A 23 days old male baby presented with features of severe shock and respiratory distress. He was born by caesarean section at term to non consanguineous parents. On examination the baby was noted to be tachypnoeic, tachycardic with cold and clammy lower extremities. Distal pulses were barely palpable in both lower limbs.
There was no structural abnormality in echocardiography. But Cardiac catheterization revealed the presence of distal aortic obstruction by a large intraluminal thrombus. An attempt to dissolve the thrombus with urokinase infusion was unsuccessful. The affected vessels were then surgically explored and a large thrombus from the distal abdominal aorta was removed. Following this the child recovered uneventfully with return of lower limb pulsation. A thrombotic profile revealed the underlying diagnosis of congenital Protein C deficiency and a simultaneous thrombophilic prothrombin gene mutation. This is an extremely unusual presentation of neonatal thrombosis treated by successful vascular surgical procedure.
A 23 days old male baby had presented with features of rapidly increasing shock, peripheral circulatory failure and breathing difficulty. He was born at term by lower segment caesarean section. Parents were non consanguineous. The birth weight of the baby was 2.65 kg. On examination, the baby was tachypnoeic with tachycardia and an extremely poor peripheral perfusion particularly in the lower limbs. The weight on admission was 2.95 kg. The respiratory rate was 76/minute while the heart rate was between 160-180/ min. The capillary refill time was prolonged, at around 5 sec with particularly poor the lower limb perfusion. He was put on Non invasive positive pressure ventilation along with high dose of inotropes and broad-spectrum intravenous antibiotics. The clinical examination revealed that his femoral and arteria dorsalis pedis pulses on both sides were barely palpable.
On further investigation there was a structurally normal heart. The USG Doppler study confirmed severely compromised lower limb circulation with possible intraluminal obstruction of the distal vessels supplying the lower limbs. Further imaging with CT angiography revealed a near complete obstruction of abdominal aorta distal to the origin of the renal arteries with a suspected intra-luminal thrombus (Figure 1). Failure of medical thrombolysis by systemic administration of high-dose Urokinase was followed by the decision of surgical exploration of the affected vessels. The affected vessels were explored through intra abdominal excision. The thrombus was removed with subsequent restoration of lower limb circulation.
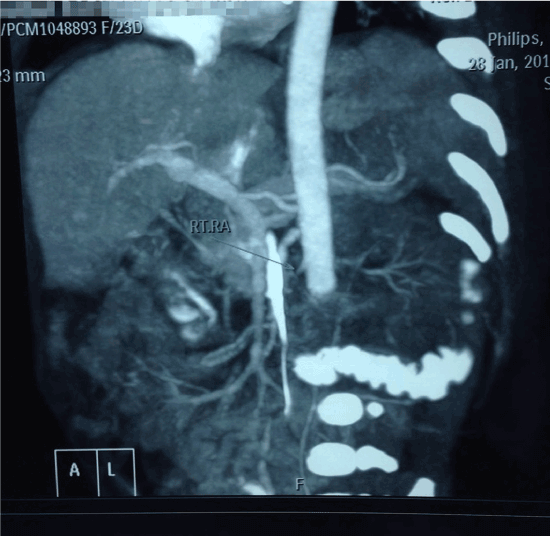
Figure 1. Renal arteries with a suspected intra-luminal thrombus
Further investigations to detect any underlying cause for the thrombosis was undertaken. This had revealed the simultaneous presence of congenital Protein C deficiency and a throbophilic Prthrombin gene mutation. The baby was released after 14 days with an advice for regular follow up.
Protein C is a glycoprotein synthesized in liver and is vitamin K dependent. Activated protein C has got anticoagulant, anti-inflammatory and cytoprotective action. The anticoagulant activity is mediated through the inactivation of coagulation factors Va and VIIIa, which in turn are required for factor X activation and thrombin production. The anti-inflammatory and cytoprotective functions of activated protein C are primarily mediated through the endothelial protein C receptor.
Congenital protein C deficiency is inherited as an autosomal dominant disorder. The gene for protein C is located in chromosome 2q13-14. This may be a heterozygous (0.14% to 0.5% of general population) or homozygous genotype. Homozygous or compound heterozygous presentation has an incidence of 1in 500000 and 1in 750000 live births respectively. The condition itself is broadly classified as Type I(Quantitative) and Type II (Qualitative) deficiency. In the type I variety, though the activated protein C level is markedly reduced, there is a remarkable variability in phenotypical presentation [1]. A simultaneous thrombophilic mutation may enhance the severity in these subjects [2]. Nucleotide change 20210G>A in the prothrombin gene was first described by Poort et al who identified this genotype in 18% of patients with a documented family history of venous thrombosis, but in only 1% of healthy controls [3]. Thrombophilia has been associated with a wide range of clinical manifestations including deep vein thrombosis and maternal or fetal morbidity and mortality. Genetic risk factors for thrombosis such as factor V Leiden, prothrombin 20210G>A, and 677C>T in the MTHFR gene have been reported to contribute to pregnancy complications including intrauterine growth retardation (IUGR), idiopathic pregnancy loss, and late intrauterine fetal demise [4,5].
- Horellou MH, Conard J, Bertina RM, Sarama M (1984) Congenital Protein C deficiency and thrombotic disease in 9 French families. Br Med J 289: 1285-1287.
- Camerlingo M, Finazzi G, Caso L (1991) Inherited protein C deficiency and nonhemorrhagic arterial stroke in young adults. Neurology 41: 1371-1373.
- Poort SR, Rosendaal FR, Reitsma PH, Bertina RM (1996) A common genetic variation in the 3′-untranslated region of the prothrombin gene is associated with elevated plasma prothrombin levels and an increase in venous thrombosis. Blood 88: 3698-3703.
- Martinelli I, Taioli E, Cetin I, Marinoni A, Gerosa S, et al. (2000) Mutations in coagulation factors in women with unexplained late fetal loss. N Engl J Med 343: 1015-1018.
- Alfirevic Z, Roberts D, Martlew V (2002) How strong is the association between maternal thrombophilia and adverse pregnancy outcome? A systematic review. Eur J Obstet Gynecol Reprod Biol 101: 6-14.

