HD patients have higher incidence of Frail, Sarcopenia and Locomotive Syndrome than general elderly due to disorders of muscle energy metabolism in uremic state, deterioration of physical activity during HD session and after HD treatment, malnutrition from restriction of protein intake, and various complications. Exercise intervention is effective in improving motor function, but because the subjects are elderly people with significant degenerative diseases of the locomotive organs, caution should be taken in choosing the type and intensity of exercise. Therefore, we examined the efficacy of exercise therapy for elderly HD patients using ergometer during HD session. The total of 95patients divided into two groups: the ergometer group (n=30) and or control group (n=33). HD patients were made to exercise three times a week for 12 months depending on their physical function. After the intervention As a result, the exercise group showed improvement both in physical and mental aspects by quality of life (QOL) evaluation, and improved by physical function. Moreover, percentage of Frail, Locomo degree decreased. Therefore, one-year intervention might improve physical function and QOL of HD patients by eliciting specific whole-body and local effects.
hemodialysis patients, elderly aerobic exercise, QOL
In Japan, the number of patients undergoing hemodialysis (HD) was reported to exceed 320,000 at the end of 2015 and it is further increasing. Among the patients, the average age at the end of 2015 was 67.54 years. It is related with the prolongation of dialysis vintage and the older age at dialysis induction, which increased in the last 10 years when compared to the past 20 years. The percentage of patients aged over 65 years increased to 65.1% in 2015 compared to 50.3% and to 33.0% in 2005 and 1995, respectively. Therefore, essential aging measures are needed in these patients. As such, it is important to prolong a healthy life span allowing people to spend their own daily life without health problems. Frailty, sarcopenia, and locomotive syndrome are well known factors that affect a healthy life span, and single and/or multiple pathological conditions, which induce disabilities of movement, the shortening of elderly care period and quality, and the close of healthy life span. These factors are closely connected with falls. Patients undergoing HD have a higher incidence of frailty, sarcopenia, and locomotive syndrome than general elderly individuals due to disorders of muscle energy metabolism in uremic state, deterioration of physical activity during HD session and after HD treatment, malnutrition from restricted protein intake, and various complications. Even in our study, these unfavorable conditions were frequently observed. It is reported that deterioration of muscle strength, overall endurance, and flexibility in HD patients induced by such complications affect less than half of healthy people. Therefore, prevention of a decline in physical activity is important. During the recent years, exercise therapy has been actively conducted in many HD institutions, since exercises during HD sessions have led to lower dropout rate and improved exercise tolerability. However, the positive outcome of exercise therapy in patients undergoing HD is generally observed in those who are aged between 35 and 54 years. Moreover, such therapy is intended for patients without impaired physical function enabling activities of daily living. There are few reports on exercise therapy for elderly patients undergoing HD who have decreased physical activity levels. We examined the efficacy of exercise therapy in elderly patients undergoing HD using ergometer during HD session via a non-randomized comparative test.
Subjects and measurements
The subjects of this study were 95 patients who were over 65 years of age and underwent outpatient maintenance HD at our clinic. They were divided into two groups: the intervention group, which underwent exercise therapy during HD, and the control group, which did not undergo exercise therapy during HD. After a one-year trial, we evaluated changes in quality of life (QOL) and physical function in the two groups. Based on the “Guidelines on rehabilitation in cardiovascular disease,” contraindications for exercise therapy in HD patients included acute myocardial infarction within two days, angina pectoris without response to medical therapy, arrhythmia with poor control, symptomatic heart failure, symptomatic aortic valve stenosis, acute pulmonary embolism, acute myocarditis, and acute aortic dissection (Figure 1). The subjects were fully informed about the protocol and provided written informed consent to participation in the study. The study protocol was approved by the Institutional Review Board of Tsukuba University of Technology (Approval number: 2015-12-15), including the ethical rules for human experimentation that are stated in the Declaration of Helsinki.
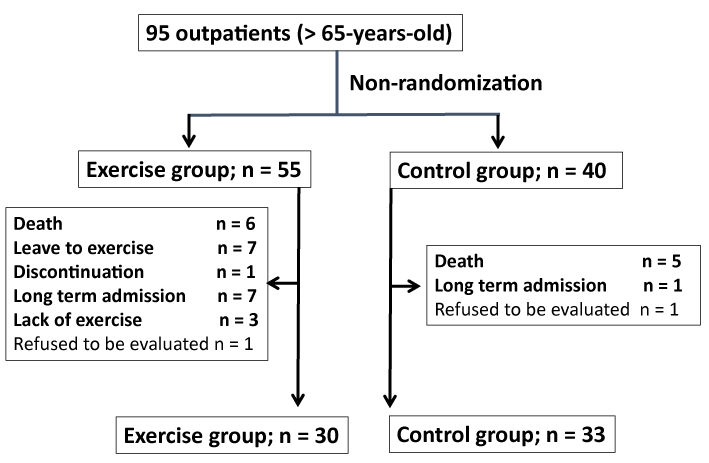
Figure 1. Patients were non-randomly divided into two groups. The number of patients who have continued the exercise therapy for one year is presented.
Exercise therapy methods
In the intervention group, exercise time was at least 30 minutes or more using ergometer during the HD session. Exercise intensity was adjusted to Borg scale score 11-13. The ergometer used two types of fully automatic type Escargo PBE-100 model and variable load type ergometer Ⅱto suit the Borg scale.
The criteria for discontinuing an exercise included resting pulse rate ≤ 40/min or ≥ 120/min, resting systolic blood pressure ≤ 70 mmHg or ≥ 200 mmHg, resting diastolic blood pressure of ≥ 120 mmHg, resting body temperature of ≥ 38℃, resting SpO2 ≤ 90% at room air. Before the exercise, patients who had palpitations, shortness of breath, chest pain, strong pain in the hip joint or the knee joint, lower leg ulcer in the part affecting the pedal, dizziness in the sitting position, cold sweats, nausea, etc. and in cases that fell under contraindications, exercise was not performed. Exclusion criteria for exercise therapy included withdrawal in cases where the patient refused to undergo physical function and mental tests.
Physical performance test
Measurement items were QOL, which was evaluated using SF-36 questionnaires, and physical motor function, which included walking speed, muscular strength, body balance ability, and exercise tolerance. For evaluating walking speed, a 10-meter walking test was performed. Grip strength was measured using digital grip force meter. Leg strength was evaluated using a 30-second standing sitting test. An open eye one leg position test was used to evaluate balance ability. The six-minute walking distance test was used to examine exercise tolerance. Each item was measured before and after one year of exercise intervention, and the measurement was performed before dialysis on the first dialysis session. Evaluation methods were as follows (Tables 1-5): 1) Frailty was examined using the J-CHS standard; 2) Diagnosis of sarcopenia was made on the basis of the Asian working group criteria. Muscle mass was evaluated using an impedance tool MLT-550N (Toray Industries, Inc.). Locomotive syndrome was evaluated using Locomo Degree 2 approved by the Japan Orthopedic Surgery Association.
Table 1. Patient demographics
| |
Exercise group ( n = 30 ) |
Control group ( n = 33) |
P |
Age (mean ± SD), yr |
73.0 ± 5.6 |
73.5±6.1 |
0.755 |
Gender (M/F) |
13/17 |
10/23 |
0.741 |
Dialysis vintage (month) |
143.7 ± 87.0 |
100.3 ± 94.2 |
0.081 |
Induction age to HD (yr) |
61.0 ± 9.8 |
65.1 ± 11.4 |
0.151 |
Diabetes mellitus (n,%) |
6, 20 |
10, 30.3 |
0.357 |
Body Weight (kg) |
51.2 ± 10.1 |
52.1 ± 12.0 |
0.757 |
Nursing Condition |
Care (%) |
23.3% |
9.1% |
0.046 |
Support (%) |
20.0% |
12.1% |
M, male; F, female; HD, hemodialysis. Values are expressed as mean ± standard deviation (SD)
Table 2. Changes in SF 36 (Physical Health)
Exercise group |
Before (± SD) |
After 1 year |
P* |
Physical functioning |
60.4 ± 28.3 |
61.4 ± 29.4 |
n.s. |
Role physical |
48.0 ± 42.0 |
63.0 ±41.5 |
< 0.05 |
Bodily pain |
68.0 ± 25.7 |
69.0 ± 31.3 |
n.s. |
General health |
49.0 ± 20.5 |
47.8 ± 20.4 |
n.s. |
Vitality |
57.8 ± 27.2 |
54.8 ± 28.0 |
n.s. |
Physical health |
56.7 ± 22.9 |
59.2 ± 26.3 |
n.s. |
Control group |
Before (± SD) |
After 1 year |
P* |
Physical functioning |
71.4 ± 24.8 |
71.6 ± 25.4 |
n.s. |
Role physical |
61.2 ± 39.9 |
62.9 ± 38.7 |
n.s. |
Bodily pain |
72.2 ± 22.4 |
69.3 ± 25.6 |
n.s. |
General health |
51.9 ± 15.9 |
49.1 ± 18.4 |
n.s. |
Vitality |
64.1 ± 20.7 |
63.8 ± 22.3 |
n.s. |
Physical health |
64.2 ± 18.6 |
63.3 ± 19.8 |
n.s. |
Values are expressed as mean ± standard deviation (SD)
Table 3. Changes in SF 36 (Mental Health)
Exercise group |
Before (± SD) |
After 1 year |
P* |
General health |
49.0 ± 20.5 |
47.8 ± 20.4 |
n.s. |
Vitality |
57.8 ± 27.2 |
54.8 ± 28.0 |
n.s. |
Social functioning |
73.5 ± 27.6 |
71.0 ± 30.6 |
n.s. |
Emotion-related role |
52.0 ± 45.2 |
73.3 ± 40.8 |
< 0.05 |
Mental health |
72.2 ± 20.2 |
57.5 ± 20.2 |
< 0.05 |
Spiritual health |
60.9 ± 24.8 |
60.9 ± 24.7 |
n.s. |
Control group |
Before (± SD) |
After 1year |
P* |
General health |
51.9 ± 15.9 |
49.1 ± 18.4 |
n.s. |
Vitality |
64.1 ± 20.7 |
63.8 ± 22.3 |
n.s. |
Social functioning |
87.9 ± 18.4 |
82.3 ± 23.7 |
n.s. |
Emotion-related role |
75.9 ± 39.7 |
77.0 ± 36.8 |
n.s. |
Mental health |
73.5 ± 19.7 |
58.6 ± 17.3 |
< 0.05 |
Spiritual health |
70.7 ± 17.2 |
66.2 ± 19.4 |
< 0.05 |
Values are expressed as mean ± standard deviation (SD)
Table 4. Physical function tests.
Exercise group |
Before (± SD) |
After 1 year |
P* |
10-m walk (sec) |
14.2 ± 15.7 |
13.2 ± 15.5 |
< 0.05 |
Hand grip (Kg) |
22.0 ± 6.4 |
20.3 ± 7.8 |
n.s. |
Standing rest test (time) |
10.7 ± 4.3 |
13.3 ± 7.1 |
< 0.01 |
6-min walk (distance, m) |
321 ± 186 |
381 ± 209 |
< 0.01 |
One-foot standing (sec) |
26.5 ± 36.5 |
21.1 ± 31.2 |
n.s. |
Control group |
Before (± SD) |
After 1 year |
P* |
10-m walk (sec) |
9.9 ± 5.9 |
10.0 ± 10.9 |
n.s. |
Hand grip (Kg) |
24.3 ± 8.1 |
23.0 ± 7.5 |
< 0.05 |
Standing rest test (time) |
11.5 ± 4.5 |
12.6 ± 4.5 |
< 0.05 |
6-min walk (distance, m) |
335 ± 196 |
401 ± 191 |
< 0.01 |
One-foot standing (sec) |
22.6 ± 29.1 |
17.2 ± 21.3 |
< 0.05 |
Values are expressed as mean ± standard deviation (SD)
Table 5. Percentage of frailty, locomotive syndrome, and sarcopenia.
| |
Exercise gruop |
Control group |
Before |
After 1 year |
Before |
After 1 year |
Frailty (%) |
32.3 |
25.8 |
25.8 |
22.6 |
Locom. Ⅱ (%) |
77.4 |
67.7 |
61.3 |
67.7 |
Sarcopenia (%) |
45.5 |
63.6 |
27.5 |
40.0 |
Locom. Ⅱ:Locomotive syndrome* stageⅡ
(*Nakamura K, et al, Clin Rev Bone Miner Metab. 2016)
Statistics
All data were analyzed using IBM SPSS Statistics version 24 (IBM Corp, Armonk, NY, USA), which included descriptive and inferential statistics packages. For parametric data, comparisons of pre- and post-intervention values at two time points were analyzed using paired t-test and unpaired t-test. A chi-squared test was used for evaluation of differences between genders and underlying diseases. All results are given as the mean ± standard deviation. The significance level was set to 0.05.
As shown in Figure 1, 95 patients were randomly assigned into the exercise therapy group (n = 55) and the non-exercise therapy group (n = 40). In the intervention group, 30 patients were able to continue the exercise therapy for one year, while 25 patients withdrew from the study due to death, leave to exercise, long-term hospitalization, exercise shortage, and refusal to be evaluated. In the non-exercise group (control), 40 patients were included, while 7 patients withdrew due to previously mentioned reasons. In total, 30 and 33 patients were included in the intervention and control groups, respectively.
The background of patients in the intervention and control groups is represented in Table 1. There was no significant difference in age, gender, dialysis history, age of induction of dialysis therapy, ratio of diabetes, and dry weight.
In the intervention group, the nursing care situation deteriorated more than in the control group.
In the intervention group, changes in the exercise burden and exercise time showed a significant increase both in exercise time and intensity, although the Borg index did not change after one year. Fourteen patients (46.7%) exercised for ≥ 60 minutes, with a maximum time of 120 minutes.
Regarding the QOL, score changes at the beginning and the end of the eight subscales of SF-36 significantly improved in the intervention group in both the physical and mental aspects of the daily function. The mean of mental health score was maintained in the exercise group (before: 60.9 ± 24.8, after: 60.9 ± 24.7), whereas in the control group it was significantly reduced (before: 70.7 ± 17.2; after: 66.2 ± 19.4; P < 0.05). The average physical health score improved in the exercise group from 56.7 ± 22.9 to 59.2 ± 26.3, whereas in the control group it decreased from 64.2 ± 18.6 to 63.3 ± 19.8.
The physical exercise function, which was tested by assessing the 10-meter walking speed, significantly improved in the intervention group from 14.2 ± 15.7 to 13.2 ± 15.5 sec (P < 0.05). Improvement was not observed in the control group (before: 9.9 ± 5.9; after: 10.0 ± 10.9), but in the intervention group, the improvement ration indicated a significant improvement (P < 0.05). Regarding muscle strength, grip strength significantly worsened in the control group (before: 24.3 ±8.1; after: 23.0 ±7.5; P < 0.05). However, in the intervention group it did not change (before: 22.0 ± 6.4; after: 20.3 ± 7.8).
Regarding the 30-second standing sitting test, 2021 Copyright OAT. All rights reservP < 0.05) in both groups (before: 10.7 ± 4.3 and 11.5 ± 4.5 in the intervention and control groups, respectively; after: 13.3 ± 7.1 and 12.6 ± 4.5 in the intervention and control groups, respectively). Regarding the improvement ratio, it is said that the threshold of life independence function is 14.5 times, and in the exercise group, the ratio exceeding this indicates a significant improvement from 11.5% to 42.3%. Regarding the six-minute walking (m) test, there was a significant improvement (P < 0.05) in both groups (before: 321 ± 186 and 335 ± 196 in the intervention and control groups, respectively; after: 381 ± 209 and 401 ± 191 in the intervention and control groups, respectively). Regarding the improvement rate, the intervention group was increased. There was no significant improvement in the open eye one-foot standing (sec) in the intervention group, but it worsened in the control group (before: 22.6 ± 29.1; after: 17.2 ± 21.3; P < 0.05).
In this study, it is compared with non-exercise group as to it was investigated whether exercise therapy using ergometer during dialysis in elderly patients undergoing HD has a significant effect on maintenance of QOL, improvement of physical activity, complication rate of frailty and locomotive syndrome. We found that the exercise group showed improvement in terms of physical and mental aspects based on QOL evaluation. Additionally, physical function evaluation of this group showed improvement in 10-meter walking time, six-minute walking distance, standing sitting rest, etc. Percentage of frailty and locomotive syndrome degree were decreased. On the other hand, there was no difference between the physical aspects, but mental deterioration was observed in the non-exercise group. Physical function tests revealed reduction in grip strength and open eyed one-foot standing time. The percentage of locomotive syndrome degree and sarcopenia increased.
Previous studies have also reported that cardiopulmonary function and endurance, lower limb muscular strength and walking ability were improved by the supine position ergometer during dialysis. However, these studies’ cohorts comprised many young people, and there was no report limited to the elderly. Even in elderly people aged ≥ 65 years, exercise efficacy by ergometer during dialysis was observed as described above. In addition, severe complications were not observed in the intervention group. It can be considered that such exercise can be performed relatively safely even by the elderly.
Moreover, the supine position ergometer movement during dialysis used in this study is aerobic exercise, and it is presumed that physical activity was maintained by training slow muscle fibers. However, unlike resistance training, which trains fast muscle fibers, it did not lead to maintenance of muscle mass and balance ability. In the future, we would like to add resistance training not only for improving physical function but also for prevention of sarcopenia.
We recognize that this study contains several limitations. First, since exercise intervention was conducted in non-randomized exercisers who showed impaired physical function, many patients with high degree of care were included in the intervention group. Second, we did not adjust for confounding factors. Finally, this observational study was performed at a single facility. We would like to conduct multicenter collaborative research in the future as well as accumulate more cases and make a statistical adjustment [1-6].
No potential conflicts of interest were disclosed.
- Statistical survey committee of the Japan Society of Dialysis Medicine: Present status of chronic dialysis therapy in our country (for examination in December, Heisei 20). Available at: http://docs.jsdt.or.jp/overview/ (Accessed: 1 September 2016).
- Fried LP, Tangen CM, Walston J, Newman AB, Hirsch C, et al. (2001) Frailty in older adults: evidence for a phenotype. J Gerontol A Biol Sci Med Sci 56: M146-156. [Crossref]
- Matsushima T (2013) Exercise in dialysis patients. Osaka Dialysis Research Meeting Journal 31: 75-78.
- Mochizuki T, Matsuo Y, Yao R, et al. (2010) Review of interventional exercise therapy for hemodialysis patients. General Rehabilitation 38: 581-588.
- Takashi N, Hitoshi A, Shunichi I, et al. (2014) Guidelines on rehabilitation in cardiovascular disease (2012 revised edition). Available at: http://www.j-circ.or.jp/guideline/pdf/JCS2012_nohara_h.pdf (Accessed: 1 May 2014)
- Storer TW, Casaburi R, Sawelson S, Kopple JD (2005) Endurance exercise training during haemodialysis improves strength, power, fatigability and physical performance in maintenance haemodialysis patients. Nephrol Dial Transplant 20: 1429-1437. [Crossref]

