to 3-phospho glycerate is coupled to the phosphorylation of ADP to ATP. The second reaction where ATP synthesis occurs is the conversion of phosphoenol pyruvate (PEP) to pyruvate. PEP is a high energy compound due to its phospho ester bond and therefore the conversion reaction of PEP to pyruvate is coupled with ADP phosphorylation. This mechanism of ATP synthesis is called substrate level phosphorylation. For complete oxidation pyruvate molecules generated in glycolysis are transferred to the mitochondrial matrix to be converted into acetyl CoA in a reaction catalyzed by the multi enzyme complex pyruvate dehydrogenase (Figure 1).
Fatty acid oxidation pathway
The fatty acid oxidation pathway intersects the TCA cycle. In 1904, Knoop in a classic experiment, decisively showed that fatty acid oxidation was a process by which two carbon units were progressively removed from the carboxy end fatty acid molecule. The process consists of four reactions and generates acyl CoA and the acyl CoA molecule shortened by two carbons, with the concomitant reduction of FAD by enzyme acyl CoA dehydrogenase and NAD+ by beta hydroxy acyl CoA dehydrogenase. This pathway is known as Beta oxidation because the Beta carbon atom is oxidized prior to when the bond between carbons beta and alpha is cleaved. The four steps of beta oxidation are continuously repeated until the acyl CoA is entirely oxidized to acyl CoA which then enters the TCA cycle.
Amino acid transamination/deamination contributes to the TCA cycle
Two points must be considered regarding the use of amino acids as fuel in energy metabolism. The first is the presence of nitrogen in amino acid composition which must be removed before amino acid becomes metabolically useful. The other is that there are at least twenty different amino acids, each of which requires a different degradation pathway. For our purpose here it is important to mention two kinds of reactions involving amino acids: transamination and deamination.
In the first kind of reaction, the enzymes amino transferases convert amino acids to their respective alpha ketoacids by transferring the amino acid group of one amino acid to an alpha ketoacid. This reaction allows the amino acid to be interconverted.
The second kind of reaction deamination removes the amino acid group of the amino acid in the form of ammonia in the liver, The oxidative deamination of glutarate results in alpha ketoglutarate (a TCA cycle intermediate) and ammonia, which is converted into urea and excreted.
Deamination reaction in other organs form ammonia that is generally incorporated into glutarate to generate glutamine which is the main transfer of amino groups in blood. Hence all amino acids through transamination/deamination reaction can be converted into intermediates of TCA cycle, directly or via conversion to pyruvate or acetyl CoA.
Tri carboxylic cycle (Kreb citric acid cycle)
Oxidation of carbohydrates, protein and fats converge on the Tri carboxylic cycle (TCA).
Inter conversion of energy between reduced co enzymes and O2 directs ATP synthesis, but how and where are NADH and FADH2 reduced? In aerobic respiration all products of nutrients degradation converge to a central pathway in the metabolism, the TCA cycle. In this pathway acetyl group of Acetyl CoA resulting from the catabolism of glucose, fatty acids and some amino acids is completely oxidized to CO2 with concomitant reduction of electron transporting co enzymes (NADH and FADH2) consisting of eight reactions. The cycle starts with condensing acetyl CoA and oxaloacetate to generate citrate. The next seven reactions regenerate oxaloacetate and include four oxidation reactions in which energy is conserved with the reduction of NAD+ and FAD co enzymes to NADH and FADH2 whose electrons will then be transferred to O2 through ETS. In addition a GTP or an ATP molecule is directly formed as an example of substrate level phosphorylation. In this case the hydrolysis of the thioester bond of the succinyl CoA with concomitant enzyme phosphorylation is coupled to the transfer of enzyme bound phosphate group to GDP or ADP. Importantly although O2 does not directly participate in this pathway, the TCA cycle only operates in aerobic conditions because the oxidized NAD+ and FAD are regenerated only in ETS. Also noteworthy is that TCA cycle intermediates, may also be used as the precursors of different biosynthetic processes Figure 1.
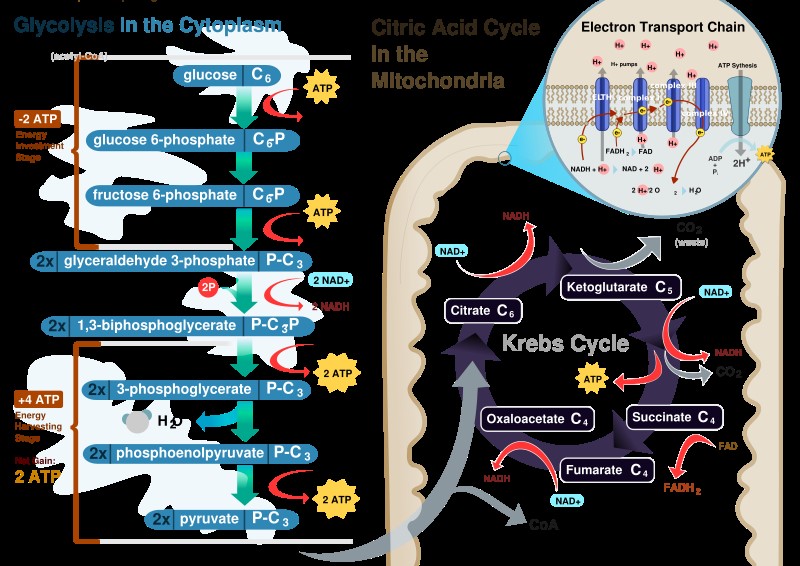
Figure 1. Source: human Physiology, Cell structure and Function.
Oxidative phosphorylation
This is the main mechanism of ATP synthesis in most human cells. The metabolic reactions are energy transducing processes in which oxidation-reduction reactions are vital for ATP synthesis. In these reactions the electrons released by oxidation of fuel molecules are transferred to two major electron carrier co enzymes, nicotinamide adenine dinucleotide (NAD+) and flavine adenine dinucleotide (FAD), that are converted to their reduced form NADH and FADH. Oxidative phosphorylation depends on the electron transport of NADH or FADH2 to O2 from water. The electrons are transferred through a number of protein complexes located in the inner mitochondrial membrane which contains chemical groups (flavin, iron – sulphur groups, heme and copper ions) capable of accepting or donating one or more electrons. The protein complexes known as the electron transport system (ETS) allow distribution of the free energy between the reduced co enzyme and the O2 and the more efficient energy conversion.
The electrons are transferred from NADH to O2 through three protein complexes. NADH dehydrogenase, cytochrome reductase and cytochrome oxidase. Electron transport between the complexes occurs through other mobile electron carriers, ubiquinone and cytochrome–c. FAD is linked to the enzyme succinate dehydrogenase of the TCA cycle and another enzyme acyl-CoA dehydrogenase of the fatty acid oxygen pathway. During the reaction catalyzed by three enzymes FAD is reduced to FADH2, these electrons are transferred to O2 through cytochrome reductase and cytochrome oxidase as described for NADH dehydrogenase electrons.
The electron transfer through the compounds of ETS is associated with proton (H+) pumping from the mitochondrial matrix to intermediate space of the mitochondria [5] (Figure 2).
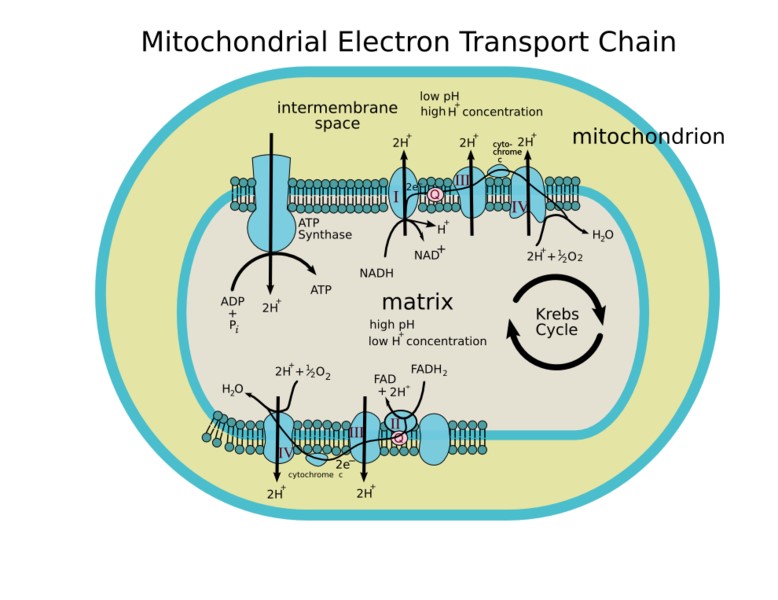
Figure 2. Source: Human Physiology, Cell structure and Function.
Regulation of citric acid cycle:
1. Substrate level
One of the contributing feature for any reaction is the availability of the various substrates involved in it. It is known that steady state concentration of the half lives of most substrates of the cycle are of the order of a few seconds.
2. Co Enzyme level
As a general rule, catabolic energy- yielding process generally requires NAD+, while anabolic energy requiring ones almost invariably require NADP+.
These two co-enzymes interact with one another according to following equation.
NADH + NADPi -> NAD+ + NADPH
<-
NAD (P) Trans hydrogenase
It is apparent that such a system would represent a sensitive control point for the regulation of the levels of these co enzymes. The relative concentration of NAD+ and NADH are important in regulating metabolic pathway. When NAD+/NADH ratio is high, the rate of citric acid cycle becomes rapid. However the activity of this cycle is retarded when NAD+/NADH ratio is low because of (a) insufficient NAD+ concentration for otherwise normal enzymatic function, and (B) re-oxidation of NADH coupled to ATP formation.
3. Enzyme levels
All mitochondria possess constant relative proportion of the various enzymes including characteristic dehydrogenases of the citric acid cycle. The observations suggest that the probability exists as genetic mechanism for the control of the synthesis of the integration of the key mitochondrial enzymes in the course of mitochondriogenesis.
4. Respiratory control
Respiratory rate depends not only on the nature and concentration of the substrate to be oxidized but also on the coupling of respiration to phosphorylation. Intact mitochondria are usually tightly coupled, so that their rate of respiration is actually controlled by the ratio (ADP/ATP). When this ratio is high respiration is promoted. (Stage 3) In contrast low ratios (i.e., high ATP concentration) decreases respiration because they bring about reverse electron reflux.
5. Accessibility of cycle intermediates
The activity of citric acid cycle is also controlled by its accessibility to Acetyl CoA of the intermediates of the cycle. This problem consists of a combination of permeability barriers and geometry.
The mitochondrial membrane itself provides a means for the admission of some substrates and exclusion of others.
6. Ketones
The accumulation of ketone bodies, aceto acetate and acetone formed by the liver in diabetes, results from the production of more acetyl CoA than can be cyclized via the kreb cycle or other synthetic reactions.
7. Control of enzyme activity
Some of the reactions in citric acid cycle require individual regulators because they are essentially irreversible under physiological conditions. There is always a danger from such reactions because they may continue till they consume available supply of substrate or co-enzyme. In general at many points stimulation or inhibition of the cycle is determined by the relative levels of NADH/NAD+, ATP/ADP, acetyl CoA/CoA or Succinyl CoA/CoA. When these ratios are high, the cell has ample supply of energy and flow through the cycle is slowed. When these ratios are low, the cell is in need of energy and flow through the cycle quickens [6].
Micronutrients i.e., vitamins and minerals are essential for life. In next section I will review their functions in general and special focus on their role in cell metabolism..
Niacin
Niacin and its derivative nicotinamide are dietary precursors of nicotinamide dinucleotide (NAD+) whichi can be phosphorylated (NADP) and reduced (NADH and NADPH). NAD+ functions in oxidation and reduction (redox) and non redox reaction.
Vitamin B3 (nicotinamide and nicotinic acid) is essential to all living cells. (journal of physiology).
The role of NAD+ metabolism in health and disease is of increased interest as the use of Niacin (Nicotinic acid) has emerged as a major therapy for treatment of hyperlipidemia and with the recognition that nicotinamide can protect tissues and NAD+ metabolism in a variety of diseased states ; including ischemia/reperfusion. In addition a growing body of evidence suggests the view that NAD+ metabolism regulates important biological effects including life span.
Niacin is a water soluble vitamin, which is also known as nicotinic acid and or vitamin B3. Its functions are as follows.
Oxidation-reduction (redox) reactions: Living organisms derive most of their energy from oxidation-reduction (redox) reactions, which are processes involving the transfer of electrons. Over 400 enzymes require Niacin co-enzymes NAD+ and NADP mainly to accept or donate electrons for redox reactions. NAD+ functions most often in energy producing reactions involving degradation (catabolism) of carbohydrates, fats and protein and alcohol. NADP functions more often in biosynthetic (anabolic reactions) such as in synthesis of all macromolecules including fatty acids and cholesterol.
Non redox reactions: The niacin co –enzyme NAD+ is substrate for at least four classes of enzymes that separate nicotinamide moiety from NAD+ and transfer ADP ribose to acceptors.
Mono-ADP-ribotransferase enzymes
In mammalian cells these enzymes transfer an ADP-ribose residue from NAD+ to a specific amino acid of a target protein, with creation of an ADP-ribosylated protein and the release of nicotinamide. Mono-ADP ribosylation reactions reversibly modify the activity of the acceptor protein such as G- proteins that bind gaunisone-5 triphosphate (GTP) and act as intermediate in a number of cell signaling pathways.
Poly-ADP-ribose polymerases (PARPS): These are enzymes that catalyze the transfer of polymers of ADP-ribose from NAD to accept proteins. PARPS appear to function in DNA repair and stress responses, cell signaling, transcription regulation, apoptosis, chromatin structure and cell differentiation, suggesting a role for NAD+ in cancer prevention.
ADP-ribosylcyclases: These catalyze the formation of cyclic ADP-ribose from ADP-ribose. Cyclic ADP-ribose works within cells to provoke the release of calcium ions from internal storage sites and probably also plays a role in cell signaling.
Sirtuins: Sirtuins are class of NAD+ dependent deacetylator enzymes that receive acetyl groups from the acetylated lysine residues of target proteins. During deacetylation process an ADP-ribose is added to the acetyl group to produce o- acetyl-ADPribose. Both acetylation and ADP-ribosylation are known post –translational modifications that affect protein activities.
To date spectrum of their biological functions include gene silencing, DNA damage repair, cell cycle regulation and cell differentiation 6.
Biotin
It is water soluble. and an essential co factor to enzymes in intermediary metabolism and a key regulator of gene expression. Biotin can not be synthesized by mammalian cells. It must be obtained by exogenous sources. Biotin functions as covalently bound co factor required for biological activity of the five known mammalian biotin dependent carboxylases 7.
Folate
Folate is critical in the metabolism of nucleic acid precursors and several amino acids as well as in methylation reactions. Folate is water soluble B-vitamin which is also known as Vitamin B 9.
One carbon metabolism: Function of folate co enzyme in the body appears to be in mediating the transfer of one carbon unit. Folate co enzymes act as acceptors and donors of one carbon units in a variety of reactions critical to the metabolism of nucleic acids and amino acids. Folate has an important metabolism interaction with riboflavin, which is a precursor of flavin adenine dinucleotide (FAD), a co enzyme required for activity of folate-metabolizing enzymes 5,10-methylene tetrahydrofolate. This active form of folate is required to form methionine from homocysteine.
Nucleic acid metabolism: Folate co enzymes play a vital role in DNA metabolism through two different pathways.
- Synthesis of DNA from its precursor thymidine and pines – is dependent on folate co enzymes.
- A folate co enzyme is required for the synthesis of methionine from homocysteine and methionine is required for the synthesis of S-adenyl-methionine (SAM). SAM is a methyl group (one carbon unit) donor—used in most methylation reactions, including the methylation of a number of sites within DNA, RNA, proteins and phospholipids. The methylation of DNA plays a role in controlling gene expression and is critical during cell differentiation. Abberations in DNA methylation have been linked to the development of cancer.
3. Amino acid metabolism
Folate co enzymes are required for the metabolism of several important amino acids namely methionine, cysteine, serene, glycine and histidine. The synthesis of methionine from homocysteine is catalyzed by methionine synthase an enzyme that requires not only folate but also vitamin B12. Concentration of homocysteine in the blood is regulated by 3 B vitamins, folate, B12 and B6.
Pantothenic acid
Pantothenic acid also known as Vitamin B5 is essential to all forms of life. Pantothenic acid is found in all tracks of life in the form of Coenzyme A, a vital co enzyme in numerous chemical reactions. It is a water soluble vitamin that is a precursor in the synthesis of CoenzymeA. CoenzymeA is essential to many biochemical reactions that sustain life. Also the phospho pantetheinyl moiety of Coenzyme is required for biological activity of several proteins including the acyl-carrier protein involved in fatty acid synthesis [7].
Riboflavin
It is a water soluble vitamin, also known as Vitamin B2. In the body, riboflavin is primarily found as an integral component of the co enzymes, flavin adenine dinucleotide (FAD) and flavin mono nucleotide (FMN). Coenzymes derived from riboflavin are termed flavo coenzymes and enzymes that use flavo coenzymes are called flavo proteins. Its functions are as follows, Oxidation-reduction (redox) reactions.
Flavo coenzymes participate in redox reactions in numerous metabolic pathways. They are critical for metabolism of carbohydrates, lipids and proteins. FAD is part of the electron transport (respiratory ) chain, which is central to energy production. In conjunction with cytochrome- P-450 flavo co enzymes also participate in the metabolism of drugs and toxins.
Anti oxidant functions
Glutathione reductase is a FAD dependent enzyme that participates in the redox cycle of glutathione. The glutathione redox cycle plays a major role in protecting organism from reactive oxygen species, such as hydroperoxide. Riboflavin deficiency has been associated with increased oxidative stress.
Glutathione peroxidase, selenium containing enzymes require two molecules of reduced glutathione to break down hydroperoxides. GPx are involved in the glutathione oxidation-reduction (redox) cycle.
Xanthine –oxidase
It is another FAD dependent enzyme which catalyzes the oxidation of hypoxanthine and xanthine to uric acid. Uric acid is one of the most effective water soluble anti oxidants in the blood. Riboflavin deficiency can result in decreased xanthine oxidase activity, reducing blood uric acid levels.
Nutrient actions of B-Complex vitamins
Flavoproteins are involved in the metabolism of several other vitamins (Vit B 6, niacin, and folate). Therefore severe riboflavin deficiency may effect many enzyme systems.
Conversion of most naturally available Vit B6, to its co enzyme from pyridoxal 5’ phosphate (PLP) requires the FMN dependent enzyme, pyridoxine 5’ phosphate oxidase (PPO). The synthesis of niacin containing co enzymes NAD+ and NADP from the amino acid tryptophan requires the FAD dependent enzyme kynurenine mono oxygenase. Severe riboflavin deficiency can decrease conversion of tryptophan to NAD+ and NADP increasing the risk of niacin deficiency.
5,10 methylenetetrahydrofolate reductase (MTHFR) is an FAD-dependent enzyme that plays an important role in maintaining the specific folate co enzyme required to form methionine from homocysteine.
Iron
Riboflavin deficiency alters iron metabolism. In humans improving riboflavin nutritional status has been found to increase hemoglobin levels [7].
Thiamine
It is a water soluble B Vitamin, B1. Thiamine pyrophosphate (TTP), the active form of thiamine is involved in several enzyme functions associated with metabolism of carbohydrates, branch chain amino acids and fatty acids. Its functions are as follows, Coenzyme function.
The synthesis of TTP from free thiamine requires magnesium, adenosine triphosphate (ATP), and the enzyme thiamine pyrophospho kinase. TPP is required as coenzyme for four multi component enzyme complexes associated with the metabolism of carbohydrates and branch chain amino acids. Pyruvate dehydrogenase, alpha ketoglutarate dehydrogenase and branch chain alpha keto acid dehydrogenase (BCKDH) each comprise a different enzyme complex found within cellular organelles called mitochondria. They catalyze the decarboxylation of pyruvate, alpha ketoglutarate and branch chain amino acids to form acetyl Coenzyme A (Co, succinyl CoA) and derivatives of branch chain amino acids respectively. All products play a critical role in the production of energy from food through their connection to the citric acid cycle. Branch chain amino acids (BCAA), including leucine, iso leucine and valine are eventually degraded into Acetyl-Co A to fuel the citric acid cycle. The catabolism of the these BCAA, also contributes to the production of cholesterol and donates nitrogen for the synthesis of the neurotransmitters, glutamate and gama aminobutyric acid (GABA).
In addition to thiamine co enzyme (TPP) each dehydrogenase complex requires a niacin containing co enzyme (NAD+), a riboflavin containing enzyme (FAD) and lipoic acid.
Transketolase catalyzes critical reactions in another metabolic pathway occurring in cytosol, known as pentose phosphate pathway. One of the most important intermediates of this pathway is ribose-5 phosphate, a phosphorylated 5-carbon sugar required for the synthesis of high energy ribonucleotides, such as ATP and gaunosine triphosphate (GTP). Nucleotides are the building blocks of nucleic acids DNA and RNA. The pentose phosphate pathway also supplies various anabolic pathways including fatty acid synthesis with the niacin containing co enzyme NADPH, which is essential for a number of biosynthetic reactions [7].
Vitamin B6
Vitamin B6 is a water soluble vitamin. and its derivatives are essential to over 100 enzymes mostly involved in protein metabolism.
Growing evidence from experimental and clinical studies suggest that systemic inflammation underlying most chronic diseases may impair Vitamin B6 metabolism.
Many biochemical reactions catalyzed by PLP-dependent enzymes are involved in essential biological processes such as hemoglobin and amino acid synthesis as well as fatty acid metabolism.
PLP also functions as a co enzyme for glycogen phosphorylase, an enzyme that catalyzes the release of glucose from stored glycogen. Much of PLP in human body is found in muscle bound to glycogen phosphorylase. PLP is also a co enzyme for reactions that generate glucose from amino acids, a process called gluconeogenesis.
Nervous system function: In the brain PLP dependent enzyme aromatic L-amino acid decarboxylase catalyzes the synthesis of two major neurotransmitters:
Serotonin from the amino acid tryptophan and dopamine from L-3,4 dihydroxy phenyl alanine (L_Dopa). Other neurotransmitters including glycine D-Serine glutamate, histamine and gamma –amino butyric acid (GABA) are also synthesized in reactions catalyzed by PLP dependent enzymes.
Hemoglobin synthesis: PLP functions as a co enzyme of 5 aminolevulinic acid synthase, which is involved in the synthesis of heme, an iron containing compound of hemoglobin. Hemoglobin is found in red blood cells and is critical for their ability to transport oxygen throughout the body. Both pyridoxal and PLP are able to bind to the hemoglobin molecule and affect its ability to pick up and release oxygen.
Tryptophan metabolism: Deficiency in another B vitamin niacin is easily prevented by adequate dietary intake. The dietary requirement of niacin and the niacin co enzymes, nicotinamide adenine dineucleotide (NAD) can be also met though to a fairly limited extent by the catabolism of the essential amino acid tryptophan in the tryptophan kynurenine pathway. Key reaction in this pathway are PLP dependen.
Hormone function: Nucleic acid synthesis [7].
Vitamin B12
Vitamin B12 has the largest and most complex chemical structure of all the vitamins. It plays an important, essential role in folate metabolism and in the synthesis of the citric acid cycle intermediate succinyl CoA.
The preservation of DNA integrity is dependent on folate and vitamin B12 availability. Vitamin B12 is essential for preservation of myelin sheath and neurons and for the synthesis of neurotransmitters. Both depression and osteoporosis have been linked to Vit B12 status and high homocysteine levels. Its functions are as follows,
Cofactor for methionine synthesis: Methyl cobalamin is required for the function of the folate-dependent enzyme, methionine synthase.. This enzyme is required for the synthesis of the amino acid methioninen from homcysteine. Methionine is in turn required for the synthesis of S-adenosyl-methionine a methyl group donor used in many biological methylation reactions including the methylation of a number of sites within DNA, RNA, and proteins. Abberent methylation of DNA and proteins which causes alterations in chromatin structure and gene expression are a common feature of cancer cells. Inadequate function of methionine synthase can lead to an accumulation of homocysteine which has been associated with increased risk of cardiovascular diseases.
Cofactor for L-methyl-co enzyme A mutase: 5-Deoxyadenosyl cobalan is required by the enzyme that catalyzes the conversion of L-methyl- malonyl –Coenzyme a to succinyl Coenzyme A
(succinyl CoA) which then enters the citric acid cycle. Succinyl CoA plays an important role in the production of energy from lipids and proteins and is also required for the synthesis of hemoglobin, the oxygen carrying pigment in red blood cells [7].
Vitamin A is a generic term that refers to fat- soluble compounds found as preformed vitamin A (retinol) in animal products. and as pro vitamin A caroteinoids in fruits and vegetables.
Vitamin A is involved in regulating the growth and specialization (differentiation) of virtually all cells in the human body.
Vitamin A has important role in embryonic development, organ formation during fetal development, normal immune function and eye development and vision.
Vitamin A compounds are essential fat soluble molecules. Their functions are as follow.
Vitamin A has important role in visual system and eye sight. It is required for normal function of retina, dim light vision and color vision.
Regulation of gene expression
Regulatory capacity of retinoic acid: By regulating the expression of over 500 retinoid-responsive genes, retinoic acid isomers play major roles in cellular proliferation and differentiation.
Immunity: It plays important role in the normal functioning of the immune system. There is also substantial evidence to suggest that RA may help prevent the development of autoimmunity.
Red blood cell production.
Zinc
Zinc deficiency is thought to interfere with vitamin A metabolism in several ways.
- Zinc deficiency results in decreased synthesis of retinol-binding protein (RBP) which transports retinol through the circulation to peripheral tissues and protects the organism against potential toxicity of retinol.
- Zinc deficiency results in decreased activity of the enzyme that releases retinol from its storage form, retinol palmitate in the liver.
- Zinc is required for the enzyme that converts retinol into retinal.
Iron. Vitamin A deficiency often co-exists with iron deficiency and may exacerbate iron deficiency anemia by altering iron metabolism [7].
Vitamin C
Vitamin C also known as ascorbic acid is a water-soluble vitamin. Humans do not have the ability to make ascorbic acid and must obtain vitamin C from the diet.
It functions as an essential cofactor in numerous enzymatic reactions e.g. in the biosynthesis of collagen, carnitine, and catecholamines, and as a potent antioxidant. Its functions are as follows.
Vitamin C is a potent reducing agent, meaning that it readily donates electrons to recipient molecules. Related to this oxidation-reduction (redox) potential, there are two major functions of vitamin C as an antioxidant and as an enzyme cofactor.
2021 Copyright OAT. All rights reserv
Vitamin C is the primary water-soluble, non enzymatic antioxidant in plasma and tissues. Even in small amounts vitamin C can protect indispensible molecules in the body such as proteins, lipids (fats), carbohydrates and nucleic acids (DNA and RNA) from damage by free radicals, and reactive oxygen species (ROS), that are generated during normal metabolism, by active immune cells and through exposure to toxins and pollutants. (e.g. certain chemotherapy drugs and cigarette smoke). Vitamin C also participates in redox recycling of other important anti oxidants ; for example is known to regulate vitamin E from its oxidized form.
Vitamin C’s role as a cofactor is also related to its redox potential. By maintaining enzyme bound metals in their reduced form, vitamin C assists mixed function oxidases in the synthesis of several critical biomolecules.Symptoms of vitamin C deficiency such as poor wound healing and lethargy result from impairment of these enzymatic reactions and insufficient collagen, carnitine, and catecholamine synthesis. Research suggests that vitamin C is involved in the metabolism of cholesterol and bile acids, which may have implications for blood cholesterol levels and the incidence of gallstones. Finally vitamin C increases the bioavailability of iron from foods by intestinal absorption of non –heme iron [7].
Vitamin D
Vitamin D can be synthesized in the skin upon exposure to sunlight and is then metabolized in the liver and kidney to the metabolically active form called1, 25-dihdroxyvitamin D. Through binding to the vitamin D receptor (VDR), 1, 25 –dihydroxyvitamin D can regulate the expression of hundreds of genes involved in skeletal muscle and other biological functions.
Vitamin D is essential for maintenance of bone mineralization through the regulation of calcium and phosphorus homeostasis, vitamin D also exhibits many non-skeletal effects particularly on the immune, endocrine and cardiovascular systems.
Its functions are as follows.
Calcium balance: Maintenance of serum calcium levels within a narrow range is vital for normal functioning of nervous system, as well as for bone growth and maintenance of bone density. Vitamin D is essential for the efficient utilization of calcium by the body.
Phosphorus balance: The regulation of calcium and phosphorus homeostasis are closely related and the calcitropic hormones PTH and 1,25 –dihydroxyvitamin D can also control serum phosphorus. Specifically 1,25-dihydroxyvitamin D increases intestinal phosphorus absorption by stimulating the expression of sodium-phosphate co transporter in the small intestine.
Cell differentiation: Cells that are dividing rapidly are said to be proliferating. Differentiation results in the specialization of cells for specific functions. In general differentiation of cells leads to decrease in proliferation, while cellular proliferation is essential for growth and wound healing, uncontrolled proliferation of cells with certain mutations may lead to cancer. The active form of vitamin D,1,25 –dihydroxyvitamin D inhibits proliferation and stimulates the differentiation of cells through binding to the VDR.
Immunity: Acting through the VDR, 1,25- dihdroxyvitamin D is a potent immune system modulator. The VDR is expressed by most cells of the immune system, including regulatory T cells and antigen –presenting cells such as dendritic cells and macrophages. Under specific circumstances, monocytes, macrophages and T cells can express the 25-hydroxyvitamin D3- alpha hydroxylase enzyme and produce 1,25-dihydroxyvitamin D, which acts locally to regulate the immune response. There is considerable scientific evidence that 1, 25 –dihdroxyvitamin D has a variety of effects on immune system function which may enhance innate immunity and inhibit the development of auto-immunity. Conversely Vitamin D deficiency may compromise the integrity of the immune system and lead to inappropriate immune responses.
Insulin secretion: The VDR is expressed by insulin-secreting cells of the pancreas and the results of animal studies suggest that 1, 25-dihydroxyvitamin D plays a role in insulin secretion under conditions of increased insulin demand. Cross –sectional and prospective studies suggest that insufficient vitamin D levels may have an adverse effect on insulin secretion and glucose tolerance in type 2 diabetes [7].
Vitamin E
Naturally occurring vitamin E includes eight fat-soluble isoforms. Body preferably uses alpha-tocopherol and only alpha tocopherol supplements can reverse vitamin E deficiency symptoms.
Its functions are as follows.
Antioxidant activity: The main function of alpha-tocopherol in humans is that of a fat-soluble antioxidant. Fats which are an integral part of all cell membranes are vulnerable to damage through lipid peroxidation by free radicals. alpha –tocopherol is uniquely suited to intercept per oxyl radicals and thus prevent a chain reaction of lipid oxidation. When a molecule of alpha-tocopherol neutralizes a free radical, it is oxidized and its antioxidant capacity is lost.. Other antioxidants such as vitamin C, are capable of regenerating the antioxidant capacity of alpha –tocopherol.
Aside from maintaining the integrity of cell membranes throughout the body, alpha-tocopherol protects the fats in low-density lipoprotein (LDLs) from oxidation. Lipoproteins are particles composed of lipids and proteins that transport fats through the blood stream. LDLs specifically transport cholesterol from the liver to the tissues of the body. Oxidized LDL have been implicated in the development of cardiovascular diseases.
Effect on cell mediated immunity: Other functions of alpha –tocopherol are likely to be related to its antioxidant capacity. For instance, alpha-tocopherol can protect the physiological properties of lipid bilayer membranes and may influence the activity of membrane proteins and enzymes. In cell culture studies alpha-tocopherol was found to improve the formation of an adhesive junction (known as immune synapse) between naïve T lymphocytes and antigen-presenting cells (APC), which eventually promoted T cell activation and proliferation [7].
Vitamin K
Naturally occurring vitamin K forms include phylloquinone (vit K 1) and a family of molecules called menaquinones (MKS or vit K2).
Vitamin K is essential cofactor for the carboxylation of glutamic acid residues in many vitamin K dependent proteins (VKDPs) that are involved in blood coagulation, bone metabolism, prevention of vessel mineralization and regulation of various cellular functions.
Vitamin K is a fat-soluble vitamin. Its functions are as follows.
Vitamin K oxidation-reduction cycle.
Although vitamin K is a fat-soluble vitamin, the body stores very small amounts that are rapidly depleted without regular dietary intake. The body recycles it through a process called the vitamin K epoxide cycle.
Coagulation (clotting): The ability to bind calcium ions (Ca2+) is required for the activity of the several vitamin-dependent clotting factors or proteins, in the coagulation cascade. The term coagulation cascade refers to a series of events, each dependent on the other, that stop bleeding through clot formation. Vitamin K dependent gamma –carboxylation of specific glutamic acid residues in those proteins makes it possible for them to bind calcium.
Skeletal formation and prevention of soft tissue calcification: Vitamin K dependent gamma carboxylation is essential to several bone related proteins, including osteocalcin anticoagulation factor, protein S, matrix y carboxylated glutamate (Gla) protein (MGP) GLa rich protein (GRP) and periostin. Osteocalcin is synthesized by osteoblasts; the synthesis of osteocalcin is regulated by active form of vitamin D. The calcin binding capacity of osteocalcin requires vit K-dependent y –carboxylation of three glutamic acid residues.
Regulation of cellular functions: Growth arrest-specific gene 6 protein (Gas 6) is a vitamin K dependent protein. It appears to be a cellular growth regulation factor with cell signaling activity. Gas 6 has been involved in diverse cellular functions, including phagocytosis cell adherence cell proliferation and protection against apoptosis. Gas 6 appears to regulate platelet signaling and vascular hemostasis [7].
Calcium
Calcium is a major constituent of bones and teeth. and also plays an essential role as a second messenger in cell signaling pathways. Circulating calcium concentrations are tightly controlled by the parathyroid hormone (PTH) and vitamin D at the expense of the skeleton, when dietary calcium intakes are inadequate. Its functions are as follows,
Cell signaling: Calcium plays a role in mediating the constriction and relaxation of blood vessels, nerve impulse transmission, muscle contraction and secretion of hormones like insulin. Excitable cells, such as skeletal muscle and nerve cells contain voltage –dependent calcium channels in their cell membranes that allow for rapid changes in calcium concentration.
For example when a nerve impulse stimulates a muscle fiber to contract, calcium channels in the cell membranes open to allow calcium ions into the muscle cells. Within the cells, the calcium ions bind to activator proteins, which help release a flood of calcium ions from storage vesicles of the endoplasmic reticulum (ER) inside the cell. The binding of calcium to the protein troponin-c initiates a series of steps that lead to muscle contraction.. The binding of calcium to the protein calmodulin activates enzymes that break down muscle glycogen to provide energy for muscle contraction. Upon completion of the action, calcium is pumped outside the cell into ER until the next activity.
Regulation of protein function: Calcium is necessary to stabilize a number of proteins, including enzymes, optimizing their activities. The binding of calcium ions is regulated for the activation of the seven vitamin K dependent clotting factors in the coagulation cascade 7.
Chromium
Chromium (Cr0) is an ubiquitous trace metal. The most stable oxidation state of chromium in biological systems is trivalent chromium (Cr3), which forms relatively inert complexes with proteins and nucleic acids. Its functions are as follows.
Trivalent chromium has been proposed to be the cofactor for a biologically active molecule that could enhance the effects of insulin on target tissues 7.
Copper
Copper is essential cofactor for oxidation –reduction reactions involving copper containing oxidases. Copper enzymes regulate various physiologic pathways such as energy production, iron metabolism, connective tissue maturation and neurotransmission.
Copper is a critical functional component of several essential enzymes known as cupro enzymes. Its functions are as follows.
Energy production: The copper dependent enzyme cytochrome- c oxidase plays a role in cellular enzyme production. By catalyzing the reduction of molecular oxygen (O2) to water (H2O), cytochrome oxidase generates an electrical gradient used by mitochondria to create the vital energy storing molecule ATP.
Connective tissue formation: Another cupro enzyme, lysyl oxidase, is required for the cross-linking of collagen and elastins, which are essential for the formation of strong and flexible connective tissue. The action of lysyl oxidase helps maintain the integrity of connective tissue in the heart and blood vessels and also plays a role in the bone formation.
Iron metabolism: Four copper containing enzymes, known as multi-copper oxidase (MCO) or ferro oxidases have the capacity to oxidize ferrous iron (Fe2+) ferric iron (Fe3+), the form of iron that can be located onto the protein transferrin for transport to the site of red blood cell function. The MCO family comprises the circulating ceruloplasmin (which represents-90% of plasma copper), the membrane bound ceruloplasmin (called GPI-ceruloplasmin) and two proteins called Hephaestin and zyklopen, found in the intestine and the placenta respectively.
Central nervous system: A number of reactions essential to normal function of the brain and nervous system are catalyzed by cupro enzymes.
Neuro transmitters synthesis: Dopamine beta hydroxylase catalyzes the conversion of dopamine to the neurotransmitter, nor epinephrine formation and maintenance of myelin. The myelin sheath is made of phospholipids whose synthesis depends on cytochrome-c oxidase activity.
Melanin formation: The cuperoenzyme, tyrosine is required for the formation of the pigment melanin. Melanin is formed in cells called, melanocytes and plays a role in the pigmentation of the hair, skin and eyes..
Antioxidant functions: Ceruloplasmin may function as an antioxidant in two different ways. Free copper and iron ions are powerful catalysts of free radical damage by binding copper. Ceruloplasmin prevents free copper ions from catalyzing oxidative damage. The ferro oxidative activity of ceruloplasmin (0xidation of ferrous iron facilitates iron loading onto its transport protein, transferrin, and may prevent free ferrous ions (Fe2+) from participating in harmful free-radical generating reactions.
Regulation of gene expression: Cellular copper levels may affect the synthesis of proteins by enhancing or inhibiting the transcription of specific genes. Copper may regulate the expression of genes by increasing the levels of intracellular oxidative stress. A number of signal transduction pathways are activated in response to oxidative stress and can lead to an increase in the expression of genes involved in detoxification of reactive oxygen species [7].
Iodine
Iodine is a key compound of thyroid hormone, which are required throughout life for normal growth, neurological development and metabolism. Iodine is a non metallic trace element [7].
Iron
Iron is an essential component of hundreds of proteins and enzymes that support essential biological functions; such as oxygen transport, energy production, and DNA synthesis. Hemoglobin, myoglobin, cytochromes and peroxidases require iron containing heme as a prosthetic group for their biological activities. It is a key element in the metabolism of all living organisms [7].
Magnesium
Magnesium is an essential mineral and a cofactor for hundreds of enzymes. Magnesium is involved in many physiologic pathways, including energy production, nucleic acid and protein synthesis, ion transport, cell signaling and also has, structural function. Its functions are as follows.
Magnesium is involved in more than 300 essential metabolic reactions, some of which are discussed below.
The metabolism of carbohydrates and fats to provide energy requires numerous
Energy production: Magnesium – dependent chemical reactions. Magnesium is required by the adenosine triphosphate ATP synthesizing protein in mitochondria. ATP the molecule that provides energy for almost all metabolic processes, exists primarily as a complex with magnesium (Mg ATP).
Synthesis of essential molecules: Magnesium is required for a number of steps during synthesis of deoxyribonucleic acid (DNA), ribonucleic acid (RNA), and proteins. Several enzymes participating in the synthesis of carbohydrates and lipids require magnesium for their activity. Glutathione, an important antioxidant requires magnesium for its synthesis.
Structural role: Magnesium plays a structural role in bone, cell membranes and chromosomes.
Ion transport across cell membranes.
Magnesium is required for active transport of ions like potassium and calcium across the cell membranes. Through its role in ion transport systems magnesium affects the conduction of nerve impulses, muscle contraction and normal heart rhythm.
Cell signaling: Cell signaling requires Mg ATP for the phosphorylation of proteins and the formation of the cell signaling molecules, cyclic adenosine monophosphate (cAMP). cAMP is involved in many processes, including the secretion of parathyroid hormone (PTH) from the parathyroid glands.
Cell migration: Calcium and magnesium levels in the fluids surrounding cells affect the migration of a number of different cell types. Such effects on cell migration may be important in wound healing [7].
Manganese
Manganese is a mineral element that is both nutritionally essential and potentially toxic. its functions are as follows,
Manganese (Mn) plays an important role in a number of physiologic processes as a constituent of multiple enzymes and an activator of other enzymes
Antioxidant functions: Manganese superoxide dismutase ( Mn SOD) is the principal antioxidant enzyme in the mitochondria. Because mitochondria consumes over 90 % of the oxygen used by the cells, they are especially vulnerable to oxidative stress. The superoxide radical is one of the reactive oxygen species produced in mitochondria during ATP synthesis. Mn SOD catalyzes the conversion of superoxide radicals to hydrogen peroxide, which can be reduced to water by other antioxidant enzymes.
Metabolism: A number of manganese-activated enzymes play important roles in the metabolism of carbohydrates, amino acids and cholesterol. Pyruvate carboxylase, a manganese containing enzyme and phosphoenolpyruvate carboxykinase (PEPCK) a manganese activated enzyme are critical in gluconeogenesis, _the production of glucose from non-carbohydrate precursors.
Arginase another manganese-containing enzyme is required by the liver for the urea cycle, a process that detoxifies ammonia generated during amino acid metabolism. In the brain, the manganese activated enzyme glutathione is an excitotoxic neurotransmitter and a precursor to an inhibitory neurotransmitter gamma-amino butyric acid (GABA).
Bone development: Manganese is the preferred cofactor of enzyme called glycosyltransferases; these enzymes are required for the synthesis of proteoglycans that are needed for the formation of cartilage and bone.
Wound healing: Wound healing is a complex process that requires increased production of collagen. Manganese is required for the activities of prolidase an enzyme that functions to promote amino acids, proline for collagen formation in human skin cells. Glycoaminoglycan synthesis which requires manganese – activated glycosyl transferases may also play an important role in wound healing [7].
Molybedenum
The molybedenum atom is part of the molybedenum cofactor in the active site of four enzymes. in humans, sulfite oxide, xanthine oxidase, aldehyde oxidase and amidoxime reducing component.
Molybedenum is an essential trace element for virtually all life forms. It functions as a cofactor for a number of enzymes that catalyze important chemical transformation in the global carbon, nitrogen and sulfur cycle. Its functions are as follows.
The biological function of the molybedenum atom is an organic molecule known as the molybedenum cofactor. (MoCo) present in the active site of MoCo –containing enzyme. In human beings molybedenum is known to function as a cofactor for four enzymes.
Sulfate oxide catalyzes the transformation of sulfite sulfate, a reaction that is necessary for the metabolism of sulfur containing amino acids (methionine and cysteine).
Xanthine oxidase catalyzes the breakdown of nucleotides (precursors to DNA and RNA) to form uric acid, which contributes to the plasma antioxidant capacity of blood.
Aldehyde oxide and xanthine oxidase catalyze hydroxylation reactions that involve a number of different molecules with similar structure. Xanthine oxidase and aldehyde oxide also play a role in the metabolism of drugs and toxins.
Mitochondrial amidoxime reducing component (mARC). Its precise function is under investigation [7].
Phosphorus
Phosphorus is an essential structural component of cell membranes and nucleic acids, but is also involved in several biological processes including bone mineralization, energy production, cell signaling through phosphorylation reactions and regulation of acid-base homeostasis. Phosphorus is an essential mineral that is required by every cell in the body for normal function. Bound to oxygen in all biological systems, phosphorus is found as phosphate in the body. Its functions are as follows,
Phosphorus is a major structural component of bone in the form of calcium-phosphate salt called hydroxyapatite. Phospholipids (e.g., phosphatydal choline) are major structural components of cell membranes. All energy production and storage are dependent on phosphorylated compounds, such as adenosine triphosphate (ATP) and creatine phosphate.. Nucleic acids (DNA) and RNA), which are responsible for the storage and transmission of genetic information are long chains of phosphate containing molecules. A number of enzymes, hormones and cell signaling molecules depend on phosphorylation for their activation. Phosphorus also helps maintain normal acid –base balance (pH) by acting as one of the body’s most important buffers. Additionally the phosphorus –containing molecules 2,3 – diphosphoglycerate (2,3 – DPG) binds to hemoglobin in red blood cells and regulate oxygen delivery to the tissues of the body [7].
Potassium
Potassium is an essential dietary mineral and electrolyte.The term electrolyte refers to a substance that dissociates into ions (charged particles) in solution, making it capable of conducting electricity. Normal body function depends on tight regulation of potassium concentration both inside and outside of cells. Its functions are as follows,
Maintenance of membrane potential: Potassium is the principal positively charged ion (cation) in the fluid inside of cells, while sodium is the principal cation in the fluid outside of cells. Potassium concentrations are about 30 times higher inside than outside cells, while sodium concentrations are more than ten times lower inside than outside cells. The concentration differences between potassium and sodium across cell membranes create an electrochemical gradient known as the membrane potential. A cell’s membrane potential is maintained by ion pumps in the cell membrane, especially the sodium potassium-ATP pumps. The pump uses ATP (energy) to pump sodium out of the cell in exchange for potassium. Their activity has been estimated to account for 20-40% of the resting energy expenditure in a typical adult. The large proportion of energy dedicated to maintaining sodium/potassium concentration gradients emphasizes the importance of this function in sustaining life. Tight control of cell membrane potential is critical for nerve impulse transmission muscle contraction, and heart function.
Cofactor for enzymes: A limited number of enzymes require the presence of potassium for their activity. The activation of sodium potassium - ATPase requires the presence of sodium and potassium. The presence of potassium is also required for the activity of pyruvate kinase, an important enzyme in carbohydrate metabolism [7].
Selenium
Selenium exerts various biological functions mainly as part of the amino acid, selenocysteine, which is found in at least 25 selenocysteine- containing proteins (selenoproteins).
Selenium is trace element that is essential in small amounts. Humans require selenium for the appropriate functioning of a number of selenium –dependent enzymes, known as selenoproteins. Its functions are as follows.
Selenoproteins: Twenty five genes coding for selenoproteins have been identified in humans.
Glutathione peroxidases: Five selenium-containing glutathione peroxidases have been identified GPx isoenzymes are all antioxidant enzymes that reduce potentially damaging reactive oxygen species. (ROS) such as hydrogen peroxide and lipid hydro peroxides to harmless products like water and alcohol by coupling their reduction with the oxidation of glutathione. Spermatogenesis and male fertility are highly dependent on GPx4 and selenoprotein P (SEPP).
Thioredoxin reductases: In mammals three selenocysteine-containing thioredoxin reductases (TRxR) have been identified. TrxRs catalyze the reduction of a wide range of substances, including thioredoxin and protein disulfide isomerase (PDI). TrxRs also serve as electron donors for the regeneration of small antioxidants, possibly recycling ascorbic acid (vitamin C), alpha lipoic acid, alpha tocopherol (vitamin E) and coenzyme Q10 from their oxidized forms. The maintenance of thioredoxin in a reduced form by TrxRs is important for regulating cell growth and survival.The protein thioredoxin together with Trx1 (OR Trx3), NADPH and FAD, constitutes the thioredoxin antioxidant system involved in the reduction of antioxidant enzymes (e.g., peroxiredoxins, methionine sulfoxide reductase, and ribonucleoside reductase) and of many oxidation-reduction (redox)- sensitive signalling proteins Trx1 is one of the most investigated selenoprotein and regarded as one of the major antioxidant enzymes and redox regeneration in mammalian cells.
Iodothyronine deiodonases. (DIOs) are selenoenzymes that catalyze the deiododenation (removal of iodine) from idothronines. DIO1 and DIO2 catalyze the deiodonation of thyroxine (T4) that generates the biologically active triiodothyronine T3 and T4 by removing iodine atom from the inner ring [7].
Sodium
Salt (sodium chloride) is essential for life. The tight regulation of the body’s sodium and chloride concentrations is so important that multiple mechanisms work in concert to control them. Its functions are as follows.
Sodium (NA+ and Cl-) are the principal ions in the fluids outside of cells (extracellular fluid), which include blood plasma. As such they play critical roles in a number of life-sustaining processes.
Maintenance of membrane potential: Sodium and chloride are electrolytes that contribute to the maintenance and charge differences across cell membranes. Potassium is the principal positively charged ion (cation) inside of cells, while sodium is the principal cation in extracellular fluid.
Nutrient absorption and transport: Absorption of sodium in the small intestine plays an important role in the absorption of chloride, amino acids, glucose and water. Similar mechanisms are involved in the reabsorption of these nutrients after they have been filtered from the blood by the kidneys. Chloride in the form of hydrochloric acid (HCL), is also an important component of gastric juice, which aids the digestion and absorption of many nutrients.
Maintenance of blood volume and blood pressure: Because sodium is the primary determinant of extra cellular fluid volume, including blood volume, a number of physiological mechanisms that regulate blood volume and blood pressure work by adjusting the body’s sodium content. In the circulatory system, pressure receptors (baroreceptors) sense changes in blood pressure and send excitatory or inhibitory signals to the nervous system and / or endocrine glands to affect sodium regulation by the kidneys. In general sodium retention results in water retention and sodium loss results in water loss [7].
Zinc
Zinc is a nutritionally essential mineral needed for catabolic, structural and regulatory functions in the body. Zinc is an essential trace element for all forms of life. Its functions are as follows.
Numerous aspects of cellular metabolism are zinc dependent.. Zinc plays important roles in growth and development, the immune response, neurological function and reproduction. On the cellular level the function of zinc can be divided into three categories, catalytic, structural and regulatory.
Catalytic role: Over 300 different enzymes depend on zinc for their ability to catalyze vital chemical reactions. Zinc dependent enzymes can be found in all known classes of enzymes.
Structural role: Zinc plays an important role in the structure of proteins and cell membranes. A finger like structure, known as zinc finger motif, stabilizes the structure of a number of proteins. For example, copper provides the catalytic activity for the antioxidant enzyme copper-zinc superoxide dismutase (Cu Zn SOD), while zinc plays a critical structural role. The structure and function of cell membranes are also affected by zinc. Loss of zinc from biological cell membranes increases their susceptibility to oxidative damage and impairs their function.
Regulatory role: Zinc finger proteins have been found to regulate gene expression by acting as transcription factors (binding to DNA) and influencing the transcription of specific genes. Zinc also plays a role in cell signaling and has been found to influence hormone release and nerve impulse transmission. Zinc has been found to play a role in apoptosis (gene directed cell death), a critical cellular regulatory process with implication for growth and development,as well as a number of chronic diseases [7].
This paper is to present a new hypothesis that will bring forth the pathogenesis of medical illnesses in human beings. It is important to review physiology in order to determine how it progresses to pathophysiology resulting in various diseases.
I have presented the role of psychosocial stress in development of diseases mediated by lipids peroxidation in my paper [1]. Further review of literature in conjunction with clinical experience has revealed that, although psychosocial stress plays important role in development of disease, other stressors if present for prolonged duration also result in development of diseases in similar manner. These stressors were described as biogenic stressors in the citation. In fact exposure to a variety of stressors such as, toxins e.g., cigarette smoke, alcohol, drugs, radiation, pathogens (bacteria or viruses) or other molecules such as pesticides, sensed as antigens will result in stress response.There is activation of HPA and SAM axes. This triggers innate immunity and if the stimuli are ongoing on chronic basis, will activate adaptive immunity.
These mechanisms utilize energy to operate effectively. This is accomplished by increase in rate of cell metabolism.
In an individual who has previously normal health, body is able to respond to the stressor, be it psychosocial or biogenic, by triggering innate immunity. Thus mobilizing its stored energy from carbohydrates and fat, which results in effective immune response. The body has adequate availability of micronutrients in the form of vitamins and minerals for energy pathways to function, and rate of metabolism is increased.
The body then replenishes these stores by increased intake of these nutrients. However when the exposure to the stressor is ongoing in the form of prolonged psychosocial stress or exposure to cigarette smoke or HIV virus or tuberculosis bacillus etc. adaptive immunity is activated. The body has initiated inflammatory response as seen by elevated C reactive protein and other inflammatory markers. This poses ongoing increased energy demand. There is continuous requirement of increase in the rate of cell metabolism. The body has to use stored energy in the form of carbohydrates, fats and even proteins to be processed by energy pathways in order to increase the metabolism. These macro nutrients require micronutrients i.e., vitamins predominantly niacin, and minerals for the energy pathways to convert their stored energy into chemical/metabolic energy in the form of ATP. Over time if these nutrients are not replenished at the pace to keep up with the energy demand, their level begins to fall. This will result in decrease in rate of metabolism, hence inadequate energy production in the face of increased metabolic requirements.
Niacin/vitamin B3 and its derivatives play a key role in energy production by participating in each energy pathway (Glycolysis; both aerobic and anaerobic, oxidative decarboxylation, Kreb citric acid cycle and oxidative phosphorylation). In addition to its role in oxidation-reduction (redox) reactions it also has important role in non redox reactions, acting as intermediates in number of cell signaling pathways, DNA repair, stress responses, transcription regulation, apoptosis, chromatin structure and cell differentiation. Biotin, a B vitamin has essential role in regulation of chromatin structure and gene expression. Folate co enzymes participate in nucleic acid and amino acid metabolism. Pantothenic acid is a precursor in the synthesis of Co enzyme A. Riboflavin participates in oxidation-reduction reactions which are critical for energy metabolism pathways, and also have a role as antioxidants. Thiamine plays important role in energy metabolism pathways.Vitamin B 6, pyridoxine plays important role in hemoglobin and amino acid synthesis and, in nervous system major neurotransmitter synthesis is catalyzed by PLP dependent enzymes. Vitamin B 12 plays important role in methylation reactions for DNA, RNA, and proteins. It also catalyzes the reaction to generate succinyl Co A which then enters the citric acid cycle.Vitamin A in addition to its role in vision, is involved in regulating the growth of virtually all cells in the human body, gene expression and normal functioning of immune system. Vitamin C is a potent antioxidant in addition to its role in biosynthesis of collagen, carnitine and catecholamines. Vitamin D in addition to its role in calcium and phosphorus balance, is a potent immune modulator. Vitamin E has antioxidant role. Vitamin K in addition to its role in coagulation also participates in regulation of cellular functions. The role of minerals has already been reviewed. They play essential roles in energy metabolism in conjunction with vitamins.
As the micronutrient deficiencies become severe, there is progressive decline in cell metabolism and lack of energy. This has serious implications. It results in impairment of function of the body slowly with lack of energy and fatigue. Mental illness is a perfect model to observe the effects of slow metabolism.The patients with Schizophrenia demonstrate negative symptoms with minimal ability to function.
Vitamin D deficiency has been documented in many diseases. It was described as an independent risk factor for heart disease with low levels of vitamin D being associated with higher presence and severity of coronary artery disease [8]. Another study found a strong association between vitamin D deficiency and Schizophrenia [9]. A study confirmed that vitamin D deficiency is associated with a substantially increased risk of all cause dementia and Alzheimer’s disease [10]. A study found that significant proportion of Erectile Dysfunction patients have vitamin D deficiency [11. There are several other studies which describe vitamin D deficiency in many other diseases [7].
This data supports the concept that vitamin D plays a significant role in pathogenesis of these diseases along with other micronutrient deficiencies rather than mere association.
An observational study of 281 HIV+ men found that higher levels of niacin intake were associated with decreased progress rate to AIDs and in prolonged survival [7].
Due to reduced metabolism macronutrients e.g. glucose, fat and proteins are not fully utilized by the cells and their level begins to rise in tissues and blood. Excess nutrients are stored to some extent depending on available energy for this process. The body in order to maintain homeostasis processes the excess macronutrients by modifying these nutrients which results in production of reactive oxygen species, inflammation and oxidative stress, causing tissue damage. This causes progressive loss of function in various tissues and organs. How body reacts to excess macronutrients is described below in the light of current research.
Hyperlipidemia is implicated in development of atherosclerosis which results in cardiovascular diseases.
Excess LDLs are oxidized. which elicit inflammatory response resulting in oxidative stress, causing tissue damage and atherosclerosis in macro circulation [12,13].
A study reported that insulin resistance is a major feature of type 2 diabetes and develops in multiple organs, including skeletal muscle, liver, adipose tissue and heart.The onset of hyperglycemia and diabetes is preceded by many years of insulin resistance [13].
This observation supports, the concept that there is decrease in utilization of glucose by cells due to slow metabolism, which results in high levels in tissues and blood, causing insulin resistance, as glucose can not be metabolized in spite of adequate insulin production.
Hyperglycemia induced ROS production triggers several cellular mechanisms including polyol and hexosamine flux, advanced glycation products (AGES), protein kinase c (PKC) activation, and NF-k B mediated vascular inflammation. PKC is responsible for different structural and functional changes in the vasculature including alterations in cellular permeability, inflammation, angiogenesis, cell growth, extra cellular matrix expansion and apoptosis. An important consequence of PKC activation is ROS generation [14].
Proteins and amino acid are not well studied but we are seeing evidence of its excess deposited as Tau protein and amyloid fibrils in Alzheimer’s disease.
A study described that alterations in the amount or the structure of Tau protein can affect its role as a stabilizer of microtubules as well as some of the processes in which it is implicated. (Tau protein promotes assembly and stabilizes microtubules, which contributes to proper function of neurons). The molecular mechanisms governing Tau aggregation are mainly represented by several post translational modifications that alter its structure and conformational state. Hence abnormal phosphorylation and function of Tau protein have gained attention as key mechanism that become Tau protein in pathological entity [15].
Currently common approach towards vitamin deficiencies is focused on deficiency in a particular vitamin due to decreased intake in diet e.g. Niacin deficiency causing Pellagra is quoted during world war with intake of corn.Vitamin D deficiency causing rickets, vitamin C deficiency causing scurvy in sailors and vitamin B 1, thiamine deficiency causing beri-beri.
However over time there has been changes in environment and life style factors. Nutrition has undergone a major change.The diet patterns are not the same. There is increasing exposure to stressors e.g. food preservatives, detergents, pesticides, cosmetics, air pollutants, fertilizers, drugs, and increasing psychosocial stress. The micronutrient deficiencies have to be approached as multi -micro nutrient deficiencies which develop due to increased demand of these nutrients due to continuous inflammation. This results in a mismatch of supply and demand causing multi morbidities. In current scenario, where all the micronutrients are deficient, replacing one nutrient such as vitamin D, because we measure vitamin D more often may not accomplish desired outcome. This has been addressed in a recent article. In complicated, chronic, multi system disease like metabolic syndrome, cancer and neuro degeneration, one can not expect that replacing one nutrient will shift primary disease outcome measures like BMI or dementia. Physiologically speaking if an individual has deficiency of several nutrients, would benefit be expected, if one nutrient were replaced but not the others [16]. Another important consideration is the quantity of micro nutrients. The individual may be taking the nutrients as recommended by the institute of medicine, outlined as Dietary Reference Intake (DRI) of vitamins, minerals and macronutrients. RDI is the intake considered to be sufficient to meet the needs of 97.5% (2 standard deviations between the mean) of the healthy people [15]. In the light of the concepts presented in this paper as the consumption of micronutrients increases with increase in chronic stress in an individual, the quantity of these nutrients have to be matched with demand and higher DRI is indicated for the optimal cell metabolism to occur, in order to meet the higher energy demand.
This has been addressed in a recent article. The principal of biochemical individuality states that the optimal dose of any nutrient will normally vary between individuals [16].
While multiple stressors are listed, which result in development of diseases by mechanisms described above, and in general they work as absolute triggers, stress indeed is a relative factor. As its is a mismatch between the stressor and the body’s resources (i.e., micronutrients) to meet the demands. For example if body has enough stores of micro and macronutrients, ordinary activities of eating a meal, maintaining body temperature or house hold chores can be performed without stress. However in a patient with Schizophrenia with body’s micronutrient (i.e., niacin) reserves being extremely diminished these activities can not be accomplished without stress.
Hence Hans Selye‘s definition of stress “The non- specific response of the body to any demand for change” is very comprehensive, as in situations of extreme mismatch of nutrients and demand even ordinary activities with minimal demand may not be accomplished, while strenuous activities can be performed with ample supply of micro and macronutrients provided permanent loss of function has not incurred due to oxidative stress in tissues and organs. In the light of above observations stress may be viewed as a mismatch between supply and demand resulting in the long term, in development of disease.
Stress and cancer
As reviewed micronutrients are essential for cell metabolism. When there is progressive deficiency of these nutrients body ranks energy production first (catabolism), at the expense of anabolic functions.
This will compromise the ability to synthesize proteins, hormones, and impair several other functions of cell signaling, gene expression, cell differentiation, DNA and RNA repair, apoptosis and so on which in the long term would result in development of cancer. In other words cancer development is the extreme sequel of chronic stress [17,18].
Optimal metabolism is a prerequisite for normal health and to maintain homeostasis. Mismatch of macro nutrients with micro nutrients results in stress, or allostatic load. Excessive intake of macronutrients, associated with insufficient micronutrients or chronic long term exposure to stressors causing micronutrient deficiency due to increased demand results in stress or allostatic load. The human body maintains homeostasis by ensuring normal metabolism. through balanced intake and utilization of macro and micronutrients. Deficiency of micronutrients (niacin being main nutrient), results in failure to achieve homeostasis. Thus causing allostatic load, slowing metabolism, decreasing the utilization of macro nutrients, which begin to accumulate and go through modification (oxidation of LDLs), anti oxidant mechanisms begin to operate to prevent oxidative stress, inflammation and tissue damage. If not replenished, micronutrient deficiencies continue to become severe, resulting in development of various disease, now commonly seen as multi -morbidities.
Chronic deficiency of micronutrients (niacin deficiency playing primary role) contribute to ageing, development of medical illnesses and cancer. Inflammation progresses in stepwise manner from low grade to severe, respectively in these conditions, and shifts the pendulum from health to various diseases.
The management approach has to focus on increasing the utilization of macronutrients by restoration of metabolism, which in turn will prevent accumulation of these nutrients, allowing body’s natural defense mechanisms to operate; failure of which (e.g. anti oxidant function) results in oxidative stress, inflammation and tissue damage.
Primary prevention: Adequate replacement of all essential micro nutrients in higher doses to match the demand, may be considered to ensure optimal cell metabolism and meet the increased demand of energy.
Secondary prevention: While providing symptomatic treatment with (Insulin in diabetes, diuretics in congestive heart failure.) replacement with essential micronutrients in higher quantities may be continued to prevent further decrease in metabolism and further organ damage due to increase in macro nutrients.
Finally stressors should be identified and eliminated when possible.
Further studies in large populations, similar to Framingham study may be designed to ascertain the role of relative deficiency of micronutrients (with predominant role of niacin deficiency); in development of medical illnesses, with strong potential for prevention and better management of diseases by maintaining optimal levels of these nutrients through diet and supplements if indicated.
- Zafar R (2015) A New Insight into Pathogenesis of Cardiovascular Diseases: Stress Induced Lipid Mediated, Vascular Diseases. Journal of Cardiovascular Diseases & Diagnosis.
- Reeves PG, Nielsen FH, Fahey GC (1993) AIN-93 purified diets for laboratory rodents: final report of the American Institute of Nutrition ad hoc writing committee on the reformulation of the AIN-76A rodent diet. J nutr 123: 1939-1951.
- GS Everly, Lating JM (2002) A Clinical Guide to The Treatment of the Human Stress Response. Third Edition.
- Bowden JA, Brestel EP, Cope WT, Mc Arthur CL, Westfall, et al. (1970) α-ketoisocaproic acid inhibition of pyruvate and α-ketoglutarate oxidative decarboxylation in rat liver slices. Biochemical medicine 4: 69-76.
- Du , Poian AT, El Ba cha T, Luz MRMP (2010) Cell Origin And Metabolism : Citation : Nutrient Utilization in Humans : Metabolism Pathways , Nature Education 3: 11
- Pyruvate oxidation and Citric Acid Cycle, Cell Respiration, Linus Pauling Institute Micronutrient Information Center OSU Oregon State University.
- Morena M, Dupuy AM, Jaussent I, Vernhet H, Gahide G, et al. (2009) A cut-off value of plasma osteoprotegerin level may predict the presence of coronary artery calcifications in chronic kidney disease patients. Nephrol Dial Transplant 24: 3389-3397. [Crossref]
- Cardiosource (2014) Beth casteel. bcasteel @acc.org 202-375-627.
- Valipour G, Saneei P, Esmaillzadeh A (2014) Serum vitamin D levels in relation to schizophrenia: a systematic review and meta-analysis of observational studies. J Clin Endocrinol Metab 99: 3863-3872. [Crossref]
- Littlejohns TJ, Henley WE, Lang IA, Annweiler C, Beauchet O, et al. (2014) Vitamin D and the risk of dementia and Alzheimer disease. Neurology 83: 920-928.
- Barassi A, Pezzilli R, Colpi GM, Corsi Romanelli MM, et al. (2014) Vitamin D and erectile dysfunction. The Journal of Sexual Medicine 11: 2792-2800.
- Inflammation in Atherosclerosis Libby P1, Ridker PM, Hansson GK; Leducq Transatlantic Network on Atherothrombosis (2009) Inflammation in atherosclerosis: from pathophysiology to practice. J Am Coll Cardiol 54: 2129-2138. [Crossref]
- Erling Falk E ( 2006 ) Pathogenesis of Atherosclerosis. J Am Coll Cardiol 47 : c 7- c 12
- Diabetes and vascular diseases : Pathophysiology , clinical consequences , And medical therapy : Part 1
- Kolarova M, García-Sierra F, Bartos A, Ricny J, Ripova D (2012) Structure and pathology of tau protein in Alzheimer disease. International Journal of Alzheimer’s Disease.
- Mischley LK (2014) Conditionally Essential Nutrients: The State of the Science. J Food Nutr 1: 1-4.
- Bianco A, Thomas E, Pomara F, Tabacchi G, Karsten B, et al. (2014) Retraction Note: Alcohol consumption and hormonal alterations related to muscle hypertrophy: a review. Nutr Metab (Lond) 11: 43. [Crossref]
- Donaldson MS (2004) Nutrition and cancer: a review of the evidence for an anti-cancer diet. Nutr J 3: 19. [Crossref]


