Background : Assessing seasonal severity of influenza is problematic; traditional indicators may not be reliable to determine the magnitude of each influenza season. We investigated why the overall impact of influenza was much higher during the 2014/2015 season despite the fact that peak influenza positivity was lower than both the 2010/2011 and 2012/2013 influenza A(H3N2) dominant influenza seasons. The magnitudes of area under the epidemic curve of influenza positivity and the ascending and descending slopes as a measure of seasonal severity were also explored.
Methods : Respiratory specimens were collected from patients and submitted for influenza testing at Public Health Ontario Laboratories. Weekly influenza positivity was calculated for each season. Number of laboratory-confirmed cases of influenza, hospitalization and case fatality rates among laboratory-confirmed cases and the number of institutional influenza outbreaks were determined for each season. Ascending and descending slopes of the seasonal epidemic curve were calculated in addition to the area under the epidemic curve.
Results : While the impact of the 2014/2015 influenza A(H3N2) season was high based on a number of indicators, some of this impact may be explained by the prolonged influenza season and the unusually low vaccine effectiveness.
Conclusions : In comparison to other H3N2 dominant seasons that were studied, influenza positivity both increased and decreased more slowly during the 2014/2015 season as compared to the other two influenza A(H3N2) dominant seasons. This may have resulted in a longer period of influenza transmission in addition to other unmeasured factors, which may explain the increased severity of the season.
influenza; influenza A virus; H3N2 subtype; indicators of influenza severity
In Canada and other non-temperate climates, influenza circulates at higher levels during the late autumn and winter, [1-4] with activity most commonly peaking between December and February when influenza A is the dominant circulating type and the early spring when influenza B is dominant [5,6]. In Ontario, Canada’s most populous province, influenza season traditionally peaks in late December/early January during periods when influenza A activity, particularly the H3N2 subtype, is dominant.
In a study looking at the duration of influenza seasons for 23 countries in Europe [7], the median length of influenza season in individual countries varied from 12-19 weeks with a median duration of 15 weeks. Similarly Baumgartner noted that seasonal influenza epidemics had duration of 4 months, regardless of climatic region (e.g. temperate) with separate waves of activity caused by different circulating types/subtypes each lasting a mean of 11 weeks. However the duration of seasonal activity may not necessarily equate to the severity of the season.
Assessing seasonal severity is problematic; the total number of laboratory-confirmed cases may not be a reliable indicator of the magnitude of each influenza season. Many factors can affect case counts including: most ill individuals do not seek medical care, varying testing practices of clinicians and use of more sensitive molecular testing in recent years and only in certain geographic regions. In Ontario, reporting of hospitalizations and deaths of patients with laboratory-confirmed influenza has varied over time, limiting its use as a stable indicator of influenza severity. The reporting of institutional influenza outbreaks has remained consistent over time as the same provincial case definition has been used since 2001, enhancing its ability to be used as a reliable indicator of the severity of the season. However, this also has limitations, as influenza A(H1N1)pdm09 is less likely to cause outbreaks in elderly residents of long-term care facilities (LTCFs) than either influenza A(H3N2) or influenza B, and most outbreaks that undergo laboratory testing in Ontario occur among elderly persons residing in LTCFs. Use of influenza positivity, while also subject to testing bias (e.g. if test methods change), may be a useful indicator of the severity and magnitude of the season. As an alternative, the magnitude of the ascending and descending slope of influenza positivity may be useful to gauge the magnitude of the season.
The purpose of our investigation was to explore why the overall impact of influenza as measured by case counts, percent positivity, number of institutional influenza outbreaks, hospitalization and mortality rates, was much higher during the 2014/2015 season despite the fact that peak influenza test positivity was lower than that of both the 2010/2011 and 2012/2013 influenza A(H3N2) dominant influenza seasons. As well we explored if the magnitude of the ascending and descending slopes of influenza positivity for influenza or the size of the area under the epidemic curve of influenza positivity (henceforth “area under the curve”) could be used as a measure of seasonal severity.
Influenza A(H3N2) dominant seasons were used as comparator seasons since H3N2 was the dominant influenza A subtype during the 2014/2015 influenza season. Influenza A(H3N2) dominant seasons subsequent to the 2009 H1N1 influenza A pandemic were chosen because polymerase chain reaction (PCR) testing, a more sensitive test method than viral culture, was not used widely at PHOL prior to that time.
Respiratory specimens were collected from patients and submitted for influenza A and B testing at Public Health Ontario Laboratories (PHOL) by culture and/or influenza real-time reverse transcriptase (rRT)-PCR and/or multiplex respiratory viral PCR (MRVP) [(Luminex Molecular Diagnostic, Toronto, Ontario, Canada) or Seeplex RV15 ACE (Seegene USA, Rockville, MD, USA)]. In the event of discrepant results between testing methods, positive results overrode indeterminate or negative ones.
Each influenza reporting season was defined as the period from September 1 to August 31 of the following year. For the purposes of this paper, weekly influenza positivity was defined as the total number of respiratory specimens testing positive for influenza, out of all respiratory specimens tested at PHOL for that week, expressed as percent positivity. Weekly influenza positivity was calculated for each season. The start of seasonal influenza A activity was defined as the second consecutive week when influenza A positivity exceeded 5% based on PHOL data. Similarly, the end of seasonal activity was defined as the second consecutive week when influenza A positivity was less than 5%. Peak influenza A activity was defined as the highest weekly value for influenza A positivity for that season.
Information about laboratory-confirmed cases of influenza, hospitalization and case fatality rates among laboratory-confirmed cases and the number of institutional influenza outbreaks were based on entries from Ontario’s 36 local health units into Ontario’s reportable disease reporting system, the integrated Public Health Information System (iPHIS) [6]. Hospitalization rates were calculated as the number of laboratory-confirmed cases of influenza that were reported as being hospitalized over a given influenza season. Fatality rates were calculated as any death reported in an influenza laboratory-confirmed case; the contribution of influenza to the death was not able to be determined.
As an additional indicator of the magnitude of the three influenza A(H3N2) dominant seasons, the area under the curve in addition to the ascending and descending slopes of the seasonal epidemic curve were calculated. The area under the curve for each season was measured as the seasonal sum of cumulative weekly percent positivity. The ascending slope for each season was calculated as a ratio of the difference in percent positivity from seasonal peak to seasonal start divided by number of weeks in between these two timeframes. Similarly, the descending slope was calculated as a ratio of the difference in percent positivity from seasonal peak to seasonal end divided by the number of weeks between these two timeframes.
Unlike the 2010/2011 and 2012/2013 seasons, the 2014/2015 influenza season included an additional week (week 53); a week 53 occurs every five to six years. A separate sensitivity analysis was conducted in order to adjust for the variability in start, peak and end week for each of the three seasons in addition to the effect of the extra week. In the sensitivity analysis we standardized each season by using the same start, peak and end week to calculate the seasonal slope to determine if results vary.
Canadian influenza vaccine effectiveness (VE) results from the Sentinel Practitioner Surveillance Network (SPSN) have been delineated for each season described in this study as VE may have also influenced influenza activity and outcomes. A description of the Canadian SPSN in which Ontario is one of four participating provinces, has been described elsewhere [8].
Data were collected as part of seasonal influenza virus surveillance; hence research ethics approval was not required.
Comparison of the severity of H3N2 dominant influenza seasons
In Ontario, by nearly every measure of severity, including laboratory-confirmed cases, institutional influenza outbreaks and hospitalization and fatality rates among laboratory-confirmed cases, the 2014/2015 season was the most severe as compared to the two other most recent influenza A(H3N2) dominant seasons, 2010/2011 and 2012/2013 (Table 1).
For all three seasons combined, a total of 111,580 respiratory specimens were tested for influenza with 16,135 (14.5%) testing positive for influenza A (Table 1). Both the number of respiratory specimens testing positive for influenza and testing volume progressively increased for each season during 2010/11, 2012/13 and 2014/15. Influenza A percent positivity in 2014/15 was higher at 14.2% than in 2010/11 (11.6%), but lower than in 2012/13 (16.8%). Peak percent positivity was the lowest during 2014-2015 season (31.9%) in comparison to both the 2010/2011 (34.3%) and the 2012/2013 (40.5%) seasons (Figure 1 & Figure 2) (Table 1).
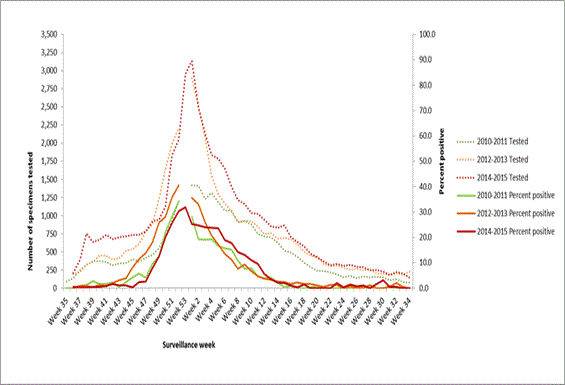
Figure 1. Respiratory specimens tested and percent positivity for influenza, by week, Ontario, 2010/11, 2012/13 and 2014/15 influenza seasons Gap in lines representing positivity is reflective of the fact that unlike the other two H3N2 seasons shown in this graph, the 2014/2015 season included a week 53.
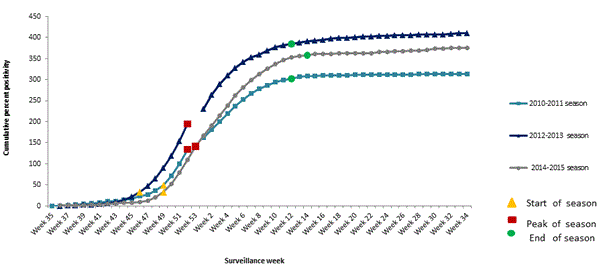
Figure 2. Beginning*, peak and end† of the season as measured by percent positivity, by week,‡ Ontario, 2010/11, 2012/13 and 2014/15 influenza seasons * The beginning of an influenza season was defined as the second consecutive week when influenza A percent positivity was higher than 5%
Hospitalization and fatality rates for laboratory-confirmed influenza cases was highest during the 2014/2015 season at 33.3 and 2.7 per 100,000 population, respectively (Table 1). The number of reported institutional influenza outbreaks was also highest during the 2014/2015 season, with 75% more institutional influenza A outbreaks reported during that season (1,049 institutional influenza A outbreaks) than the next most severe season, 2012/2013 (600 outbreaks).
Overall, there were 38% more influenza-positive specimens, 58% more reported laboratory-confirmed influenza cases, 49% more hospitalized influenza cases, 45% more fatal influenza cases and 102% more institutional influenza outbreaks in the 2014/2015 season than the average number of the two previous H3N2 dominant seasons. Most of the difference in the number of influenza-positive specimens occurred in the 2010/2011 season as compared to the 2014/2015 season (113% increase), while there was only a 2% increase in 2014/2015 as compared to 2012/2013.
Influenza VE varies from year to year, with the H3N2 subtype exhibiting the most variability. VE for the 2014/2015 influenza season as per the Canadian SPSN was essentially zero [9], the lowest VE in the 10 years of the network’s existence as compared to an average of 40% for the 2010/2011 and 2012/2013 seasons.
During the 2010/2011 and 2012/2013 seasons, peak influenza positivity reached 34% and 41% respectively. Despite the fact that the impact of influenza infection appeared to be most severe during the 2014/2015 season, peak positivity (31.9%) was lower that season as compared to either of the prior two influenza A(H3N2) dominant seasons (Tables 1 and 2).
Table 1.Comparison of seasonal influenza activity, Ontario, 2010/11, 2012/13 and 2014/15 influenza seasons
|
|
2010/11
|
2012/13
|
2014/15*
|
|
Number of respiratory specimens tested
|
26,635
|
38,480
|
46,465
|
|
Number (%) of specimens influenza A positive
|
3,095 (11.6%)
|
6,454 (16.8%)
|
6,586 (14.2%)
|
|
Peak percent positivity of influenza A specimens
|
34.3%
|
40.5%
|
31.9%
|
|
Number of laboratory-confirmed influenza A cases†
|
6,049
|
8,722
|
11,637
|
|
Number (rate per 100,000 population) of laboratory-confirmed influenza A hospitalizations
|
2,342 (17.75/100,000)
|
3,698 (27.4/100,000)
|
4,514* (33.3/100,000)
|
|
Number (rate per 100,000) of laboratory- confirmed influenza A case fatalities
|
202 (1.53/100,000)
|
292 (2.2/100,000)
|
359*
(2.7/100,000)
|
|
Number of institutional influenza A outbreaks
|
437
|
600
|
1,049
|
|
Influenza vaccine effectiveness (H3N2 subtype)
|
39% 13
(95% Confidence Interval, 14%-57%)
|
41% [14]
(95% Confidence Interval, 17%-59%)
|
-16% [9]
(95% Confidence Interval, -49%-9%)
|
* Reporting changes during the 2014/2015 influenza season likely resulted in under-reporting of the number of hospitalizations and deaths for that season
†While the number of influenza A-positive specimens is based on PHOL data, the number of influenza A cases includes all laboratory-confirmed cases reported through the integrated Public Health Information System (iPHIS) by Ontario’s public health units, which includes data from all laboratories in Ontario that test for influenza
Table 2.Start, peak and end of seasonal influenza activity, 2010/2011, 2012/2013 and 2014/2015 seasons, Ontario
|
Influenza season
|
Start week*
|
Influenza positivity at start week
|
Peak week of influenza positivity
|
Peak influenza positivity
|
End week†
|
Influenza positivity in end week
|
Difference in weeks between start and peak
|
Difference in influenza positivity between peak and start of the season
|
Ascending slope (% per week)
|
Difference in weeks between end and seasonal peak
|
Difference in influenza positivity between peak end of the season
|
Descending slope (% per week)
|
|
2010-2011
|
49
|
12.5
|
52
|
34.3
|
12
|
3.9
|
4
|
21.8
|
5.5
|
13
|
30.4
|
2.33
|
|
2012-2013
|
46
|
11.3
|
52
|
40.5
|
12
|
3.7
|
7
|
29.2
|
4.2
|
13
|
36.8
|
2.83
|
|
2014-2015
|
49
|
12.5
|
53
|
31.9
|
14
|
2.2
|
5
|
19.4
|
3.9
|
15
|
29.7
|
1.98
|
* Start week is defined as the second consecutive week when influenza positivity is >5%
† End week is defined as the second consecutive week when influenza positivity is <5%
Comparison of the area under the curve and slope of influenza positivity
The size of the area under the curve, which represents the cumulative sum of percent positivity, theoretically should reflect severity of the season, with the most severe season having the greatest area under the curve (or the highest sum of cumulative percent positivity). However, this was not the case as the size of the area under the curve mirrored overall percent positivity being the highest during 2012/2013 season. Specifically, the area under the curve was 324.7%, 426.2 % and 388.4% for the 2010/2011, 2012/2013 and 2014/2015 seasons, respectively. The ascending slope of the epidemic curve from seasonal start (week 49) to peak week (53) for respiratory specimens tested for influenza was the least steep at 3.9 percent per week during the 2014/2015 season compared to 5.5 and 4.2 percent per week, during the two other H3N2 dominant seasons indicating the slower rise of influenza activity (Table 2). Similarly, of all H3N2 dominant seasons since 2009, the downward slope following peak influenza activity was the least steep for the 2014/2015 season at 1.9 percent per week compared to 2.3 and 2.8 percent per week, respectively, indicating a prolonged decrease of activity. Additionally, the ascending slope in 2014/2015 was the least steep of these seasons, indicating a slower rise to the peak, and longer duration to arrive at a fixed peak. (Table 2)
In sensitivity analysis, when keeping the influenza season constant (i.e. using the same start (week 46) and end week (week 15), and using week 52 or week 53 as the peak week for 2014-2015 season), both the ascending and descending slopes remained the least steep for 2014-2015 season. Specifically the ascending and descending slopes were 3.7 and 1.9 percent per week respectively using week 52 as the peak for the 2014/2015 season as compared to 4 and 1.9 percent per week when week 53 was used as the peak week. Overall, influenza positivity increased and decreased more slowly during the 2014/2015 season as compared to the other two influenza A(H3N2) dominant seasons.
In this study we have demonstrated that while the impact of the 2014/2015 influenza A(H3N2) season was high based on a number of indicators such as the number of laboratory-confirmed cases and rates of hospitalization and deaths among laboratory-confirmed cases, some of this impact may be explained by the prolonged influenza season and the unusually low vaccine effectiveness.
One of the difficulties in comparing the 2014/2015 to prior H3N2 dominant seasons is that influenza case reporting in Ontario changed during the 2014/2015 season. During that season, recommendations in regards to follow-up of laboratory-confirmed cases performed by local public health changed from a follow-up of all cases prior to December 24, 2014, to following up only one out of every five laboratory-confirmed influenza cases subsequent to that date. This likely resulted in under-reporting of the number of cases that were hospitalized or had a fatal outcome during the 2014/2015 season as compared to previous influenza seasons. However despite the decreased follow up, rates of hospitalizations and deaths reported for laboratory-confirmed cases were higher during the 2014/2015 season. Reporting of institutional influenza outbreaks did not change however, and the number of such outbreaks reported in the 2014/2015 season increased substantially as compared to the two prior H3N2 dominant seasons, further indicating the greater impact of the 2014/2015 season as compared to previous seasons.
One reason we were not able to observe the true burden of the 2014/2015 season using PHOL data was that we excluded results from influenza-positive specimens (referrals) tested by other laboratories since we could not determine percent positivity for these samples. While the number of referrals (influenza positive samples forwarded for subtyping) was small for the 2010/2011 season (15), for the 2012/2013 and 2014/2015 seasons, this increased to 78 and 2,360 respectively, likely representing increased influenza testing done by other (non-PHOL) Ontario laboratories. For the 2014/2015 season, this represented 40% of all influenza positive specimens tested at PHOL. Given the increase in referrals in 2014/2015 as compared to the prior H3N2 dominant seasons, excluding referrals from our analysis may have obscured the true magnitude (i.e. number of influenza A positive specimens) during the 2014/2015 season.
Knowing the reproductive number (Ro) for each season could have helped us to interpret seasonal comparisons since overall attack rates are influenced by changes in the reproductive number [10,11]. However, we did not have sufficient data to determine Ro for any season.
2021 Copyright OAT. All rights reserv
PHOL performs the majority of testing for influenza and other respiratory viruses in the province of Ontario (population 13.8 million), but other microbiology laboratories also perform these tests. Therefore, it is important to note that PHOL data do not represent the total number of influenza-positive specimens identified in Ontario. PHOL is also the provincial reference laboratory for respiratory virus testing; other laboratories, most frequently hospital laboratories, will send influenza positive specimens to PHOL for subtyping (termed “referrals”). Referral specimens were excluded from analyses since we are not able to ascertain the total number of influenza-negative specimens tested by these laboratories and including influenza positive specimens only would have falsely increased test positivity.
Early during the 2014/2015 influenza season, a re-emerging enterovirus serotype [enterovirus D68 (EV-D68)] was widely reported by the United States Centers for Disease Control and Prevention to cause severe respiratory illness in children. These reports may have resulted in a higher number of respiratory specimens submitted to PHOL early during the 2014/2015 season, since Ontario, as well as other Canadian provinces, reported EV-D68 detections during September and October, 2014. This may have prompted clinicians to increase respiratory specimen submissions and thus reduced influenza positivity early in the season, and therefore may have dampened the increase of the ascending slope. However, the last EV-D68 positive specimen was detected on October 17, 2014 (week 42), with requests for EV-D68 testing declining subsequent to this date. Therefore it is unlikely that increased respiratory specimen submissions for EV-D68 testing affected influenza positivity, since the start of the influenza season occurred during week 49, 2014, well after peak specimen submission for EV-D68 testing [12].
Overall the 2014/2015 influenza A(H3N2) season was unprecedented in terms of total number of respiratory specimens tested, number of laboratory-confirmed cases, number of institutional influenza outbreaks and rates of hospitalization and death among laboratory-confirmed cases for any post-pandemic influenza A(H3N2)-dominant season. Given the magnitude of the impact of the 2014/2015 season, it would be expected that the percent positivity would be as high as or higher than the previous two H3N2 dominant seasons in conjunction with the greater severity observed, however this was not the case. The area under the curve was the highest during the 2012-2013 season, likely reflective of the fact that peak percent positivity as well as overall percent positivity were the highest during that season. This indicates that percent positivity alone cannot be a robust measurement of severity for influenza season. In comparison to other H3N2 dominant seasons that were studied, the ascending and descending slopes of the 2014/2015 influenza season were the least steep. This reflected the fact that influenza positivity both increased and decreased more slowly during the 2014/2015 season as compared to the other two influenza A(H3N2) dominant seasons. This may have resulted in a longer period of influenza transmission provincially, in addition to other unmeasured factors, which may explain the higher number of laboratory-confirmed cases and institutional influenza outbreaks, as well as higher hospitalization and death rates among cases [13].
Contributing to the effects of the extended 2014/2015 season was the extraordinarily low influenza vaccine effectiveness (essentially zero) for the dominant H3N2 subtype. A new H3N2 clade circulated during the 2014/2015 season which was not contained in that season’s vaccine, thus individuals were exposed to this new clade for a prolonged period of time, potentially resulting in greater transmission and ultimately, serious outcomes in populations at higher risk of complications.
Once seasonal influenza activity starts, the ability to anticipate its duration and impact can help inform the timing of vaccine delivery, guidance in the use of empiric influenza antivirals and anticipating healthcare system capacity issues. However monitoring percent positivity or area under the curve alone cannot define the magnitude of seasonal influenza activity. Trends in the number of reported laboratory-confirmed cases and hospitalization and death rates should be considered in addition to analyzing vaccine effectiveness and monitoring circulating influenza strains which may result in reduced immunity and prolonged transmission.
Conceived, analyzed the data, and wrote the manuscript – Anne-Luise Winter, Adriana Peci, Lennon Li, Jonathan B. Gubbay.
This paper did not receive any grants or other sources of funding.
The authors declare that they have no competing interests.
- Statement on Seasonal Influenza Vaccine for 2014-2015. [http://www.phac-aspc.gc.ca/naci-ccni/flu-grippe-eng.php].
- Sakai T, Suzuki H, Sasaki A, Saito R, Tanabe N, et al. (2004) Geographic and temporal trends in influenzalike illness, Japan, 1992-1999. Emerg Infect Dis 10: 1822-1826. [crossref]
- Fowlkes A, Giorgi A, Erdman D, Temte J, Goodin K, et al. (2014) Viruses associated with acute respiratory infections and influenza-like illness among outpatients from the Influenza Incidence Surveillance Project, 2010-2011. J Infect Dis 209: 1715-1725. [crossref]
- Azziz Baumgartner E, Dao CN, Nasreen S, Bhuiyan MU, Mah-E-Muneer S, et al. (2012) Seasonality, timing, and climate drivers of influenza activity worldwide. J Infect Dis 206: 838-846. [crossref]
- Centers for Disease Control and Prevention. [http://www.cdc.gov/flu/about/season/flu-season.htm].
- Ontario Respiratory Pathogen Bulletin. [http://www.publichealthontario.ca/en/ServicesAndTools/SurveillanceServices/Pages/Ontario-Respiratory-Virus-Bulletin.aspx]
- Paget J, Marquet R, Meijer A, van der VK (2007) Influenza activity in Europe during eight seasons (1999-2007): an evaluation of the indicators used to measure activity and an assessment of the timing, length and course of peak activity (spread) across Europe. BMC Infectious Diseases 7: 141.
- Skowronski DM, Chambers C, Sabaiduc S, De Serres G, Winter AL, et al. (2015) Integrated Sentinel Surveillance Linking Genetic, Antigenic, and Epidemiologic Monitoring of Influenza Vaccine-Virus Relatedness and Effectiveness During the 2013-2014 Influenza Season. J Infect Dis 212: 726-739. [crossref]
- Skowronski DM, Chambers C, Sabaiduc S, De Serres G, Winter AL, et al. (2016) A Perfect Storm: Impact of Genomic Variation and Serial Vaccination on Low Influenza Vaccine Effectiveness During the 2014–2015 Season. Clin Infect Dis 63: 21-32. [crossref]
- Biggerstaff M, Cauchemez S, Reed C, Gambhir M, Finelli L (2014) Estimates of the reproduction number for seasonal, pandemic, and zoonotic influenza: a systematic review of the literature. BMC Infectious Diseases 14: 480.
- Zhang S (2011) Estimating Transmissibility of Seasonal Influenza Virus by Surveillance Data. J Data Science 9: 55-64.
- Peci A, Winter AL, Warshawsky B, Booth TF, Eshaghi A, et al. (2015) Epidemiology of Enterovirus D68 in Ontario. PLoS One 10: e0142841. [crossref]
- Skowronski DM, Janua NZ, De Serres G, Winter AL, Dickinson JA, et al. (2012) A Sentinel Platform to Evaluate Influenza Vaccine Effectiveness and New Variant Circulation, Canada 2010-2011 Season. Clin Infect Dis 55: 332-342. [crossref]
- Skowronski DM, Janjua NZ, De Serres G, Sabaiduc S, Eshaghi A, et al. (2014) Low 2012-13 influenza vaccine effectiveness associated with mutation in the egg-adapted H3N2 vaccine strain not antigenic drift in circulating viruses. PLoS One 9: e92153. [crossref]


