2021 Copyright OAT. All rights reserv
Abstract
Background: Thyroid cancer induces thrombosis. However, the studies on thyroid cancer and risk of venous thrombosis and hypercoagulability are scarce.
Objective: To describe the first case report of cerebral venous thrombosis in patient with thyroid cancer and review the available evidence of venous thrombosis and thyroid cancer using MEDLINE database: Thyroid cancer, papillary; Thyroid cancer, follicular; Thyroid carcinoma, anaplastic; Thyroid cancer, Hurthle cell; Thyroid cancer, medullary; Venous thromboembolism; Pulmonary embolism; Deep vein thrombosis; Venous thrombosis; Cerebral venous thrombosis; Paraneoplastic syndrome; Hypercoagulable state.
Methods and results: We report the case of a 71-year-old female with sudden neurological symptoms. Brain magnetic resonance imaging and magnetic resonance venography confirmed the presence of cerebral venous thrombosis. She was treated with low molecular weight heparin. Twenty-Four hours after admission, patient presented acute dyspnea and pleuritic chest pain. Multidetector computed tomographic pulmonary angiography showed a pulmonary embolism. Thoracoabdominal computed tomography scan showed a right-sided cervical adenopathy and a solitary thyroid mass. Cytological examination of the mass confirmed the presence of papillary thyroid carcinoma.
Conclusion: We report cerebral venous thrombosis and pulmonary embolism in a patient with papillary thyroid carcinoma. Thyroid cancer may predispose a prothrombotic state.
Key words
venous thrombosis, thyroid cancer, cerebral venous thrombosis, pulmonary embolism, paraneoplastic syndrome, hypercoagulable state
Introduction
Malignancy is known to be one of the predisposing factors of venous thrombosis due to its hypercoagulable state. Risk of venous thrombosis has been different among various malignancies. Cancer patients commonly demonstrate abnormal levels of coagulations factors, such as elevated fibrinogen, FV, FVIII, FIX, and FXI, fibrinogen degradation products, and platelet counts [1].
The incidence of venous thrombosis in patients with thyroid cancer is to 3.1 per 1,000 patients [2]. Thyroid cancer induces thrombosis through compression, angioinvasion, or possibly a prothrombotic state. However, the studies on thyroid neoplasms and risk of venous thrombosis and hypercoagulability are scarce. Alterations of coagulation-fibrinolytic system in thyroid cancer have not been extensively studied. Mean platelet volume and its elevation have been suggested as a biomarker for the risk of papillary thyroid cancer in patients with thyroid nodule [3]. In a systematic review, older age (>60 years of age) and proximity to the time of diagnosis have been reported as possible risk factors of venous thrombosis in thyroid cancer [4]. Venous thromboembolism including deep vein thrombosis and pulmonary embolism are the most common thrombotic events [4]. Thyroid cancer may not only predispose a prothrombotic state per se, but also its treatment (e.g., biologically targeted chemotherapies and suppressive levothyroxine treatment) may add to that risk [5].
In this paper, we report a case of cerebral venous thrombosis (CVT) and pulmonary embolism (PE) in patient with thyroid cancer. Review of the literature revealed no previously reported CVT associated with thyroid carcinoma. We also made a systematic literature review of thrombosis in thyroid cancer, with a description of the clinical characteristics, treatment and out-comes of this rare paraneoplastic syndrome.
Case report
A 71-year-old non-diabetic, non-hypertensive woman presented with severe persistent headache of two days duration which was acute in onset and was not relieved by any oral analgesic. Patient described the headache of moderate to severe intensity, progressively worsening, and located in the occipital and frontal areas, associated with nausea and vomiting. There was no history of fever, visual disturbance, seizures, trauma, or head injury. She had two children with no history of spontaneous abortion, complicated puerperium, use of hormonal therapy or constitutional symptoms. Her vital signs and general physical examination were normal. On neurological examination, the cranial nerves were intact and Kernig's and Brudzinski's signs were negative. On ophthalmological examination, both eyes exhibited normal pupillary reflex with no restriction in the movement of extraocular muscles and no papilledema was noted. Initial laboratory tests, including a complete blood count, and creatine and electrolyte measurement, were normal. The D-dimer value was middle-grade elevated. A non-contrast computed tomography (CT) scan of brain was normal. Patient was admitted for severe, headache refractory to conservative therapy and patient received intravenous fluids, oxygen, analgesic agents like nonsteroidal anti-inflammatory drugs and morphine, and ondansetron for nausea. Over the course of hospitalization, patient’s pain improved from severe (score 8-10) to moderate (score 4-10). A magnetic resonance imaging (MRI) scan of the head was obtained. The magnetic resonance venography showed thrombosis of the left and right transverse sinuses (Figure 1). Anticoagulant therapy with low-molecular-weight heparin was started. Twenty-Four hours after admission, patient presented acute dyspnea and right sided pleuritic chest pain. Multidetector computed tomographic pulmonary angiography showed a thrombus located in the right pulmonary artery (Figure 2). A Doppler ultrasound of lower-extremity not revealed deep venous thrombosis. Thrombophilia screening was performed which included homocysteine, Anti-beta 2 glycoprotein, Anti-Cardiolipin, Lupus anticoagulant, antithrombin, protein C, protein S, factor V Leiden, C677T MTHFR and prothrombin G20210A mutations screen were all negative. Levels of factor VIII, IX, XI and XIII were normal. An echocardiogram performed was normal. On hematological investigations, erythropoietin level and the leukocyte alkaline phosphatase score were normal, and Bcr-abl translocation gene and JAK2 V617F mutation were negatives. A cervical and thoracoabdominal CT scan revealed a thyroid mass (Figure 3). An Ultrasound-Guided Fine Needle Aspiration was performed. Cytological examination of the mass confirmed the presence of papillary thyroid carcinoma (Figure 4). Surgery was considered for treatment and total thyroidectomy was performed. The patient progressed favorably, and she was subsequently discharged from the hospital. The anticoagulant treatment was continued for a total treatment time of six months. At 6-month follow up, she was symptom-free and follow-up MRI brain and computed tomographic pulmonary angiography revealed no residual abnormality. For thyroid neoplasia assessment, contrast enhanced CT scan was done 3, 6 and 12 months after the surgery which revealed the disease to be in complete remission. She is doing well even after two years from the diagnosis of cerebral venous thrombosis, pulmonary embolism and thyroid carcinoma.

Figure 1. Coronal view of brain magnetic resonance imaging with venography demonstrating lack of flow in the left and right transverse sinus.
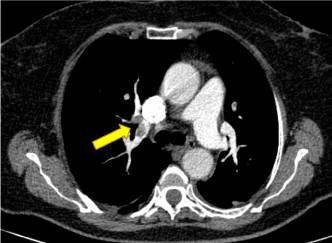
Figure 2. Multidetector computed tomographic pulmonary angiography shows a thrombus located in the right pulmonary artery.
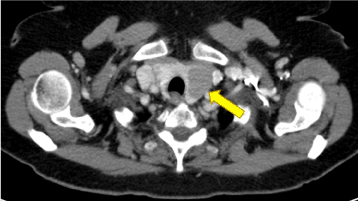
Figure 3. Cervical computed tomographic scan showing a thyroid nodule from the left thyroid lobe that measures approximately 2.9 × 2.8 cm in greatest axial dimensions.
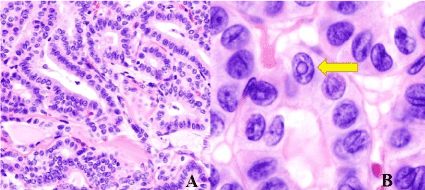
Figure 4. Micrograph showing that the papillae in papillary thyroid carcinoma are composed of cuboidal cells (A). Enlarged histopathological image of papillary carcinoma of the thyroid gland demonstrating diagnostic features (nuclear clearing and overlapping nuclei) (B).
Discussion
Occurrence of CVT and PE found simultaneously is unusual. Clinical risk factors for venous thromboembolism include increasing age, prothrombotic conditions, either genetic or acquired, prolonged immobility, major surgery, trauma, obesity, oral contraceptives, puerperium and pregnancy, nephritic syndrome and infection and malignancy [6]. In the diagnosis of CVT and PE, our patient had none of these risk factors.
Cancer is recognized risk factor for venous thromboembolism. The prevalence of occult cancer in patients with an unprovoked venous thromboembolism seems to be lower (<4%). Venous thrombosis can be the first manifestation of an undiagnosed cancer. Until recently, the literature suggested that up to 10% of such patients would be diagnosed with a cancer in the year after their diagnosis of venous thrombosis [7]. The primary site of cancer is particularly important with highest rates of venous thrombosis observed in patients with brain, pancreas, ovary, lung cancers, and hematologic malignancies including lymphoma and myeloma [8].
Thyroid cancer annual incidence is roughly 1% of all new malignant diseases and has increased over the last decade, mainly due to improved ability to diagnose malignant transformation in small thyroid nodules [9]. Most thyroid cancers manifest as asymptomatic nodules. The true effect of thyroid cancer on the risk of venous thrombosis is still unknown. Thyroid cancer induces thrombosis through compression, angioinvasion, or possibly a prothrombotic state [4]. However, the studies on thyroid cancer and risk of venous thrombosis and hypercoagulability are scarce. In 1879 Kaufmann and Graham reported the ?rst two cases of vascular thrombus associated with thyroid cancer: one in the jugular vein and the other in the thyroid vein [10]. A comprehensive review of the literature shows only few cases of venous thrombosis and thyroid cancer. A hypercoagulable state has been considered in the previous reports of a case of widespread medullary carcinoma with recurrent stroke, nonbacterial endocarditis, elevated D-dimer and normal fibrinogen [11], a case of metastatic medullary thyroid cancer with deep vein thrombosis and pulmonary embolism and mild elevation of FVIII, fibrinogen, and antithrombin III [12], a case of metastatic papillary carcinoma of the thyroid with internal jugular vein thrombosis [13] and a case of cerebral artery thrombosis and thyroid cancer with metastasis to lungs and lymph nodes [14]. We report a rare case of paraneoplastic syndrome: CVT and PE as first manifestation of thyroid carcinoma.
Cerebrovascular events may be the first clinical manifestations in cancer patients or may develop during the course of malignant disease [15]. The risk of CVT has been calculated to be 5-fold increase in patients with cancer. Cerebral venous thrombosis has been reported to be associated with various cancers like squamous cell cervical cancer [16], non-Hodgkin's lymphoma [17], and breast cancer [18]. Patients are especially at risk of developing CVT the first year after diagnosis of malignancy [19]. Review of the literature revealed no previously reported CVT associated with thyroid carcinoma. The risk factors associated with the development of CVT in patients with cancer include direct tumor compression, tumor invasion of cerebral sinuses, the hypercoagulable state associated with cancer, surgery, hospital admissions and immobilization, the presence of an indwelling central catheter, chemotherapy, use of erythropoiesis-stimulating agents and new molecular-targeted therapies such as antiangiogenic agents [20-24]. In our case, local factors such as compression and invasion, were excluded as causes. Cerebral venous thrombosis should always be kept in the differential diagnosis of any simple headaches or neurologic deficits in a patient with cancer even without cerebral metastases and even if the disease is in complete remission. In the case study reported above, headache, nausea and vomiting were the preceding symptoms. Cerebral venous thrombosis and PE is a rare disease. In our case, association of CVT and PE may be explained by a global prothrombotic state for the thyroid neoplasia. PE may be related to a detached thrombus from the cranial venous sinus in our case because neurological symptoms appeared earlier than those of the respiratory symptoms. We think that CVT might have formed before the pulmonary thrombus.
D-dimer has a high negative predictive value in patients with isolated headache for excluding CVT, so D-Dimer levels should be included in any blood work if there is a suspicion of CVT [25]. However, cancer-associated hypercoagulation is difficult to diagnose in the individual because the common coagulation markers including D-dimer lack specificity and sensitivity [26]. The interaction between metastatic disease and blood coagulation have been the object of extensive research leading to the perception that circulating tumor cells may not just accelerate clot formation, but that successful metastasis of several tumor cell types depends on activation of coagulation [27]. D-dimer levels were significantly higher in patients with metastatic disease, indicating that cancer-associated hypercoagulation is more prevalent in this group of patients. As CVT has got a favorable outcome unlike other neurological syndromes, early diagnosis with MRI/MR venography and rapid institution of anticoagulant therapy should be considered [1]. Low-molecular-weight heparin is the recommended class of anticoagulants for the treatment of cancer-associated VTE. Evidence-based treatment guidelines recommend low molecular weight heparin for at least the first 3-6 months for cancer-associated CVT [28,29].
In conclusion, the cause of CVT and PE in this patient was most likely related to the thyroid cancer. Cancer is a strong risk factor for CVT and PE, and venous thrombosis can be the first manifestation of a thyroid cancer.
References
- Mina A, Favaloro EJ, Koutts J (2007) Hemostatic dysfunction associated with endocrine disorders as a major risk factor and cause of human morbidity and mortality: a comprehensive meta-review. Semin Thromb Hemost 33: 798-809. [Crossref]
- Walker AJ, Card TR, West J, Crooks C, Grainge MJ (2013) Incidence of venous thromboembolism in patients with cancer - a cohort study using linked United Kingdom databases. Eur J Cancer 49: 1404-1413. [Crossref]
- Baldane S, Ipekci SH, Sozen M, Kebapcilar L (2015) Mean platelet volume could be a possible biomarker for papillary thyroid carcinomas. Asian Pac J Cancer Prev 16: 2671-2674. [Crossref]
- Ordookhani A, Motazedi A, Burman KD (2017) Thrombosis in Thyroid Cancer. Int J Endocrinol Metab 16: e57897. [Crossref]
- Viola D, Valerio L, Molinaro E, Agate L, Bottici V, et al. (2016) Treatment of advanced thyroid cancer with targeted therapies: ten years of experience. Endocr Relat Cancer 23: R185-R205. [Crossref]
- Ferro JM, Canhão P (2014) Cerebral venous sinus thrombosis: update on diagnosis and management. Curr Cardiol Rep 16: 523. [Crossref]
- Lee AY, Levine MN (2003) Venous thromboembolism and cancer: risks and outcomes. Circulation 107: I17-21. [Crossref]
- Kuderer NM, Lyman GH (2014) Guidelines for treatment and prevention of venous thromboembolism among patients with cancer. Thromb Res 133: S122-S127. [Crossref]
- Burman KD, Wartofsky L (2015) CLINICAL PRACTICE. Thyroid Nodules. N Engl J Med 373: 2347-2356. [Crossref]
- Al-Jarrah Q, Abou-Foul A, Heis H (2014) Intravascular extension of papillary thyroid carcinoma to the internal jugular vein: A case report. Int J Surg Case Rep 5: 551-553. [Crossref]
- Lal G, Brennan TV, Hambleton J, Clark OH (2003) Coagulopathy, marantic endocarditis, and cerebrovascular accidents as paraneoplastic features in medullary thyroid cancer-case report and review of the literature. Thyroid 13: 601-605. [Crossref]
- Tiede DJ, Tefferi A, Kochhar R, Thompson GB, Hay ID (1994) Paraneoplastic cholestasis and hypercoagulability associated with medullary thyroid carcinoma. Resolution with tumor debulking. Cancer 73: 702-705.
- Raveh E1, Cohen M, Shpitzer T, Feinmesser R (1995) Carcinoma of the thyroid: a cause of hypercoagulability? Ear Nose Throat J 74: 110-112. [Crossref]
- Banerjee AK, Chopra JS (1972) Cerebral embolism from a thyroid carcinoma. Report of a case. Arch Neurol 27: 186-187. [Crossref]
- Iqbal N, Sharma A (2013) Cerebral venous thrombosis: a mimic of brain metastases in colorectal cancer associated with a better prognosis. Case Rep Oncol Med 2013: 109412. [Crossref]
- López-Peláez MF, Millán JM, De Vergas J (2000) Fatal cerebral venous sinus thrombosis as major complication of metastatic cervical mass: computed tomography and magnetic resonance findings. J Laryngol Otol 114: 798-801. [Crossref]
- Enevoldson TP, Russell RW (1990) Cerebral venous thrombosis: new causes for an old syndrome? Q J Med 77: 1255-1275. [Crossref]
- Soda T, Edagawa K, Tsuji K, Dehara M, Nakajima Y, et al. (2008) A case of deep cerebral venous thrombosis associated with breast cancer. Clinical Neurology 48: 646-650. [Crossref]
- Silvis SM, Hiltunen S, Lindgren E, Jood K, Zuurbier SM, et al. (2018) Cancer and risk of cerebral venous thrombosis: a case-control study. J Thromb Haemost 16: 90-95. [Crossref]
- Raizer JJ, DeAngelis LM (2000) Cerebral sinus thrombosis diagnosed by MRI and MR venography in cancer patients. Neurology 54: 1222-1226. [Crossref]
- Rogers LR (2004) Cerebrovascular complications in patients with cancer. Semin Neurol 24: 453-460. [Crossref]
- Astudillo L, Lacroix-Triki M, Cousin F, Chevreau C (2007) A rarely diagnosed paraneoplastic syndrome: cerebral venous thrombosis. Rev Med Interne 28: 716-717.
- Stone MJ, Bogen SA (2012) Evidence-based focused review of management of hyperviscosity syndrome. Blood 119: 2205-2208. [Crossref]
- Sung L, Sook-young W, Seonwoo K, Young K, Won K, et al. (2015) Cross-sectional study of patients with diffuse large b-cell lymphoma: Assessing the effect of host status, tumor burden, and inflammatory activity on venous thromboembolism. Cancer Res Treat 2: 1-10. [Crossref]
- Filippidis A, Kapsalaki E, Patramani G, Fountas KN (2009) Cerebral venous sinus thrombosis: review of the demographics, pathophysiology, current diagnosis, and treatment. Neurosurg Focus 27: E3. [Crossref]
- Lee AY (2002) Cancer and thromboembolic disease: pathogenic mechanisms. Cancer Treat Rev 28: 137-140. [Crossref]
- Hejna M, Raderer M, Zielinski CC (1999) Inhibition of metastases by anticoagulants. J Natl Cancer Inst 91: 22-36. [Crossref]
- Nishioka J, Goodin S (2007) Low-molecular-weight heparin in cancer-associated thrombosis: treatment, secondary prevention, and survival. J Oncol Pharm Pract 13: 85-97.
- Coutinho JM (2015) Cerebral venous thrombosis. J Thromb Haemost 13: S238-S244. [Crossref]




