Brainstem encephalitis may be a serious complication of enteroviral infection. Mainly due to human enterovirus A71, it has become a real health issue in Asia Oceania. In Europe, infection remains sporadic or causes small epidemics. Here we report an original case of Coxsackie B3 brainstem encephalitis, the first one to the best of our knowledge. A healthy five years old girl developed brainstem involvement signs. Clinical examination showed no fever, normal heart frequency and normal blood pressure. She presented a self-limited divergent squint which lasted 2 hours followed by oculomotor troubles and a right facial palsy. Biological tests performed on cerebrospinal fluid were not informative. The cerebral Magnetic Resonance Imaging was compatible with a rhombencephalitis. She totally recovered one month after this episode.
Enterovirus was detected by real-time PCR on the nasopharyngeal and the stool sample collected on day of admission but was not detected on the cerebrospinal fluid. Coxsackie B3 was genotyped by sequencing of VP1 protein coding sequence. Despite the absence of enterovirus detection on the cerebrospinal fluid, the diagnosis of enterovirus rhombencephalitis was classified as probable by the French National Reference Centre for enterovirus. Our description highlights the necessity of nasopharyngeal and stool sampling in order to extend the diagnosis with the aim of learning about enterovirus circulation in Europe as well.
coxsackie B3; rhombencephalitis; neurological complication; sporadic
Most enteroviral infections are asymptomatic, but some may lead to neurological manifestations, mostly benign meningitis. Serious central nervous system involvements are possible like meningo-encephalitis, brainstem encephalitis and in particular flaccid palsy [1]. These severe neurological cases are predominantly due to Human Enterovirus A71 (HEV A71) and it has become a real health issue in Asia Oceania mainly, where major epidemics occur every 2-3 years [2].
In Europe, two large HEV A71 epidemics occurred in Bulgaria (44 deaths) in 1975 and Hungary (47 deaths) in 1978 [2,3]. Since 1978 in Europe HEV A71 infections have remained sporadic or caused small outbreaks. Between April and May 2016 in Catalonia (Spain) an outbreak reported 87 infections with neurological complications, 22 of them in intensive care units. No fatalities were recorded [3]. Rhombencephalitis is mainly due to HEV A71 as well, and only a very few cases have already been reported with another enterovirus [4,5].
Here we describe a case of a rhombencephalitis due to coxsackie B3. It belongs to the Human Enterovirus B (HEV B) genus.
A five year old girl with no medical history except for a varicella infection 2 months ago attended the paediatric emergency unit on 16 May 2016 presenting a self-limited divergent squint which lasted 2 hours the morning directly before this. She had been suffering from a vesperal headache and a loss of appetite for 10 days.
Clinical examination showed no fever, normal heart frequency and normal blood pressure. She was conscious and oriented. No meningeal syndrome was shown. She presented oculomotor troubles (ocular apraxia and multidirectional nystagmus) and a right facial palsy. A discrete cerebellar syndrome was observed with balance disorder.
Blood chemistry and haematological cell count did not reveal any abnormalities, the CRP was normal (0.7mg/L). The cerebral Magnetic Resonance Imaging (MRI) showed an isolated posterior median pons hyper signal (Figure 1) without restriction of the conduction on apparent diffusion coefficient map and no mass effect on the lumen of the fourth ventricle. The electromyogram was not in favour of a peripheral nervous system disorder (Guillain-Barré or Miller Fisher Syndrome). A lack of conduction was observed on medullar and lower bulbo-medullar areas.
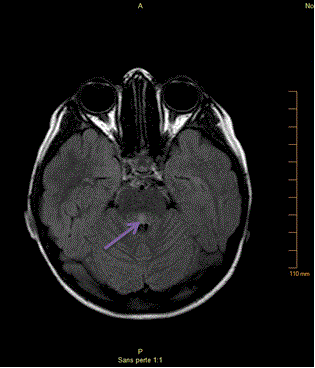
Figure 1. Cerebral MRI T1-weight image, axial slice.Isolated posterior median pons hyper signal.
The serologic status was negative for Lyme disease, Campylobacter jejuni, Campylobacter fetus, Mycoplasma pneumoniae, Herpes simplex virus and virus B19. She was previously immunized (IgG positive) for cytomegalovirus (CMV), Epstein-Barr virus (EBV), varicella-zoster virus (VZV) and human herpes virus 6 (HHV6). Testing for antibodies directed against gangliosides was negative. The CSF was clear, without cells, proteins, glucose and chlorides were normal (0.24 g/L, 2.77 mmol/L and 124 mmol/L respectively). The CMV, HSV, VZV and enterovirus PCR were negative in CSF. Tested in CSF, antibodies anti-VZV (IgG and IgM) were negative.
Enterovirus was detected on a nasopharyngeal and a stool sample collected on day of admission by real-time PCR. Coxsackie B3 was genotyped at the National Reference Centre for Enterovirus at the University Hospital of Clermont-Ferrand by sequencing of VP1 protein coding sequence [6]. The girl was treated by methylprednisolone bolus of 600 mg once a day for 5 days. A discreet clinical improvement was observed during the eight days of hospitalization and a total recovery noted during the consultation one month later.
According to the National Reference Centre for enterovirus definition (defined by paediatric neurologist and intensive care specialists of Necker Hospital and reported on the declaration form of serious cases available on National Reference Centre for enterovirus website: http://cnr.chu-clermontferrand.fr/CNR/Pages/BulletinsInfos.aspx), the diagnosis of enterovirus rhombencephalitis was classified as probable in the absence of other aetiology.
To the best of our knowledge no other cases of coxsackie B3 rhombencephalitis have been described to date.
Indeed coxsackie B viruses are well known to cause aseptic meningitis and meningo-encephalitis mainly in newborn [7] and immunocompromised patients [7-9]. Myocarditis/pericarditis [10-13], neonatal sepsis [14,15] but also a few cases of type 1 diabetes, pancreatitis, hepatitis [11], uveitis [16] have also been reported. In a few cases of hand, foot and mouth disease, Coxsackie B3 can also be found [17].
Coxsackie B3 was only detected in nasopharyngeal and stool samples, but not in the CSF. The presence of enterovirus-RNA is not always possible, and, for example, some cases of enterovirus brainstem encephalitis have already been described without any CSF positive enterovirus detection [3,15,18,19]. The detection of the virus in the nasopharyngeal and stool specimen and the absence of such detection in CSF confirm the necessity of peripheral sampling in order to extend the virological diagnosis. In one case, enterovirus was only detectable in cerebral biopsies [8]. Furthermore other bioassays could be uninformative, in particular pleocytosis can be absent at the moment the sample is collected [1].
Epidemics of severe neurologic involvement due to enterovirus are an important health issue mainly in Asia Oceania. In Europe due to the Catalonia outbreak and a recent alert of the French paediatrician’s society reporting the increasing number of severe neurological cases due to non-polio enterovirus in France [20], clinicians are encouraged to collect stool and respiratory specimens from patients suggestive of severe neurological disorders and to report these cases, with the aim of learning about enterovirus circulation in Europe. Our description confirms the need for this survey.
2021 Copyright OAT. All rights reserv
- Wang SM, Liu CC, Tseng HW, Wang JR, Huang CC, et al. (1999) Clinical spectrum of enterovirus 71 infection in children in southern Taiwan, with an emphasis on neurological complications. Clin Infect Dis 29: 184-190. [Crossref]
- Solomon T, Lewthwaite P, Perera D, Cardosa MJ, McMinn P, et al. (2010) Virology, epidemiology, pathogenesis, and control of enterovirus 71. Lancet Infect Dis 10: 778-790. [Crossref]
- European Centre for Disease Prevention and Control (ECDC). Outbreak of enterovirus A71 with severe neurological symptoms among children in Catalonia, Spain. 14 June 2016. Stockholm: ECDC. 14 Jun 2016. Available from: http://ecdc.europa.eu/en/publications/Publications/07-06-2016-RRA-Enterovirus%2071-Spain.pdf.
- Brecht M, Jyoti R, McGuire W, Chauhan M (2010) A case of neonatal coxsackie B virus brainstem encephalitis. J Paediatr Child Health 46: 699-701. [Crossref]
- Xu W, Liu CF, Yan L, Li JJ, Wang LJ, et al. (2012) Distribution of enteroviruses in hospitalized children with hand, foot and mouth disease and relationship between pathogens and nervous system complications. Virol J 9: 8. [Crossref]
- Audrey M, Christine A, Cecile H, Yanne M, Martine C, et al. (2006) Prospective identification of HEV-B enteroviruses during the 2005 outbreak. J Med Virol 78: 1624-1634.
- Ronellenfitsch S, Tabatabai J, Böttcher S, Diedrich S, Frommhold D, et al. (2014) First report of a Chinese strain of coxsackie B3 virus infection in a newborn in Germany in 2011: a case report. J Med Case Rep 8: 164. [Crossref]
- Berger JR, Chumley W, Pittman T, Given C, Nuovo G (2006) Persistent Coxsackie B encephalitis: Report of a case and review of the literature. J Neurovirol 12: 511-516. [Crossref]
- Palacios T, Bartelt L, Scheld W, Lopes MB, Kelting SM, et al. (2015) Fatal Coxsackie meningoencephalitis in a patient with B-cell lymphopenia and hypogammaglobulinemia following rituximab therapy. Ann Allergy Asthma Immunol 115: 148-150. [Crossref]
- Cen Z, Guo Y, Kong Q, Zhou Q, Wu W (2015) IL-10-producing B cells involved in the pathogenesis of Coxsackie virus B3-induced acute viral myocarditis. Int J Clin Exp Pathol 8: 830-835. [Crossref]
- Persichino J, Garrison R, Krishnan R, Sutjita M (2016) Effusive-constrictive pericarditis, hepatitis, and pancreatitis in a patient with possible coxsackievirus B infection: a case report. BMC Infect Dis 16: 375. [Crossref]
- El-Asmar J, Kharfan-Dabaja MA, Ayala E (2016) Acute pericarditis and tamponade from Coxsackie B3 in an adult Hematopoietic-Cell-Allograft recipient: A rare but potentially serious complication. Hematol Oncol Stem Cell Ther 9: 82-85. [Crossref].
- Karavidas A, Lazaros G, Noutsias M, Matzaraki V, Danias PG, et al. (2011) Recurrent coxsackie B viral myocarditis leading to progressive impairment of left ventricular function over 8 years. Int. J. Cardiol 151: e65-e67. [Crossref]
- Wang L, Dong C, Chen DE, Song Z (2014) Visceral pathology of acute systemic injury in newborn mice on the onset of Coxsackie virus infection. Int J Clin Exp Pathol 7: 890-904. [Crossref]
- de Graaf H, Pelosi E, Cooper A, Pappachan J, Sykes K, et al. (2016) Severe Enterovirus Infections in Hospitalized Children in the South of England: Clinical Phenotypes and Causative Genotypes. Pediatr Infect Dis J 35: 723-727. [Crossref]
- Förster W, Bialasiewicz AA, Busse H (1993) Coxsackievirus B3-associated panuveitis. Br J Ophthalmol 77: 182-183. [Crossref]
- Guan H, Wang J, Wang C, Yang M, Liu L, et al. (2015) Etiology of Multiple Non-EV71 and Non-CVA16 Enteroviruses Associated with Hand, Foot and Mouth Disease in Jinan, China, 2009-June 2013. PLoS One 10: e0142733. [Crossref]
- Goto K, Sanefuji M, Kusuhara K, Nishimura Y, Shimizu H, et al. (2009) Rhombencephalitis and coxsackievirus A16. Emerg Infect Dis 15: 1689-1691. [Crossref]
- Enterovirus detections associated with severe neurological symptoms in children and adults in European countries, Rapid Risk Assessment. Online. Accessed: 01-08-2016.
- Alerte infections sévères entérovirus, Société Française de Pédiatrie. Online. Accessed: 23-Nov-2016.

