Objective: In the paper, the authors examine the surfaces of the bovine articular cartilage at varying pH solutions by measuring the friction coefficient.
Design: Cartilage pairs with the same sign of charges have been investigated: positively (+/+), negatively (-/-) and neutral (IEP) cartilage surfaces under aqueous buffer solution.
Results: We present evidence that the (cartilage/cartilage) pair charged with the identical positive or negative charge density exhibited the same value of the friction coefficient. Interfacial energy the “bell-shaped curve” of spherical lipid bilayers a model membrane described regarding the electrostatic repulsion of the cartilage surfaces is supporting friction curve interpretation.
Conclusion: We found that charged surfaces had a smaller friction coefficient than neutral (cartilage/cartilage) pair. It was found that the friction was mostly dependent on the charge densities of the cartilage surface and the friction between charged surfaces is lower than that of neutral cartilage surfaces.
charged articular cartilage, bilayer of phospholipids, friction on charged surfaces, interfacial energy bell-shaped curve
The two natural surfaces charged with the same sign and sliding against one another require better understanding [1-4]. Due to this, using the lamellar repulsion model, the strong dependence of the friction from both the cartilage surface charge density and the surface wettability [1,5] is supporting (cartilage/cartilage) friction. The natural cartilage of animal joints has very low friction for the sliding velocity - a few centimeters per second under a load of 18 MPa [6]. The lipid bilayers on the cartilage surface (surface active phospholipid, SAL) of the healthy joint cartilage contain mainly phosphatidylcholine (41%), phosphatidylethanolamine (27%), and sphingomyelin (32%) of total phospholipids [3,7-9]. Liposomes and lamellar phases are composite structures made of phospholipids in synovial fluid and bilayers adsorbed on cartilage surface. The two models for natural cartilage surfaces boundary lubrication at different pH imparted by phospholipids bilayers on cartilage surface are given by Figure 1.
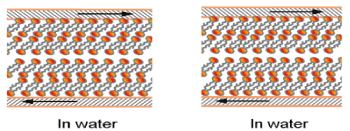
(a) Below IEP, pH 1 to ~4 (b)After IEP, pH ~ 4 to pH 7.4 and above
(a): (Me3N+) (CH2)2 PO4H-R1R2 (b): (Me3N+)(-OH) (CH2)2 PO4--R1R2
At pH~ 4 IEP: (Me3N+) (CH2)2 PO4H-R1R2 D (Me3N+)(-OH) (CH2)2 PO4--R1R2
Figure 1. Model for natural cartilage surfaces imparted by phospholipids bilayers:
(a) Hydrophilic and positively charged (Me3N+-) cartilage surface in pH 1 to 4 below IEP
(b) Hydrophilic and negatively charged (-PO4--) cartilage surface in pH 4 to 7.4 and above and Equilibrium in isoelectric point, at pH~4, IEP
The paper presents the application of the lamellar-repulsion model of the charge density of the interaction between amphoteric surfaces to depict the friction of the sliding surfaces made of cartilage [1-3]. By this model, the low friction may be observed while repulsive articular cartilage surfaces are sliding against one another with the layer of the hydration water, which is located between the interface gap and is operating as a lubricant. Because of that, the friction may be lowered by the bilayers lamellar slippage and repulsion over short distances.
The negatively charged surface of cartilage with synovial fluid at pH 7.4 is supported under load by lamellar slippage of bilayers, thus resulting in low friction. Phospholipids which are in the synovial fluid or form the AC surface bilayers were experimentally proven to play a crucial role as a lubricant [7-9].
In this paper, we studied the friction of the amphoteric soft matter (cartilage/cartilage) pair surfaces vs. pH buffer aqueous solution.. The amphoteric cartilage surface in pH range 1 to 6.5 change charge from positive (-NH3+→ -NH2) to uncharged state, or approaching neutral, IEP, and turns to negative (-PO4H → -PO4-) with pH increasing. The friction coefficient of (cartilage/cartilage) pair was measured in the following cases: both cartilages were positively charged (-NH3+)/(-NH3+), then negatively charged (-PO4-)/(-PO4-) and finally neutral surface and it was shown that charge the cartilage surface is a significant factor of the frictional force. Studying the friction in (cartilage/cartilage) pairs of bovine origin over the pH of buffer solutions is fascinating to observe the electrostatic mechanism of joint lubrication.
The samples of articular cartilage were taken from the knees of an aged-one-to-two-years-old ox. Osteochondral plugs, which diameters were 5 and 10 mm long, were collected from lateral and medial femoral condyles with the use of a circular stainless steel cutter. Then, the articular cartilage discs were cut in such a way to form 3-mm plugs with underlying bone. Then, the samples were stored at 253 K in 0.15 M NaCl (pH = 6.9) and, before testing, they were fully defrosted. Next, the discs of articular cartilage were glued to the disc and attached to the surface made of stainless steel. Finally, friction tests were carried out.
To prepare the buffer solutions 0.2 M sodium hydroxide was added to 100 mL of a solution made of 0.04 M acids: acetic (80% of the solution), phosphoric and boric acid. A sodium hydroxide solution was used at the temperature 22oC to adjust a suitable buffer pH [10]. The electrolyte pH was controlled using a pH-meter in the process of the measurements.
Friction test
The friction coefficient (f) was measured with the usage of the ITeR sliding friction tester pin-on-disc tribotester at room temperature. The tribotester measured the friction between two samples of articular cartilage which were soaked in the buffor solution playing a role of lubricating fluid and exposed to a load with sliding velocities for some time. The speed of sliding discs was very low (1mm/s in 2 and 5 min), and the load was 15 N (1.2 MPa), what is relevant to the natural physiological conditions. Before running the test, the specimen of articular cartilage was put to buffer solution for 60 min. Then, five tests were carried out with the usage of fresh articular cartilage discs for each experiment which is described in [5].
The friction coefficient of (cartilage/cartilage) pair vs. pH
Following Amonton’s law, the frictional force (F) occurring between two sliding solids is proportional to the load (W) and the coefficient of the proportionality, (f), called the friction coefficient, (f) = F/W.
The friction tests were conducted before the IEP using pairs of positively charged cartilage surfaces (-NH3+/-NH3+) (curves no 1 and 2). Tests for curves 3, 4 and 5 were carried out after the IEP using pairs of negatively charged cartilage surfaces (-PO4- /-PO4-), while curve 6 was obtained at pH ~ 4.5 using cartilage surfaces at IEP with no net electrical charges (NH3+ (CH2)n PO4- R1R2) (Figure 2A).
Figure 2 (A) shows less symmetrical “bell-shaped curve” of the friction coefficient of mammal’s cartilage (BC) concerning time and pH. As it is shown in Figure 2 (A) it can be observed that for different cartilages there are observed different values of the friction; curves in part (a) should relate to their amphoteric character and charge density. The presented experimental results show that the friction is highly reliant on the electrostatic interaction between two surfaces of cartilage. The observed low friction of two sliding cartilage sides having the same charge is associated with the electrostatic repulsion of a charged (cartilage/ cartilage) pair.
The changes of AC charge density were caused by the variation of pH of the buffer solution, which produced different charge densities of articular cartilage. The curve was obtained at pH 2.5 and 3.5 before IEP, for a pair charged positively of (-NH3+/-NH3+) and curves at pH 6.0, 7.4 and 9.0 were chosen after IEP), for pairs charged negatively of (-PO4- /-PO4-), while the curve at pH 4.5 (IEP) was obtained for no net charges.
Figure 2 (A) shows the pH profiles of the coefficient of friction, the coefficient increases with the decrease of in charge density of the cartilage approaching IEP, showed highest value of friction. The figure clearly shows that the coefficient of friction decreases with the increase in the negative charge density (-PO4-). Such radical change is prescribed to material with amphoteric properties. With increase of pH friction approached minimum value, and with increase pH friction run unchanged.
The different values of friction for various cartilage pH as shown in Figure 2 (B) should be associated with their charge density. The friction coefficient of the (cartilage/cartilage) tribological pair surfaces with positively charged surface (+/+) at pH 2.5 and 3.5, with negatively charged surface (-/-) at pH 6.0, 7.4, and 9.0 and at isoelectric point, IEP (±/±) at a pH 4.5, it was found that the friction was largely dependent on the charge densities of the cartilage surface.
The interfacial energy bell-shaped curve of the phospholipid membrane vs. pH
The interfacial energy (ϒ) of spherical lipid bilayer formed from phospholipids (PLs) vs. pH “bell-shaped curve”, Figure 2(C) with the isoelectric point, IEP, 4.0 ± 0.2 is based on literature data [1] and [4] (quoted from Table 1).
The isoelectric point, IEP, is at a pH of ~ 4 when a PL molecule carries none net electrical charge H2N (CH2)n PO4H-R1R2 D H3N+ (CH2)n PO4--R1R2. Changes of the interfacial energy correspond to the amino (-NH3+→ -NH2) transition with a low pH, after IEP the phosphate (-PO4H → -PO4-) transition at a higher pH range of 4 to 6.5. The isoelectric point, IEP, with the pH at which a phospholipid molecule carries no net electrical charge
IEP ; H2N (CH2)n PO4H-R1R2 D H3N+(CH2)n PO4--R1R2
The isoelectric point (IEP) and an interfacial energy (IE) (mJ/m2) increases when hydrophilic head diameter of lipid decreases is summarized as follows: phosphatidylserine (IEP = 3.80, IE = 2.93), phosphatidylcholine (4.12, 3.53), phosphatidylethanolamine (4.18, 4.06), and sphingomyelin (4.01, 4.42) (quoted from Table 1 in [4]. Changes in interfacial energy correspond well to the amino group (-NH3+→ -NH2) transition at a low pH of 0.2 to 4, after IEP pH 4.0, and the phosphate group (-PO4H → -PO4-) transition at a higher pH range of 4.0 – 6.5.
Nature equipped cartilage with the synovial fluid in the form of an efficient amphoteric phospholipidic buffer mixture. The actual concentrations of [-PO4-] and [-PO4H] influence the effectiveness (or buffer capacity, β) expressed by the molar quantity of [OH-] added, ΔH+ is molar amount of added (or removed) protons to change of the pH, so ΔpH results in equation, (ΔH+) /ΔpH = β [1].
The relation between the buffer capacity (β) and components ratio is presented in Figure 2(B) curve (cartilage) in the physiological range 7.4 ± 1.0. Gradients of the tangents to the curve belong to the interval (6.5 to 8.5) and is inversely proportional to the buffer capacity (β = Δ (EInt Energy) /ΔpH), Figure 2 (C) and β = Δ (f) /ΔpH), Figure 2(A).
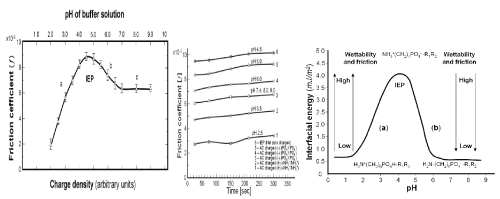
Figure 2. (A) The friction coefficient (f) for (cartilage/cartilage) tribopair vs pH of the universal buffer solutions at different pH values (2.0 - 9.0), under 15N loading and 1 mm/s sliding velocity for 120s, (B) pH profiles of friction coefficient when (cartilage/cartilage) pair slid against each other for 300s positively charged surfaces (+/+) at pH 2.5 and 3.5; and negatively charged surfaces (-/-) at pH 6.0, and 7.4, 8.0 and 9.0; and at isoelectric point (±/±) at pH 4.5, S.D 10 to 15%. (C) The interfacial surface energy (ϒ) of spherical lipid bilayer model „bell-shaped curve” vs. pH of buffer solution. (a) ( pH 2.0 to 4.0 (-NH3+→ -NH2) (b) (pH 4.0 to 6.5 ) (-PO4H → -PO4-)
It was experimentally proved that the charged surface of cartilage resulted from lower friction than that measured at pH ~ 4 (IEP) for no net charges. The low friction characterizes low interlayer (contact angle ~ 0o), lamellar slippage (Figure 3b) of bilayers and short-range repulsion. The interfaces of AC surfaces negatively charged (-PO4-) and contribution of the hyaluronan, proteoglycans (PGs), a glycoprotein termed lubricin and lamellar PLs phases [2,11] support the concept of the lamellar-repulsive mechanism.
Figure (3a) shows transformation of hydrophilic bilayer to hydrophobic monolayer during drying process at room condition. A tree of ‘lamellar-repulsive mechanism’ of joints lubrication shown in Figure (3b). Surface amorphous multilayer (SAL) membrane provides the boundary lamellar-repulsive hydration lubrication. The lamellar-repulsive mechanism is supported by PL lamellar phases and charged macromolecules present in the synovial fluid between two charged cartilage surfaces.
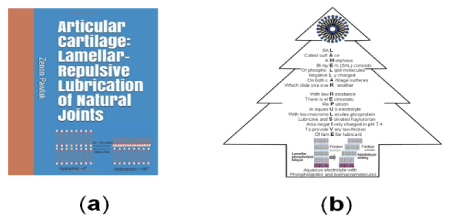
Figure 3. Book cover page ‘Articular Cartilage: Lamellar Repulsive-Lubrication of Natural Joints’ [1], (b) Tree of ‘Lamellar-repulsive mechanism’ of joints lubrication
In this article, we have examined the influence of pH on the friction lipid bilayers on the cartilage surface. The friction between (cartilage/cartilage) pair carrying the same sign of charges with positively charged surface (+/+) at pH 2.5 and 3.5, with negatively charged surface (-/-) at pH 6.0, 7.4, and 9.0 and at isoelectric point, IEP (±/±) at a pH 4.5 has been investigated in buffer aqueous solution. It is demonstrated that the friction was largely dependent on the electrostatic interaction between two charged of the cartilage surfaces. Due to these observations the negatively charged range 7.4 ± 1 covering natural lubrication draw most our attention. At pH 7.4, the negatively charged bilayers of PLs demonstrated very high buffer effectiveness (β) transmitted by a phospholipid, (ΔH+) /ΔpH = β. It suggests that the lamellar-repulsive hydration mechanism can explain lubrication in joints. In the paper, there is shown that the curve of the friction coefficient Figure 2 and interfacial energy curve Figure 2 have an isoelectric point, IEP, and the “bell-shaped curve” or the Gaussian curve. Moreover, slippage of lamellar bilayers and short-range repulsion between the interfaces of the negatively charged cartilage surfaces are the main determinants of the low frictional properties of the joint.
- Pawlak Z (2018) Articular Cartilage: lamellar-repulsive lubrication of natural joints, Kindle direct publishing pp161.
- Williams PF, Powell GL, LaBerge M (1993) Sliding friction analysis of phosphatidylcholine as a boundary lubricant for articular cartilage J Engin Medicine 207: 59-66.
- Hills BA (2000) Boundary lubrication in vivo. Proc Inst Mech Eng H 214: 83-94. [Crossref]
- Petelska AD and Figaszewski ZA (2009) pH effect of the sphingomyelin membrane interfacial tension. J Membrane Biol 230: 11-19.
- Pawlak Z, Gadomski A, Sojka M, Urbaniak W, Beldowski P (2016) The amphoteric effect on friction between the bovine cartilage/cartilage surfaces under slightly sheared hydration lubrication mode. Coll. Surfaces B: Biointerfaces 146: 452-458.
- McCutchen CW (1959) Sponge-hydrostatic and weeping bearings. Nature 184: 1284-1285. [Crossref]
- Ballantine GC, Stachowiak GW (2002) The effects of lipid depletion on osteoarthritic wear. Wear 253: 385-93.
- Kosinska MK, Liebisch G, Lochni G, Wilhelm J, Klein H, et al. (2013) A lipidomic study of phospholipid classes and species in human synovial fluid. Arthritis Rheum 65: 2323-2333.
- Sarma AV, Powell GL, LaBerg M (2001) Phospholipid composition of articular cartilage boundary lubricant, Journal of Orthopedic Research 19: 671-676.
- Britton HTK, Robinson RA (1931) Universal buffer solutions and the dissociation constant of veronal. J Chem Soc 10: 1456-1462.
- Guimarães MV, Terra DL, Lana JFSD, Morais PAG (2019) Impact on the quality of life of patients suffering from osteoarthritis of the knee after intra-articular administration of bone marrow mononuclear cells. Clin Med Invest 4: 2-5.



