Purpose: Mental imagery exercise (MIE) is an innovative technique that has various clinical applications, particularly in physical therapy and rehabilitation. Some Studies had shown the effectiveness of MIE in physical therapy and rehabilitation, particularly for numerous types of sport injuries. The aim of this present review is to provide a critical overview of the current literature on the effectiveness of mental imagery exercise as an innovative physical therapy and rehabilitation technique with respect to the following goals: (i) improving muscular strength, (ii) regaining range of motion, (iii) decreasing pain, (iv) increasing self-efficacy and (v) reducing” kinesiophobia” in knee after Anterior Cruciate Ligament Reconstruction (ACLR).
Method: For accomplishing this objective, our review was focused on the articles published in accordance with the following parameters: English language journals, conducted on patients with Anterior Cruciate Ligament Reconstruction, on both genders, and those that tackle comparison of mental imagery exercises with other exercises. In addition, the PubMed search strategy was used for the identification of the included studies.
Results: Our results showed that some authors suggested a significant improvement in muscle strength and pain reduction after Mental imagery exercise, while other investigators provided results regarding a lack of significance in these variables (i.e. muscle strength and pain). However, all studies had provided evidence related to improved knee range of motion, decreased kinesiophobia, and enhancement in self-efficacy after MIE.
Conclusion: In conclusion, the effectiveness of mental imagery exercise after Anterior Cruciate Ligament Reconstruction could be considered as evidence-based practice in physical therapy and rehabilitation protocol.
Anterior cruciate ligament reconstruction, mental imagery, motor imagery, mental practice, rehabilitation
Anterior Cruciate Ligament (ACL) is one of the most important stabilizing ligaments in the knee joint. Many studies have focused on the mechanisms of ACL injury such as landing on a hyperextended knee, pivoting on planted foot, and sudden or abrupt deceleration [1-3].
Pain, swelling, limited range of motion, progressive muscle weakness, instability, and inability to return to play are the most important clinical outcomes of ACL injury [4]. Moreover, studies that have considered biomechanics, identified three main kinematic consequences of the injury like loss of control of anterior translation of the tibia, a modification of the axis of rotation in the knee joint, and poor synchronization between the lateral femoral condyle and tibia [5]. Therefore, surgical intervention for ACL reconstruction have been recommended as the first choice [6] alongside with physical therapy and rehabilitation strategies which could deeply influence the outcomes [7]. Thus, the role of physical therapy began from the earliest possible phase by using modalities and in particular exercise therapy [8].
Furthermore, many recent studies have suggested that the ACL physical therapy and rehabilitation protocols did not only affect the proprioception, but also, had a great influence on the psychological dimensions which are in a close relationship and are reciprocally influenced by the body conditions [7]. These dimensions were studied for a long time in the past decades and had been proven to enhance the physical therapy and the rehabilitation outcomes in sport injury [4,9].
As one of the cognitive- behavioural approaches, mental imagery exercise, is defined as “Mental imagery is a fully immersive multi-sensory procedure that associates as numerous senses to create a mental image and process it without the presence of external stimuli” [10,11]. There is evidence in support of improving the sensorimotor deficits after ACLR [12] by enhancing the post-operative abilities and performances [13-15], as it has been shown that mental imagery exercises induce activation of the motor region in the brain and consequently, enhance the muscle activity, performance and motor learning [16]. Jackson et al., Hadian et al. provided evidence that the same neurological adaptations in the learning process were found after mental imagery exercises when compared to regular physical exercises [17,18]. Yue and Cole previously explained this suggestion in detail in 1992. They considered that the strength gains after MIE were related to the central programming of a maximal voluntary contraction (MVC) [19]. In addition, in 2010, Akamatsu et al. found that there were significant changes in the Frontal Theta frequency while using cortical activation approach in ACL patients [5].
Additionally, many studies have suggested that mental imagery exercises may allow patients to cope with adverse psychological states related to the injury by controlling some factors such as anxiety after injury, depression and self-confidence [4,9,14,15,20]. In other words, mental imagery is known as an innovative technique in sports injury rehabilitation [21].
Accordingly, researchers suggested different types of mental imagery exercises such as, visual, auditory and kinaesthetic imagery ones [22,23], which were identified to make a global sensory-motor experience in reference to perception.
Thus, despite the difference in impact between the external visual imagery achieved by recruiting a video of a third person and the kinaesthetic imagery [24,25], results in this field remain irrelevant.
Within this context, the aim of this study is to define the evidence-based role of mental imagery exercises in ACLR physical therapy and rehabilitation concerning the enhancement of muscle strength, ROM, knee stability and other psychological effects.
Search strategy (Annexes 1 and 2)
A comprehensive literature search was performed using the following electronic databases: Medline/ PubMed, Scopus, Web of Science, Cochrane library and PEDro after 2001 till Dec. 2019.
Inclusion criteria:
RCT and experimental studies were included,
Studies performed after 2001 to limit our review on the most recent studies in this field,
Articles recruiting ACLR patients for both genders,
Articles comparing mental imagery exercises to other techniques of rehabilitation,
Articles published in English language.
Exclusion criteria
Studies in which the participants were suffering from other knee pathologies, or bilateral knee surgery,
Studies recruiting patients with diabetes, hemophilia, cancer, osteoporosis, or cardiovascular diseases.
Characteristics of the included studies: A total of 567 articles were identified through the literature search (PISMA Flow Chart in Figure 1), 10 duplicate articles were removed, which left 557 articles to study. Out of those studies, 514 articles were excluded after a review of the title and abstract and the full-text versions. The remaining 43 potentially relevant articles were screened, and 37 articles were excluded for not including a specific sample of patients experiencing anterior cruciate ligament reconstruction (n=16), or a pure psychosocial intervention (n=9), or for study design reasons (n=7) and for other reasons (n=6) (Supplementary Appendix 1 and 2).
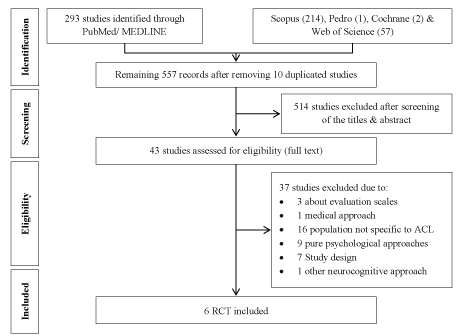
Figure 1. PRISMA Chart.
Sex-relevant studies [4,7,9,20,22,26] met the inclusion criteria and then were encompassed and categorized as shown in (Table 1).
Table 1. Risk of Bias.
| |
R. Maddison, et al. [29] |
Stefano Zaffagnini, et al. [7] |
R. Maddison et al. [9] |
Cupal and Brewer [4] |
Lebon et al. [26] |
Lebon et al. [22] |
|
|
Random sequence generation |
|
|
|
|
|
|
Allocation concealment |
|
|
|
|
|
|
Selective reporting |
|
|
|
|
|
|
Other sources of bias |
|
|
|
|
|
|
Participants and personnel |
|
|
|
|
|
|
|
Low risk of bias |
Outcome assessment |
|
|
|
|
|
|
|
High risk of bias |
Incomplete data |
|
|
|
|
|
|
|
Not clear |
The characteristics of the included studies were presented in Table 2. A total of 237 patients (175 males and 62 females) after anterior cruciate ligament reconstruction were recruited. The patients’ ages ranged from 15 to 53 years. All patients underwent arthroscopic anterior cruciate ligament reconstruction.
Table 2. Characteristics of included studies. IF: Impact Factor, PEDro: PEDro scores.
Article |
Author |
Year |
Title |
Journal |
IF |
PEDro |
Level of Evidence |
1 |
R. Maddison, et al. [29] |
2012 |
Guided imagery to improve functional outcomes post-anterior cruciate ligament repair |
Scandinavian Journal of Medicine & Science in Sports |
4.621 |
7 |
1B |
2 |
Stefano Zaffagnini, et al. [7] |
2013 |
The Video insight_ method: improving rehabilitation following anterior cruciate ligament reconstruction—a preliminary study |
Knee Surgery, Sports Traumatology, Arthroscopy |
3.21 |
9 |
1B |
3 |
R. Maddison et al. [9] |
2006 |
Modeling and rehabilitation following anterior cruciate ligament reconstruction |
Annals of Behavioral Medicine |
3.118 |
5 |
1B |
4 |
Cupal and
Brewer [4] |
2001 |
Effects of Relaxation and Guided Imagery on Knee Strength, Re-injury Anxiety, and Pain Following Anterior Cruciate Ligament Reconstruction |
Rehabilitation Psychology |
3.79 |
8 |
1B |
5 |
Lebon et al. [26] |
2010 |
Benefits of Motor Imagery Training on Muscle Strength |
Journal of Strength and Conditioning Research |
2.06 |
7 |
1B |
6 |
Lebon et al. [22] |
2012 |
Increased Muscle Activation Following Motor Imagery During the Rehabilitation of the Anterior Cruciate Ligament |
Applied Psychophysiology Biofeedback |
1.347 |
5 |
1B |
Methodological quality of the Randomized Controlled Studies: All studies recruited are classified with level 1B of evidence and using the Pedro scale (i.e. based on 11-items), the methodological quality of included studies was assessed. A valid and reliable scale for rating of the quality of randomized trials [27,28]. Giving a score for ten of the 11 items on the scale performed the evaluation and a summed score from 0 to 10 with higher scores reflects a higher methodological quality. Two colleagues, who had no conflict of interest with our review, scored each study. The score was taken by dividing the sum of the two evaluations by 2 then comparing it with previous evaluations cited in previous systematic reviews or mentioned in Pedro site. As a result, scores were from 5 to 9 over 10 (Table 2).
Risk of bias (Table 1): Using the Cochrane Risk of Bias Tool, two authors evaluated the risk of bias in the 6 recruited studies.
The Cochrane risk of bias tool was released in 2008 and updated in 2011. It is based on seven bias domains: sequence generation and allocation concealment (both within the domain of selection bias or allocation bias), blinding of participants and personnel (performance bias), blinding of outcome assessors (detection bias), incomplete outcome data (attrition bias), selective reporting (reporting bias) and an auxiliary domain: “other bias.” For each bias domain, the tool urges users to assign a judgment of “high,” “low” or “unclear” risk of bias and to document the basis for their judgments
Table 1 mentioned that 23.8% of studies have high risk of bias, 11.9% with unclear risk of bias, and the rest are with low risk of bias. Hence, the recruited studies have good methodological qualities according to Cochrane Risk of Bias Tool.
Effects of VRI (Table 3):
Table 3. Results of included studies. MIE: Mental Imagery Exercise; ACL: Anterior Cruciate Ligament; ACLR: Anterior Cruciate Ligament Reconstruction; ROM: Range of Motion; MVC: Maximal Voluntary Contraction; PRISMA: Preferred Reporting Items for Systematic Reviews and Meta-Analysis; EG: Experimental Group; CG: Control Group; IKDC: International Knee Documentation Committee Questionnaire; TSK: Tampa Scale of Kinesiophobia; wks.: weeks; Sig.: Significant; VAS: Visual Analogue Scale; EMG: Electromyography; MR: Maximal Repetition
Article |
Population |
Protocol |
Outcome measures |
Results |
1 |
N=21, 62 % males
34.86 years
(± 8.84 Y)
|
Experimental Group (EG):
Brief relaxation sequence at the beginning of each session
Mental rehearsal activities:
- Focus on the physiological processes occurring during each stage of recovery,
- Suggestions to promote positive coping responses to treatment,
- Imagery modalities (visual and kinesthetic) [25]
- Listening to CDs of the imagery session between appointments.
Cognitive, motivational, and healing imagery [30]
Control Group (CG):
Standard rehabilitation cares |
Knee strength:
Cybex 6000 isokinetic dynamometer Knee
Laxity:
KT1000t Arthrometer
(Baseline and 6-month post-operatively)
Neurobiological factors:
Adrenaline, noradrenaline, and dopamine
(24-h urine sample collected at baseline, 2, 6, and 12-week post-ACLR Rehabilitation)
Rehabilitation self-efficacy:
Athletic Injury Self-Efficacy Questionnaire (AISEQ)
Rehabilitation imagery:
Athletic Injury Imagery Questionnaire-2 (AIIQ-2) [15] |
Knee strength extension:
No statistical differences at 180°/s (P< 0.67), or at 60°/s (P< 0.48) at 6 months
Knee laxity:
Sig. difference: P < 0.05
Greater in the CG (M=- 4.75, SE 2.31) compared with the EG (M= - 1.96, SE 3.15)
Neurobiological Factors:
Sig. decreases in Noradrenalin (P=0.02), and dopamine (P<0.001)
Adrenaline decreases but not sig. effect (P=0.92).
Rehabilitation self-efficacy:
Sig. interaction group × time (P=0.01)
Rehabilitation imagery:
Sig. result for EG with P = 0.05 for motivational, 0.01 for cognitive and 0.001 for healing imagery |
2 |
N=101
Mean 3.0- ± 0.2 months follow-up
Age at surgery was 33.0 ± 17.0 years |
Group A, experimental (EG):
Video that produce positive and therapeutic ‘‘insight’’
Group B, control (CG):
Video that produce ‘insight’’ unfavorable to the psychological recovery
Instructions:
Patients were instructed to watch the video 3 times a week for the first 2 months during the execution of the same conventional rehabilitative protocol |
- Tegner
- IKDC
- Physical and mental SF-36 scores
- Tampa Scale of Kinesiophobia (TSK)
- Time to crutches discharge
Evaluation in pre- and 3 months post-surgery |
IKDC and TKS:
Sig. improvements in EG compared to CG at follow-up for:
IKDC (82.0 ± 13.8 vs. 71.0 ± 19.7, p = 0.0470)
TKS (28.1 ± 6.0 vs. 32.0 ± 5.8, p = 0.0141)
Time to crutches discharge (20.9 ± 5.0 vs. 26.5 ± 8.2 days, p = 0.0012)
Tegner, SF-36 & mental status: Not sig.
TKS & time to crutches discharge:
Sig. correlation (r=0.35, p=0.0121) |
3 |
N=54
68 % males
15 to 53 years |
Modeling intervention (EG):
2 copying model videos:
1st video: shows exercises to improve flex. or ext. walking with and without crutches for 2 wks. post-surgery and prior to discharge,
2nd video: 2 to 6 wks. Post-operative: edited interviews and various action shots of persons performing tasks (ex. walking)
Control Group (CG):
No intervention |
Psychological measures:
- Expected pain and anxiety
- Actual pain
- Anxiety (state-trait anxiety inventory)
- Rehabilitation self-efficacy: crutches self-efficacy, walking self-efficacy and exercise self-efficacy
Functional milestones:
- IKDC
- Crutches usage
- ROM
|
Psychological measures:
Anxiety:
Increases in both groups
Pain:
Expected pain: sig. condition effect (P<0.05)
Actual pain: not sig. condition effect (P=0.52)
Self-efficacy to perform rehabilitation tasks:
Crutches self-efficacy: sig. group differences (P<0.01). EG has greater confidence to walk without crutches
Walking self-efficacy: sig. difference at predischarge (P=0.01)
Exercise self-efficacy: sig. difference at predischarge (P=0.01)
Functional milestones:
IKDC: EG significantly better at 6 wks.
ROM: Not sig. condition effect at 2 and 6 wks. (P<0.76 and 0.36 respectively)
Crutch use: Sig. group difference (P<0.01) EG sent less time on crutches |
4
|
N=30
28.2 ± 8.2 Y
Males and females
|
Physiotherapy started 3 days post operation
Treatment Group (EG):
Relaxation and guided imagery S.:
Videotapes 10 S. /6 months; 1 S. every 2 wks.
Placebo Group (PG):
Attention, encouragement, and support
Control Group (CG):
No intervention |
- Knee strength (Cybex 6000 isokinetic dynamometer)
- Re-injury anxiety
- Pain
- Activity level (Likert-type scale)
|
Knee strength:
Sig. increase at 24 wks. (P<0.05)
Re-injury anxiety and pain:
Sig. decrease for EG at 24 weeks post-surgery
P<0.01 and reduction was more extent for EG (M = 1.09 and 0.54 respectively) |
5 |
N=19
19.75 ± 1.72 years
Healthy students
|
The 2 groups performed identical bench press and leg press exercises
Experimental Group (EG):
9 participants visualize and feel the correspondent contractions during the rest period:
Imagery and actual contraction
Control Group (CG):
10 participants carried out a neutral task |
- The maximal voluntary contraction (MVC) and the maximal number of repetitions (MR) using 80% of the pre-test MVC weight
- Muscle strength in lower and upper limbs
- Anthropometric measures: circumferences of arm, chest and thighs
|
The Leg Press:
Muscle strength: Increase in EG and CG MVC: Significantly higher in EG than in CG (P < 0.05)
MR: Sig. interaction between the leg press MR and the group (EG vs. CG) (P = 0.076)
The Bench Press:
No difference between the EG and CG in MVC & MR
Anthropometric measures:
No sig. difference between pre- and posttest (P> 0.05) |
6 |
N=12
10 men and 2 women
18 to 40 years (28.5 ± 5.0 Y) |
Both groups received 12 sessions of traditional rehabilitation (a 30-min every 2 days):
Strengthening, massages, passive joint mobilization, electro stimulation, cycling without strain and cryotherapy.
Experimental Group (EG):
The instructions were designed to make the patients using kinesthetic imagery rather than pure visual MI
Control Group (CG):
Neutral task: “do not perform any mental training based on movement (e.g., mental calculation or crosswords)” |
Self-estimation of pain:
Visual Analog Scale (VAS)
Activation of vastus medialis:
EMG
Motor ability of lower limb
Lower Extremity Functional Scale (LEFS)
Anthropometrical measures:
Knee circumference
Thigh circumference
ROM of the knee (by goniometer) |
Activation of vastus medialis:
Significantly greater in the EG (P = 0.02)
Self-estimation of pain:
Sig. pain decrease in both groups,
No group significant difference
Motor ability of lower limb
LEFS scores: No sig. difference between groups
Anthropometrical measures:
No sig. group difference (P>0.05)
Circumferences during last session provide evidence of a sig. muscle atrophy in both groups |
Maddison et al. in 2012 found that the use of mental rehearsal activities with 21 patients with ACLR led to an improvement of knee stability (knee laxity was greater with control group than experimental group (M=- 4.75, SE 2.31) and (M =- 1.96, SE 3.15) respectively). In addition, the rehabilitation self-efficacy and the neurobiological factors changed significantly within the mental imagery group more than the control group (P=0.01) [29,30].
Moreover, Zaffagnini et al. in 2013 reported a significant improvement in the IKDC and TKS assessments, with positive video-insight compared to with negative video-insight interventions (IKDC (82.0 ± 13.8 vs. 71.0 ± 19.7, p=0.0470) and TKS (28.1 ± 6.0 vs. 32.0 ± 5.8, p=0.0141)). Also, the time to use crutches changed significantly with first group (20.9 ± 5.0 vs. 26.5 ± 8.2 days, p=0.0012) [7].
The modelling-video intervention had been proven to be more effective than the one with no intervention, with a significant decrease of expected pain (P<0.05) and an increase of crutches self-efficacy, walking self-efficacy and exercises self-efficacy (P ≤ 0.01. Also, the IKDC changed significantly with experimental group after 6 weeks of treatment [9].
Similarly, the relaxation combined with guided imagery led to a significant increase in knee strength (P<0.05) with a decrease of re-injury anxiety (P<0.01) after 24 weeks post-treatment as compared with placebo or control groups [4].
Moreover, Lebon et al. showed that combining the imagery with actual contraction led to a significant increase of muscle strength and MVC (P<0.05) when compared to imagery alone [26].
Finally, in another study Lebon et al. stated that the use of kinesthetic imagery post-ACLR compared to neutral tasks induced a significant increase of the activation of vastus medialis (P=0.02) with a decrease of pain (P<0.05) [22].
A comprehensive literature search was carried out using Medline/ PubMed, Scopus, Web of Science, Cochrane library and PEDro in order to find out the effectiveness of MIE in accelerating the recovery after sport injury [9,14,20]. Therefore, the main aims of this review were to explain the probable effects and potential mechanisms underlying the functional and psychological measures of MIE.
Functional measures: Based on our research, imagery exercises showed to be an effective technique in improving functional outcomes such as muscle strength, ROM walking ability and increasing knee stability [20,22,26].
The positive effect of guided imagery exercise on knee stability reported by Maddison et al. [20], which was in line with Glaser’s results [31], was correlated with a decrease in the level of adrenaline and dopamine reflecting lower level of stress, improved healing (KT 1000 scores) and reduced knee laxity in the intervention group. In another study, Maddison et al. showed significant improvement in the knee functional outcomes assessed by IKDC and during crutches usage. They attributed these changes due to an early stimulus for improving the muscle strength after 6 weeks of training using modelling videotape of functional exercises [9].
Using video insight method, similar findings were reported by Zaffagnini et al. [7], as they suggested that adding a video instruction or feedback to the conventional rehabilitation protocol could positively affect the short-term clinical outcomes (i.e. 3 months follow-up).
In the neurophysiological aspect, Cupal and Brewer suggested the possible role of MIE on improvement of muscle strength and functional measures. They studied the effect of combined relaxation and MIE [4] after ACLR and attributed the improvement to the modulation of corticospinal excitability [32,33] and the modification of programming in the brain [19], a larger brain area that was responsible for controlling these muscles might increase this improvement [26,34]. Lebon et al. confirmed these suggestions, when they recorded an increase in EMG activity without any changes in muscle circumferences after MIE training. Their findings might be related to neurophysiological rather structural modifications in the muscles [34,35].
In a different point of view, Lebon et al. stressed on the effect of combination of MIE and physical training. They reported a more significant increase of strength in the lower limb muscles compared with the upper limb muscles [26]. Consequently, we believe that the generalization of reorganization’s concept related to the cortical representation needs more studies with larger sample size.
Sidaway et al. and Ranganthan et al. [34,36] opened another window by suggesting that the mental repetitions of a muscle contraction might lead to stronger outputs from the brain to target muscle by recruiting more motor units or reducing the inhibitory inputs on the motoneurone pool. We also agree that the neural plasticity could act as a promoting factor in this regard, which is in accordance with previous findings in evoked potential recording and electrophysiological tools after stroke [37,38] and limb amputations [39] as well as after knee arthroplasty [40].
Lebon et al. reported an improvement of 37.86 degrees in knee ROM by using motor imagery added to the conventional rehabilitation protocol. However, Maddison et al. in a modelling design study reported no significant improvement regarding the ROM assessed 3 and 6 weeks after ACLR [9].
It is worthy to mention that other variables may affect the impact of MIE on functional and psychological outcomes of the patients. Olsson & Nyberg found that the imagery perspective, the complexity of task imagined, and previous physical experience must also be considered among the factors that produce an optimal result in sport rehabilitation [41].
Psychological measures: pain, anxiety and self-evaluation of the treatment efficacy: It was established that neurocognitive intervention based on mental imagery exercises could reduce anxiety [42-44]. With this regard, Cupal et al. mentioned that the combination of relaxation and mental imagery exercises led to significant decrease of pain and re-injury anxiety [4,42]. However, we believe that the small sample size (10 participants) was a limitation for the generalization of their findings. Lebon et al. [22], focusing on motor recovery, reported a significant pain decrease in both control and experimental groups (Table 1). They considered that taking analgesic by participants might be a primary cause for the absence of significant differences among groups. This could have been a weakness in their study and perhaps biased the effect of MIE on pain relief.
More evidence was provided by Maddison et al. who reported a reduction in preoperative perceptions of anxiety and pain as well as an increase in postoperative rehabilitation self-efficacy (i.e., use of crutches, predischarge walking and exercise self-efficacy) and in motivation after modelling video intervention in ACLR patients. Their results showed a significant condition effect for perception of the expected pain (but not for the actual pain) and a decrease in rehabilitation self-efficacy in both groups (i.e. MIE and control). However, it was more effective for MIE group, especially at 2 and 6 weeks after surgery. Thus, they concluded that the decrease of patients’ expectation after surgery was because of the symptomatic factors such as pain, muscle weakness and limited ROM [45-48]. Other variables such as self-consideration and intrinsic motivation were studied by Zaffagnini et al. who reported significant improvements by using specific images [7].
The aim of this study was to explore the effectiveness of mental imagery exercises as a neurocognitive-based approach on ACLR patients. Accordingly, we found out a large number of researches that suggested that MIE increase muscle strength, range of motion, and knee stability. It, also, decreases pain and kinesiophobia when implemented by professional and experienced physiotherapists.
However, the results of the current review showed that there are still some debates in this regard particularly when MIE was used alone. Accordingly, we suggest that the combined method including mental imagery and active physical exercises will be the most effective technique [18,26]. Thus, for recommending a guideline, more evidence is needed in future studies through well-designed protocols with a larger sample size.
• Only 6 randomized control trials with good methodological quality handled the effect of mental imagery with patients after ACLR.
• These RCTs show that there are still some debates regarding the protocol to be used to increase muscle strength, range of motion, and knee stability and decrease kinesiophobia.
Competing interests: The authors declare that they have no competing interests.
Declaration of Conflicts of Interest: The author(s) declared no potential conflicts of interest with respect to the ethical and research considerations in this publication.
Funding: The author(s) received no financial support for the research, and/or publication of this article.
Authors' contributions:
• Hussein Ziab: main author, collecting data, analysis, and discussion.
• Rami Mazbouh and Rahimeh Mahmoodi: Reviewing and editing.
• Tony Gebran, Salam Hussein, Mohammad Wehbe, Amir Kanj and Mohammad Taha: data collection, screening, evaluating level of evidence.
• Mohammad Reza Hadian: corresponding author, editing and reviewing data analysis and discussion.
We would like to thank Mr Ali Mansour, Physiotherapist from USA for his valuable contribution in the language editing.
Ethics approval: Not applicable
Consent to participate: Not applicable
Consent for publication: Not applicable
Availability of Data and Materials: Not applicable
- Medvecky MJ, Nelson S (2015) Kinesiophobia and Return to Sports After Anterior Cruciate Ligament Reconstruction. Conn Med 79: 155-157. [Crossref]
- Kobayashi H, Kanamura T, Koshida S, Miyashita K, Okado T, et al. (2010) Mechanisms of the Anterior Cruciate Ligament Injury in Sports Activities: A Twenty-Year Clinical Research of 1,700 Athletes. J Sports Sci Med 9: 669-675. [Crossref]
- Shimokochi Y, Shultz SJ (2008) Mechanisms of Noncontact Anterior Cruciate Ligament Injury. J Athl Train 43: 396-408. [Crossref]
- Cupal DD, Brewer BW (2001) Effects of relaxation and guided imagery on knee strength, reinjury anxiety, and pain following anterior cruciate ligament reconstruction. Rehabil Psychol 46: 28-43.
- Akamatsu Y, Mitsugi N, Taki N, Takeuchi R, Saito T (2010) Simultaneous anterior cruciate ligament reconstruction and opening wedge high tibial osteotomy: Report of four cases. The Knee 17: 114-118. [Crossref]
- Eberhardt C, Jäger A, Schwetlick G, Rauschmann MA (2002) [History of surgery of the anterior cruciate ligament]. Orthopade 31: 702-709. [Crossref]
- Zaffagnini S, Russo RL, Marcheggiani Muccioli GM, Marcacci M (2013) The Videoinsight® method: improving rehabilitation following anterior cruciate ligament reconstruction--a preliminary study. Knee Surg Sports Traumatol Arthrosc 21: 851-858. [Crossref]
- Biggs A, Jenkins WL, Urch SE, Shelbourne KD (2009) Rehabilitation for Patients Following ACL Reconstruction: A Knee Symmetry Model. N Am J Sports Phys Ther 4: 2-12. [Crossref]
- Maddison R, Prapavessis H, Clatworthy M (2006) Modeling and rehabilitation following anterior cruciate ligament reconstruction. Ann Behav Med 31: 89-98. [Crossref]
- Cumming J, Williams SE (2012) The Role of Imagery in Performance. In SM Murphy (Ed.), Oxford library of psychology. The Oxford handbook of sport and performance psychology. Oxford University Press. pp: 213-232.
- Di Corrado D, Guarnera M, Guerrera CS, Maldonato NM, Di Nuovo S, et al. (2020) Mental Imagery Skills in Competitive Young Athletes and Non-athletes. Front Psychol 11: 633. [Crossref]
- Slimani M, Tod D, Chaabene H, Miarka B, Chamari K (2016) Effects of Mental Imagery on Muscular Strength in Healthy and Patient Participants: A Systematic Review. J Sports Sci Med 15: 434-450. [Crossref]
- Ievleva L, Orlick T (1991) Mental Links to Enhanced Healing: An Exploratory Study. Sport Psychol 5: 25-40.
- Sordoni C, Hall C, Forwell L (2000) The use of imagery by athletes during injury rehabilitation. J Sport Rehabil 9: 329-338.
- Sordoni C, Hall C, Forwell L (2002) The use of imagery in athletic injury rehabilitation and its relationship to self-efficacy. Physiotherapy Canada 54: 177-185.
- Guillot A, Collet C (2008) Construction of the Motor Imagery Integrative Model in Sport: a review and theoretical investigation of motor imagery use. Int Rev Sport Exerc Psychol 1:31-44.
- Jackson PL, Lafleur MF, Malouin F, Richards CL, Doyon J (2003) Functional cerebral reorganization following motor sequence learning through mental practice with motor imagery. NeuroImage 20: 1171-1180. [Crossref]
- Hedayati R, Oliaee GR, Hadian MR, Talebian Moghaddam S, Baghery H (2004) The Effect of Mental Practice on Increasing Maximal Muscle Strength in 20-30-year-old Healthy women. J Rafsanjan Univ Med Sci 3: 119-125.
- Yue G, Cole KJ (1992) Strength increases from the motor program: comparison of training with maximal voluntary and imagined muscle contractions. J Neurophysiol 67: 1114-1123. [Crossref]
- Maddison R, Prapavessis H, Clatworthy M, Hall C, Foley L, et al. (2012) Guided imagery to improve functional outcomes post-anterior cruciate ligament repair: randomized-controlled pilot trial. Scand J Med Sci Sports 22: 816-821. [Crossref]
- Slimani M, Chamari K, Boudhiba D, Chéour F (2016) Imagery use-motor learning and sport performance relationships.
- Lebon F, Guillot A, Collet C (2012) Increased muscle activation following motor imagery during the rehabilitation of the anterior cruciate ligament. Appl Psychophysiol Biofeedback 37: 45-51. [Crossref]
- Kosslyn SM, Segar C, Pani J, Hillger LA (1990) When is imagery used in everyday life? A diary study. J Ment Imag 14: 131-152.
- Ranganathan VK, Kuykendall T, Siemionow V, Yue GH (2002) Level of mental effort determines training induced strength increases. Abstr Soc Neurosci 32.
- Guillot A, Collet C, Dittmar A (2004) Relationship Between Visual and Kinesthetic Imagery, Field Dependence-Independence, and Complex Motor Skills. J Psychophysiol 18: 190-198.
- Lebon F, Collet C, Guillot A (2010) Benefits of motor imagery training on muscle strength. J Strength Cond Res. 24: 1680-1687. [Crossref]
- De Morton NA (2009) The PEDro scale is a valid measure of the methodological quality of clinical trials: a demographic study. Aust J Physiother 55: 129-133. [Crossref]
- Maher CG, Sherrington C, Herbert RD, Moseley AM, Elkins M (2003) Reliability of the PEDro scale for rating quality of randomized controlled trials. Phys Ther 83: 713-721. [Crossref]
- Maddison R, Prapavessis H, Clatworthy M, Hall C, Foley L, et al. (2012) Guided imagery to improve functional outcomes post-anterior cruciate ligament repair: randomized-controlled pilot trial. Scand J Med Sci Sports 22: 816-821. [Crossref]
- Driediger M, Hall C, Callow N (2006) Imagery use by injured athletes: a qualitative analysis. J Sports Sci 24: 261-271. [Crossref]
- Glaser R, Kiecolt-Glaser JK, Marucha PT, MacCallum RC, Laskowski BF, et al. (1999) Stress-related changes in proinflammatory cytokine production in wounds. Arch Gen Psychiatry 56: 450-456. [Crossref]
- Stinear CM, Byblow WD, Steyvers M, Levin O, Swinnen SP (2006) Kinesthetic, but not visual, motor imagery modulates corticomotor excitability. Exp Brain Res 168: 157-164. [Crossref]
- Lotze M, Montoya P, Erb M, Hülsmann E, Flor H, et al. (1999) Activation of cortical and cerebellar motor areas during executed and imagined hand movements: an fMRI study. J Cogn Neurosci 11: 491-501. [Crossref]
- Ranganathan VK, Siemionow V, Liu JZ, Sahgal V, Yue GH (2004) From mental power to muscle power--gaining strength by using the mind. Neuropsychologia 42: 944-956. [Crossref]
- Zijdewind I, Toering ST, Bessem B, Van Der Laan O, Diercks RL (2003) Effects of imagery motor training on torque production of ankle plantar flexor muscles. Muscle Nerve 28: 168-173. [Crossref]
- Sidaway B, Trzaska AR (2005) Can Mental Practice Increase Ankle Dorsiflexor Torque? Phys Ther 85: 1053-1060. [Crossref]
- Dimyan MA, Cohen LG (2011) Neuroplasticity in the context of motor rehabilitation after stroke. Nat Rev Neurol 7: 76-85. [Crossref]
- Hernandez A (2015) Evoked Potentials as Neurophysiologic Tools to Evaluate Stroke. J Neurol Stroke 2: 00046.
- Karl A, Birbaumer N, Lutzenberger W, Cohen LG, Flor H (2001) Reorganization of Motor and Somatosensory Cortex in Upper Extremity Amputees with Phantom Limb Pain. J Neurosci 21: 3609-3618. [Crossref]
- Moukarzel M, Guillot A, Di Rienzo F, Hoyek N (2019) The therapeutic role of motor imagery during the chronic phase after total knee arthroplasty: a pilot randomized controlled trial. Eur J Phys Rehabil Med 55: 806-815. [Crossref]
- Olsson C-J, Nyberg L (2010) Motor imagery: if you can’t do it, you won’t think it. Scand J Med Sci Sports 20: 711-715. [Crossref]
- Pearson J, Naselaris T, Holmes EA, Kosslyn SM (2015) Mental Imagery: Functional Mechanisms and Clinical Applications. Trends Cogn Sci 19: 590-602. [Crossref]
- VandeCreek L (1998) Innovations in Clinical Practice: A Source Book. Sarasota, FL: Professional Resource Exchange Inc.
- Evans L, Hare R, Mullen R (2006) Imagery Use During Rehabilitation from Injury. J Imag Res Sport Phys Act 7: 1.
- Bandura A (1997) Self-Efficacy: The Exercise of Control. 1st edition. New York: Worth Publishers.
- Flint FA (1992) The psychological effects of modeling in athletic injury rehabilitation.
- Melamed BG, Siegel LJ (1975) Reduction of anxiety in children facing hospitalization and surgery by use of filmed modeling. J Consult Clin Psychol 43: 511-521. [Crossref]
- Padilla GV, Grant MM, Rains BL, Hansen BC, Bergstrom N, et al. (1981) Distress reduction and the effects of preparatory teaching films and patient control. Res Nurs Health 4: 375-387. [Crossref]

