Introduction
The human body is colonized by 100 trillion of microorganisms, including bacteria, archaea, viruses and very tiny eukaryotes [1]. In particular, the total number of bacteria in a reference man is reported to be around 3.8·1013. The total number of fungal cells, the so-called ‘mycobiome’ is orders of magnitude smaller than that of bacterial cells [2]. Together with other less abundant (<0.1%) microorganisms fungi are component of the rare biosphere [3,4] which significantly impact human health protection from pathogens [5].
The definition of the 2021 Copyright OAT. All rights reservrminology confusion since the term “microbiota” and “microbiome” are often used interchangeably. According to a correct definition, the term “microbiota” has to be referred to the microbial community associated with humans and the term “microbiome” both to microbes and the genes they share with humans.
Early study on human microbiome started in the 1680s by Antonie van Leeuwenhoek [6,7] highlighting for the first time the profound differences in the microbial community at different body sites.
Once these difference became obvious severals large-scale microbiome projects have been launched all around the world [8-10] in order to explain the reasons behind microbial diversity and factor affecting it by mean of both culture-dependent and independent methods.
Advances in molecular and genetic techniques as well growing interest within the field resulted in an increase of the number of scientific publications on the topic, especially after the so-called “Microbiome Boom” between 2012 and 2013s (Figure 1).
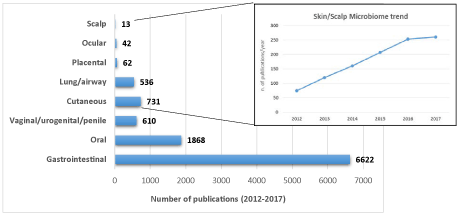
Figure 1. Microbiome research on different body sites. Number of results obtained by using the following keywords: “microbiome”, “microbiota”, “microflora”, “<body site>” on PubMed (retrieved 31 December 2017)
Eubiotic microbiome for the human health
Different species of bacteria, fungi, and virus may inhabit the human body. Bacterial members belong mainly to Actinobacteria, Firmicutes, Proteobacteria, and Bacteroidetes phyla [11-14]. Each individual posses unique microbial communities residing in different anatomic site and each of this site harbor a peculiar microbial community, performing a specific function [7,15-18].
More similarity has been found between microbial communities at the same body site than between different body sites in the same individual [7]. At the same time, a large degree of intrapersonal variation has been found even in the absence of disease [7,19].
Microbiome biodiversity evolves fast from birth till the first year of life [20,21] continuously increasing its complexity with age [22-25].
Since human microbiome is characterized by a great variability the identification of a group of microorganisms related to health status is not simple [26,27]. For the first time, in 2014, Shafqat and co-authors [28] changed the perspective by linking the health status to a core of metabolic and biological functions elicited by the microbiome in each body sited, independently from the microorganisms that would provide these functions. This is strictly related to higher consistency of the metabolic pathways across people compared to microbiome diversity [26]. Therefore in several recent studies high microbial diversity has been correlated to health [29] and temporal stability [30]. A healthy microbiome corresponds with a status of balance beneficial for the host. This state is also known as eubiosis. When extrinsic or intrinsic factors alter this balance microbiome switches to dysbiosis, losing in most cases, its biodiversity.
Many different factors can influence microbiome biodiversity either positively and negatively. Among these i) age [20,21,31]; ii) environmental factors [32]; iii) diet [33-36]; iv) inheritance [37]; v) antibiotics consumption [38,39]. To maintain is state of healthiness, the microbiome has to possess two main property: “resistance” and “resilience”, that ‘s to say the ability to return to the previous healthy state [26,40].
There is an increasing evidence that lower degree of resistance and especially resilience may lead to several human disease predisposition as to the prevalence of various human disorders [41] as a consequence of microbiome dysbiosis.
Disease such as dental caries, bacterial vaginosis, obesity, autoimmune disease, diabetes, gastrointestinal and dermatological diseases have been linked to changing in microbial eubiotic balance [41]. More and more evidence is also accumulating as regards intercorrelation between microbiome, especially the intestinal one, and central nervous system and behavior [42,43]. More interesting, recently, the presence of a larger axis, the gut-brain-skin axis has been validated [44,45] paving the way to new therapeutic approaches.
The cutaneous and scalp microbiome
As the largest organ in human body [46,47] skin hosts an enormously diverse arrangement of the microorganism [48]. They belonging mainly to genus Corynebacterium, Propionibacterium, and Staphylococcus [11-14]. Microorganisms from this genus are differently predominant in each skin sites [14,49] and this depending on variability in terms of water content, r of sebaceous glands, temperature, exposure to the environment [14]. For example drier sites showed Proteobacteria as the main phyla; on the contrary skin sites enriched in sebaceous glands are populated mainly by species belonging to Actinobacteria such as suborders of Propionibacterineae and Corynebacterineae.
Study the role of skin microbiome for human health is acquiring importance day by day. First studies have been focused on pathogens affecting the skin but more recent works highlighted the crucial role of resident microbial communities in skin healthiness [50-52]. Microorganisms, establishing a symbiotic relationship with the skin, and this results, in the majority of the time, as beneficial both for microbial community and for the host [53-55]. Many skin conditions have been linked to an imbalance in this symbiotic mutualistic relationship [50,56-58]. For example, a diminishing in the abundance of Propionibacterium and Actinobacteriacae has been linked to persistent skin infections [59]. Other studies reported the higher expression of Firmicutes and lower expression of Actinobacteria in psoriatic lesions [47,60]. A modification of the microbiome has also been shown to be implicated in another chronic inflammatory condition, atopic dermatitis [47]. The implication of Propionibacterium acnes in acne development is well established [61] but, more recently the involvement of other microorganisms has also been postulated [52]. Therefore, it has been shown that Staphylococcus epidermidis and Staphylococcus aureus inversely correlate each other and shifting of this correlation may increase the risk of skin diseases' onset [50].
Study of skin microbiome represent a novel diagnostic and therapeutic approach to many skin conditions, also including that strictly related to the scalp.
Although obviously sharing some characteristics with skin, scalp presents some distinctive tracts such as thickness, hair growth, more blood vessels and more sebaceous glands. These unique features expose scalp area to different peculiar diseases. As part of the skin organ, the scalp is in a symbiotic relationship with microbial communities that inhabits it and, since scalp unique features, this community is expected to be peculiar. As shown in Figure 1, a number of publications on the microbiome related to scalp area is very limited and published works are mainly related to the role of scalp microbiome in dandruff [62,63]. Thanks to advances in sequencing and metagenomic, new tools are currently available to study the role of the microbiome.
No studies are currently reported as regards microbiome involvement in scalp conditions related to hair growth, such as androgenetic alopecia, alopecia areata, and scarring alopecia. Considering the impact of such disease on human health and their increasing incidence, the understanding on the impact of the microbiome and related changes could represent an improvement in clinical practice as well a tool for the development of advanced targeted therapeutic approaches.
Here we show our preliminary results on a panel of about 15 subjects for group affected by androgenetic alopecia (AGA), alopecia areata (AA), and lichen planopilaris (LPP), respectively. Each group has been compared to a panel of healthy subjects.
The main bacterial species (P. acnes, S. epidermidis, and S.aureus) inhabiting the scalp subject’s were identified by quantitative PCR using species-specific primers.
All scalp conditions analyzed were correlated with a higher incidence of P. acnes compared to the population used as control (p<0,005). Additionally, in subjects affected by AGA ratio between S. epidermidis and S. aureus diminished. This diminishing is more evident in subjects affected by AA where S. aureus becomes predominant on S. epidermidis. The role of P. acnes in the pathogenesis of hair casts and alopecia has previously been hypothesized [64] and our study represents the first clinical evidence correlating microbiome unbalancing to scalp hair disorders.
In subjects affected by LPP, a form of scarring alopecia, the predominance of P. acnes persists but is less evident than in the previous diseases analyzed (Figure 2). This could be linked to the loss of sebaceous glands commonly found in patients affected by LPP [65]. Our results showed also a higher S. epidermidis/S. aureus. It would be interesting, in future works, to analyze how this ratio would change according to a different stage of inflammation and in more lesional forms.
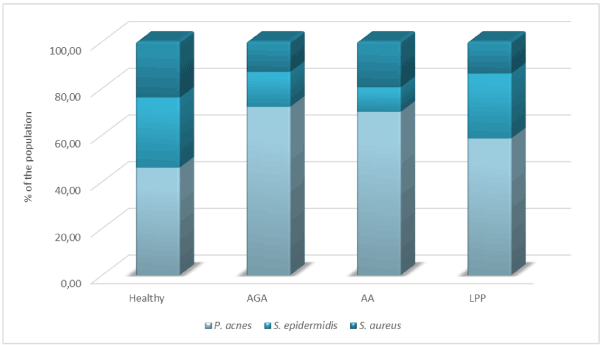
Figure 2. RT-qPCR quantification (% of the population) of main bacterial species (P. acnes, S. epidermidis, and S.aureus) inhabiting the scalp in the subject’s affected by androgenetic alopecia (AGA), alopecia areata (AA), and lichen planopilaris (LPP), respectively. N=15
Conclusion
Nowadays, the study of human microbiome represents a novel diagnostic and therapeutic approach to treat many human conditions, also including that strictly related to skin and scalp.
The findings we included in the present work represent just an overview of a larger pioneer study on the involvement of changing of the microbiome in scalp diseases, especially that related to hair growth.
Even just preliminary, our results strongly highlighted, for the first time, the role exerted by unbalancing on the normal resident microbial community in hair growth-related conditions.
References
- Sender R, Fuchs S, Milo R (2016) Revised Estimates for the Number of Human and Bacteria Cells in the Body. PLoS Biol 14: e1002533.
- Huffnagle GB, Noverr MC (2013) The emerging world of the fungal microbiome. Trends Microbiol 21: 334-41.
- Sogin ML, Morrison HG, Huber JA, Mark Welch D, Huse SM, et al. (2006) Microbial diversity in the deep sea and the underexplored "rare biosphere". Proc Natl Acad Sci U S A 103: 12115-12120.
- Dethlefsen L, Huse S, Sogin ML, Relman DA (2008) The pervasive effects of an antibiotic on the human gut microbiota, as revealed by deep 16S rRNA sequencing. PLoS Biol 6: e280. [Crossref]
- Hajishengallis G, Liang S, Payne MA, Hashim A, Jotwani R, et al. (2011) Low-abundance biofilm species orchestrates inflammatory periodontal disease through the commensal microbiota and complement. Cell Host Microbe 10: 497-506.
- van Leeuwenhoek A: An abstract of a Letter from Antonie van Leeuwenhoek (1683) About Animals in the scrurf of the Teeth. Philosophical Transactions of the Royal Society of London 1684, 14: 568-574.
- Qin J, Li R, Raes J, Arumugam M, Burgdorf KS, et al (2010) A human gut microbial gene catalogue established by metagenomic sequencing. Nature 464: 59-65.
- Costello EK, Lauber CL, Hamady M, Fierer N, Gordon JI, et al. (2009) Bacterial community variation in human body habitats across space and time. Science 326: 1694-97. [Crossref]
- Qin J, Li Y, Cai Z, Li S, Zhu J, et al (2012) A metagenome-wide association study of gut microbiota in type 2 diabetes. Nature 490: 55-60.
- Li J, Jia H, Cai X, Zhong H, Feng Q, et al. (2014) An integrated catalog of reference genes in the human gut microbiome. Nat Biotechnol 32: 834-841. [Crossref]
- Dethlefsen L, McFall-Ngai M, Relman DA (2007) An ecological and evolutionary perspective on human-microbe mutualism and disease. Nature 449: 811-818. [Crossref]
- Grice EA, Kong HH, Renaud G, Young AC, Bouffard GG, et al. (2008) A diversity profile of the human skin microbiota. Genome Res 18:1043-50. [Crossref]
- Reid G, Younes JA, Van der Mei HC, Gloor GB, Knight R et al. (2011) Microbiota restoration: natural and supplemented recovery of human microbial communities. Nat Rev Microbiol 9: 27-38.
- Huttenhower C, Gevers D, Knight R, Abubucker S, Badger JH, et al. (2012) Structure, function and diversity of the healthy human microbiome. Nature 486: 207-214.
- Grice EA, Kong HH, Conlan S, Deming CB, Davis J, et al. (2009) Topographical and temporal diversity of the human skin microbiome. Science 324: 1190-1192.
- Ravel J, Gajer P, Abdo Z, Schneider GM, Koenig SS, et al. (2011) Vaginal microbiome of reproductive-age women. Proc Natl Acad Sci U S A 108: 4680-4687.
- Wade WG (2013) The oral microbiome in health and disease. Pharmacol Res 69: 137-143.
- Rajilic-Stojanovic M, de Vos WM (2014) The first 1000 cultured species of the human gastrointestinal microbiota. FEMS Microbiol Rev 38: 996-1047.
- Palmer C, Bik EM, DiGiulio DB, Relman DA, Brown PO, et al. (2007) Development of the human infant intestinal microbiota. PLoS Biol 5: e177.
- Eggesbo M, Moen B, Peddada S, Baird D, Rugtveit J, et al. (2011) Development of gut microbiota in infants not exposed to medical interventions. Acta Pathol Microbiol Immunol Scand 119: 17-35.
- Morgan XC, Segata N, Huttenhower C (2013) Biodiversity and functional genomics in the human microbiome. Trends in genetics 29: 51-58.
- Yatsunenko T, Rey FE, Manary MJ, Trehan I, Dominguez-Bello MG, et al. (2012) Human gut microbiome viewed across age and geography. Nature 7402: 222-227.
- Ding T, Schloss PD (2014) Dynamics and associations of microbial community types across the human body. Nature 509: 357-360.
- Goodrich JK, Davenport ER, Waters JL, Clark AG, Ley RE, et al. (2016) Cross-species comparisons of host genetic associations with the microbiome. Science 29: 532-535
- Moya A, Ferrer M (2016) Functional redundancy-induced stability of gut microbiota subjected to disturbance. Trends Microbiol 24: 402-113.
- Shafquat A, Joice R, Simmons SL, Huttenhower C (2014). Functional and phylogenetic assembly of microbial communities in the human microbiome. Trends Microbiol 22: 261-266.
- Lloyd-Price J, Abu-Ali G, Huttenhower C (2016) The healthy human microbiome. Genome Medicine 8: 51.
- Turnbaugh PJ, Ley RE, Hamady M, Fraser-Liggett CM, Knight R, et al. (2007) The human microbiome project. Nature 449: 804-810.
- Mariat D, Firmesse O, Levenez F, Guimar?es V, Sokol H (2009) The Firmicutes/ Bacteroidetes ratio of the human microbiota changes with age. BMC Microbiol 9: 9-123.
- Flores GE, Caporaso JG, Henley JB, Rideout JR, Domogala D, et al. (2014) Temporal variability is a personalized feature of the human microbiome. Genome Biol 15: 1-531.
- Blaser MJ, Falkow S (2009) What are the consequences of the disappearing human microbiota? Nat Rev Microbiol 7: 887-894.
- De Filippo C, Cavalieri D, Di Paola M, Ramazzotti M, Poullet JB, et al. (2010) Impact of diet in shaping gut microbiota revealed by a comparative study in children from Europe and rural Africa. Proc Natl Acad Sci U S A 107: 14691-14696.
- Turnbaugh PJ, Ridaura VK, Faith JJ, Rey FE, Knight R, et al. (2009) The effect of diet on the human gut microbiome: a metagenomic analysis in humanized gnotobiotic mice. Sci Transl Med 11: 6ra14. [Crossref]
- Cho I, Blaser MJ (2012) The Human Microbiome: at the interface of health and disease. Nature reviews Genetics 13: 260-270. [Crossref]
- David LA, Maurice CF, Carmody RN, Gootenberg DB, Button JE, et al. (2014) Diet rapidly and reproducibly alters the human gut microbiome. Nature 505: 559-563.
- Gorvitovskaia A, Holmes SP, Huse SM, et al. (2016) Interpreting prevotella and bacteroides as biomarkers of diet and lifestyle. Microbiome 12: 4-15.
- Grice EA, Segre JA (2012) The human microbiome: Our second genome. Annu Rev Genomics Hum Genet 13: 151-170. [Crossref]
- Cho I, Yamanishi S, Cox L, Methe BA, Zavadil J, et al. (2012) Antibiotics in early life alter the murine colonic microbiome and adiposity. Nature 488: 621-626.
- Livanos AE, Greiner TU, Vangay P, Pathmasiri W, Stewart D, et al. (2016) Antibioticmediated gut microbiome perturbation accelerates development of type 1 diabetes in mice. Nat Microbiol 1: 16140. [Crossref]
- Bäckhed F, Fraser CM, Ringel Y, Sanders ME, Sartor RB, et al. (2012) Defining a healthy human gut microbiome: current concepts, future directions, and clinical applications. Cell Host Microbe12: 611-622.
- Pflughoeft KJ, Versalovic J (2017) Human microbiome in health and disease. Annu Rev Pathol 7: 99-122.
- Dinan TG, Cryan JF (2017) The microbiome-gut-brain axis in health and disease. Gastroenterol Clin North Am 46: 77-89.
- Li Q, Han Y, Dy ABC, Hagerman RJ, et al. (2017) The gut microbiota and autism spectrum disorders. Frontiers in Cellular Neuroscience 11: 120.
- Arck P, Handjiski B, Hagen E, Pincus M, Bruenahl C, et al. (2010) Is there a 'gut-brain-skin axis'? Exp Dermatol 19: 401-405. [Crossref]
- Bowe WP, Logan AC (2011) Acne vulgaris, probiotics and the gut-brain-skin axis - back to the future? Gut Pathogens 3:1.
- Grice EA, Segre JA (2011) The skin microbiome. Nat Rev Microbiol 9: 244-253.
- Ribeiro A, Estanqueiro M, Oliveira M, Sousa Lobo J (2015) Main benefits and applicability of plant extracts in skin care products. Cosmetics 2: 48-65.
- Findley K, Grice EA (2014) The skin microbiome: A focus on pathogens and their association with skin disease. PLoS Pathog 10: e1004436. [Crossref]
- Kong HH (2011) Skin microbiome: genomics-based insights into the diversity and role of skinmicrobes. Trends Mol Med 17:320-28. [Crossref]
- Cogen AL, Nizet V, Gallo RL (2009) Skin microbiota: A source of disease or defence? Br J Dermatol 158: 442-455.
- Grice EA (2014) The skin microbiome: potential for novel diagnostic and therapeutic approaches to cutaneous disease. Sem Cut Med Surg 33: 98-103. [Crossref]
- Dreno B, Martin R, Moyal D, Henley JB, Khammari A, et al. (2017) Skin microbiome and acne vulgaris: Staphylococcus, a new actor in acne. Exp Dermatol 26: 798-803. [Crossref]
- Noble WC (2004) Staphylococci on the skin. In The Skin Microflora and Microbial Skin Disease; Noble, WC, Edn.Cambridge University Press: London, UK,135-152.
- Katsuyama M, Ichikawa H, Ogawa S, Ikezawa Z (2005) A novel method to control the balance of skin microflora: Part 1. Attack on biofilm of Staphylococcus aureus without antibiotics. J Dermatol Sci 38 197-205. [Crossref]
- Lambers H, Piessens S, Bloem A, Pronk H, Finkel P, et al. (2006) Natural skin surface pH is on average below 5, which is beneficial for its resident flora. Int J Cosmet Sci 28: 359-370. [Crossref]
- Brogden NK, Mehalick L, Fischer CL, Wertz PW, Brogden KA, et al. (2012) The emerging role of peptides and lipids as antimicrobial epidermal barriers and modulators of local inflammation. Skin Pharmacol Physiol 25: 167-181. [Crossref]
- Zeeuwen PL, Kleerebezem M, Timmerman HM, Schalkwijk J (2013) Microbiome and skin diseases. Curr Opin Allergy Clin Immunol 13: 514-20. [Crossref]
- Belkaid Y, Hand T (2014) Role of the microbiota in immunity and inflammation. Cell 157: 121-141.
- Frank DN, Feazel LM, Bessesen MT, Price CS, Janoff EN, et al. (2010) The human nasalmicrobiota and Staphylococcus aureus carriage PLoS ONE 5: e10598. [Crossref]
- Patel RV, Lebwohl M (2011) In the clinic. Psoriasis. Annals of internal medicine. 155 ITC2-1,àíICT2-15.
- Omer H, McDowell A, Alexeyev OA (2017) Understanding the role of propionibacterium acnes in acne vulgaris: The critical importance of skin sampling methodologies. Clin Dermatol 35: 118-129. [Crossref]
- Clavaud C, Jourdain R, Bar-Hen A, Tichit M, Bouchier C, et al. (2013) Dandruff is associated with disequilibrium in the proportion of the major bacterial and fungal populations colonizing the scalp. PLoS One 8: e58203. [Crossref]
- Xu Z, Wang Z, Yuan C, Liu X, Yang F, et al (2016) Dandruff is associated with the conjoined interactions between host and microorganisms. Scientific Reports 6: 24877.
- Wang E, Lee JS-S, Hee TH (2012) Is Propionibacterium acnes associated with hair casts and alopecia? Int J Trichol 4: 93-97. [Crossref]
- Tayyebi Meibodi N, Asadi Kani F, Nahidi Y, Bordbar Azari J, Sadeghian H, et al. (2012) Lichen planopilaris: Histopathological study of vertical sections of scalp biopsies in 44 patients. Iranian Red Crescent Med J 14: 501-502. [Crossref]


