Abstract
Rheumatoid arthritis (RA) is consider as an independent cardiovascular (CV) risk factor and it is associated with increased cardiovascular disease (CVD) mortality and morbidity. Also, RA patients have increased risk of sub-clinical vascular disease compared to their matched controls as shown by high prevalence of carotid artery disease. This subclinical vascular disease was evaluated using the carotid artery intima-media thickness (cIMT) as measured non-invasively using B-mode ultrasound and has been proposed as an early manifestation of atherosclerosis. This literature review aims at analysing the role of cIMT in investigating the subclinical atherosclerosis in RA.
Key words
Rheumatoid arthritis, carotid intima media thickness, ultrasound, cardiovascular disease, atherosclerosis, inflammation
Introduction
Rheumatoid arthritis (RA) patients have significantly increased mortality and morbidity as a result of CVD and cerebro-vascular disease [1]. Laboratory, clinical and epidemiological studies suggest that immune dysregulation and systemic inflammation play an important role in the accelerated atherosclerosis of RA [2,3]. Although cardiovascular (CV) events are major consequences of RA, these complications develop over years, and the time course of epidemiologic and clinical studies has been reduced by using B-mode ultrasound measurement of carotid intima-media thickness (cIMT) and carotid plaque to study early atherosclerotic changes non-invasively [4] (Figure 1). cIMT values measured by ultrasound correlate closely with direct measurement of local and systemic atherosclerotic burden in pathology studies and with clinical CV endpoints [4-6]. Single cIMT measurement is of correspondent to the commonly used risk factors for the prediction of CVD [7]. Therefore, number of studies demonstrated the presence impaired cIMT in RA [8-21], and the change in the cIMT found to be helpful in assessing possible effect of RA drug treatment on CVD risk [22].
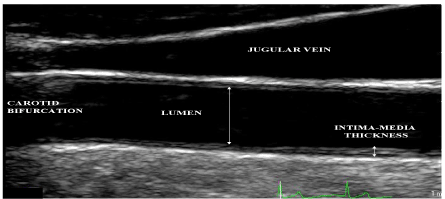
Figure 1. Ultrasound scan of the carotid artery, with labelled anatomic and ultrasonic features.
Ultrasonographic assessment of common carotid atherosclerosis is a feasible, reliable, valid and cost-effective method for both population studies and clinical trials of atherosclerosis progression and regression [23]. Several studies have demonstrated sub-clinical atherosclerosis in established and in early RA patients, either by increased cIMT or presence of carotid atherosclerotic plaque [8,11-13,15,20,21,24].
Even though atherosclerotic disease may remain asymptomatic for decades, its first manifestation can be severe, even fatal. For this reason, early primary prevention of atherosclerosis and efforts to ameliorate its progression has become important goals in medicine. The non-invasive nature of B mode ultrasonography makes it ideal for screening the presence of CVD and for longitudinal studies. cIMT measures of the far wall for both right and left carotid arteries were obtained. Using the far wall has the advantage that it is the clearest target for measurement and the easiest segment to re-image in a reproducible fashion, Figure 1. Dedicated software for the automated measurement of the cIMT are available, eliminating reader bias and variability that significantly contributes to the noise associated with cIMT measurement. It also reduces measurement error related to repeated measurements in long studies and variability between the measurements [25]. Crouse, et al. found that the repeatability of IMT measurements varies among segments and is greatest for far walls and common segments [26].
Furthermore, it is known that vascular structure changes very slowly over time, thus it been proposed that IMT measured at a single time point, is likely to be an excellent reflection of an individual’s past exposure to risk factors.
Carotid intima-media thickness (cIMT) as a proxy measure for atherosclerosis
In the 1980s, several studies concluded that B-mode ultrasound imaging represent a useful approach for measurement of cIMT [4,27-31]. Since then, great number of studies analysed the relationship of cIMT to different diseases including RA. cIMT is a well-validated surrogate measure of the risk of coronary and cerebrovascular disease in an individual. Higher cIMT has been shown to predict future ischemic cardiac and cerebral events among people in asymptomatic populations [32,33]. It been shown that the strength of the association between cIMT and myocardial infarction was at least as strong as the association seen with traditional risk factors [34].
In several prospective follow-up studies cIMT has been used as a determinant of an outcome, or as an outcome variable to study determinants of progression of vessel wall abnormalities [35-37]. cIMT thickness may be used in clinical trials to study the efficacy of non-pharmacological and pharmacological interventions on progression or regression of the atherosclerotic process. This application of the measurement assumes that changes in cIMT parallel changes in cardiovascular risk, through a common mechanism.
Basic principles of ultrasound physics and imaging
When ultrasound pulse waves that propagate through the body come to the boundary between two tissues a certain amount of sound energy is reflected at the interface, while the remainder propagates on into the second tissue. The amount that is reflected depends on the difference in acoustic impedance of the two tissues. Acoustic impedance is defined as the product of material density and the speed of sound. When ultrasound pulse (wave) propagates through the layers of the carotid artery, reflections of echoes may occur at the interface between adventitia and media, media and intima, intima and lumen, if a large enough difference in acoustic impedance exists between the different tissue structures. Whether the reflected echoes will be detected or not depends on the amount of reflected energy in relation to the sensitivity of the ultrasound instrumentation. Better resolution is achievable at higher transducer frequencies, on other hand attenuation; sound energy loss because of scattering and absorption become more pronounced at higher frequencies. Therefore, compromise between resolution and depth of penetration which affect the attenuation degree is required. A good ultrasound imaging of carotid artery can be achieved by a transducer with a wavelength between 5 and 10 MHz depending on how deep the vessel is [38].
Feasibility, reliability, validity and cost effectiveness of cIMT ultrasound
A carotid artery ultrasound is of interest because of it is easy accessibility. It has been suggested that ultrasonographic assessment of cIMT is a feasible, reliable, valid and cost-effective method for both population studies and clinical trials of atherosclerosis progression and regression [23,39]. The reliability of this measurement has been well characterized by several groups. Its reproducibility decreases with increasing values of cIMT, but overall figures indicate inter-observer coefficients of variation of 5-10% [40,41], although some reports are as high as 18% [42,43].In addition, it is inexpensive, can be used in asymptomatic subjects, can be carried out repeatedly, thus reducing the necessary sample size, is safe, and can be used to quantitatively measure structural changes in the wall of superficial large arteries [39].
What is normal cIMT?
The American Heart Association Committee has attempted to define the normal arterial intima. In their view, some arterial segments have particularly thick intima, related to the carotid bifurcation. This bifurcation-related thickening has been termed “eccentric intimal thickening”, “intimal cushion”, or “intimal pad”. In straight, non-branching arteries, such as the distal 1 cm of the common carotid artery (CCA), “normal” intimal thickness is small and comprises a single layer of attenuated endothelial cells over a thin layer of sub-endothelial connective tissue. Both layers give a measure of ≈ 0.02 mm thickness. Because of this small size they cannot be discerned with B mode ultrasound techniques. Therefore, the majority of IMT in a normal CCA is composed of the tunica media [23]. It has been reported that the average thickness of the combined media and intima in a normal CCA varies in middle aged men between 0.7 and 1.2 mm, depending on age [44] (Figure 2).
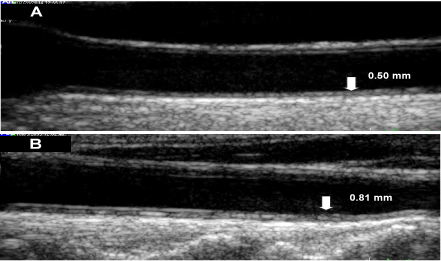
Figure 2. Ultrasound scans of the carotid artery (A) 45-year-old male (B) 70-year- old male; both are RA patients and free of traditional cardiovascular risk factors.
On the other hand, it has been demonstrated that the intimal layer is very thin in straight, non-branching arteries where median intimal cell layer account for 2.5% (0.02-mm) and the median IMT of approximately 97.5% (0.8 mm) [45]. As a result, most of the observed IMT in these regions is from the media rather than intima. In more diseased arteries, the proportions likely approach 20 % intima and 80% media. However, plaque formation is largely a result of intimal thickening. It is therefore possible that IMT fails to accurately reflect risk for CAD [46].
cIMT, age and sex
Different risk factors may vary in its relevance at different stages of atherogenesis. Stensland and colleagues have suggested that CV risk factors that have a stable effect across age could be important in initiating atherosclerosis, whereas risk factors with increasing effect with age, such as hypertension and dyslipidemia, may be more associated with the progression of atherosclerosis [47].
The EVA study [48] reported an atherosclerotic progression rate of approximately 0.01 mm per year in the common cIMT after 4 years in a study population composed of volunteers aged 59-71 years. Comparison of the results of the studies that estimated the rate of progression from cross-sectional observation yields a progression rate for the CCA IMT of 0.0092 mm per year for women and 0.0099 mm per year for men. Hannawi, et al. found that cIMT at early onset rheumatoid arthritis was significantly associated with male gender, and there was a similar trend for cIMT progression. They explained that it might be a result of overwhelming effect of RA-associated inflammation on the usual sex-cIMT progression relationship [21]. Others suggested that the sex differences in terms of cIMT are partly attributable to differences in carotid lumen diameter and may therefore reflect differences in physiology rather than differences in vascular damage [49].
Usual protocol for common cIMT assessment
Carotid intima-media thickness (cIMT) imaging
cIMT ultrasound examination is carried out at the room temperature. Patients coffee consumption and cigarette smoking in the day of the study. cIMT of the CCA, it is recorded while the subject in the supine position and the head slightly extended. the recording session usually take approximately 30 minutes. The most commonly used practice is to record cIMT is three times of each side, with anterior, lateral oblique and posterior oblique views, in order to increase the visualization of the arc of carotid wall. This method permits the determination of the proper mean values for IMT. Gain settings may be altered on individual basis to optimize the images view. When suitable images were obtained (at end-diastole), these to be frozen on the ultrasound monitor for capture.
cIMT measurement
Analyses best carried out on frozen images via ECG triggering (lead II) - top of R-wave- to minimize variability depending on changes in the IMT and lumen diameter occurring during the cardiac cycle [23,50]. The interface of the distal CCA was marked over a length of 10 mm. The beginning of the dilatation of the distal CCA served as a reference point for the start of the measurement. The measures only involved the far wall IMT, according to the leading-edge principle. IMT is defined as the distance from the leading edge of the first echogenic (bright) line to the leading edge of the second line; each tissue interface produces an echo if there is sufficient difference in structure, i.e. in acoustic impedance; this allows detection of small structures whose dimension is beyond the resolution limit (Figure 3). Thus, the anatomical location of a biological structure is always defined by the leading edge, and the thickness of an anatomical entity as the distance between the leading edge of two different echoes [51]. Using the mean of several cIMT measurement rather than a single maximum measure, is generally an accepted method of measurement [25]. Some use the average IMT of six frozen images to be calculated and mean cIMT ((left + right)/2) was taken as a measure of the wall thickness of the distal CCA [25].
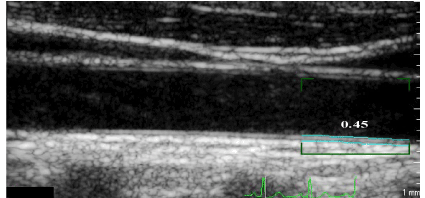
Figure 3. Ultrasound of the common carotid artery, showing the automated IMT measurement.
Measurements should be made at a mineralized (“hard”) plaque, as the echogenic shadow resulting from mineralization usually renders the medial-adventitial interface invisible. As the ultrasonography interfaces are less sharp in the carotid bulb and as the carotid bulb may not be accessible for all subjects, the IMT should not be measured at the carotid bulb even though the IMT is typically greater there than in the CCA. Anatomically, such ultrasound measurement of IMT has been shown to closely relate to the histological measurement of combined IMT in pathological specimens [4,52].
Carotid plaque is defined as a focal widening relative to the adjacent segments, with protrusion into the lumen composed of calcified deposits (hard plaque), non-calcified deposits (soft plaque) or a combination of calcification and non-calcified material (Figure 4). Plaque is considered present if visualized in the full diameter of the vessel; i.e. both the proximal and the distal parts of the plaque are attached to the typical double-lined intima-media structure and the double lines are also visible on the opposite side of the lumen [53].
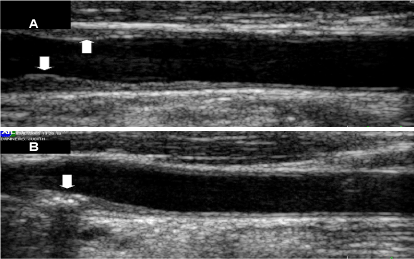
Figure 4. Ultrasound scan of the carotid artery, showing (A) non-calcified, (B) calcified carotid artery plaques.
On the other hand, it is conceivable that certain risk factors might have different associations with different segments of the carotid artery [54]. If site-specific effects dominate IMT progression in the carotid artery, its measurement may not uniformly reflect systemic atherosclerosis presence or progression. Moreover, the estimates based on a segment of 1 cm distal to the carotid bifurcation of the far wall alone might underestimate the true strength of demonstrated associations between cIMT and the atherosclerosis risk including RA or any other CVD risk factors [54]. Additionally, stronger association of inflammation with IMT of the bifurcation and internal carotid artery compared with associations with the IMT of the CCA might be suggested on the bases of the greater turbulence of flow identified at those sites. Data from the asymptomatic carotid artery progression study (ACAPS) indicate that averaging across greater numbers of walls increases the stability of the measure [55].
Limitations of cIMT assessment
cIMT measurement has number of limitations. Firstly, the thickening may not reflect the atherosclerosis but may merely be an adaptive response to changes in shear stress, lumen diameter, tensile stress and pressure [32,56]. Therefore, it has been suggested that cIMT should be considered relative to the CCA diameter. Secondly, compared with other large arteries such as the coronary arteries, atherosclerosis of the CCA tends to develop relatively late in life [32,57]. Thirdly, the reproducibility of cIMT evaluation depends on the performance of the sonographer/reader and also on the technique used for image processing [58]. Fourthly, one of the limitations when comparing the degree of atherosclerosis in peripheral and coronary arteries is that different methods are used. IMT is the main variable studied in carotid artery ultrasonography, whereas with coronary angiography, residual lumen or relative stenosis is measured and not wall thickness [59]. Glagov, et al. suggested that a large increase in wall thickness due to atherosclerosis may be seen both in coronary and in carotid arteries before a decrease in lumen diameter and stenosis development occur. Therefore, it is to be expected that ultrasound and coronary angiography will give different findings during the early stage of atherosclerosis [56], which might indicate that at early stages of atherosclerosis, cIMT may not represents coronary atherosclerosis. Fifthly, although some studies have demonstrated that cIMT corresponds well with pathological findings [60], others show that the distance between the two leading edges in the far wall corresponds best with total wall thickness, including adventitia, and not with the extent of the intima-media complex alone [61]. Sixthly, gray scale ultrasound is limited in its ability to detect progression or regression of carotid atherosclerosis over time in unselected, asymptomatic subjects [62]. Seventhly, disorders of the arterial wall, such as myointimal hyperplasia and/or hypertrophy, due to carotid wall shear stress and/or tensile stress alteration may induce a compensatory and/or pathological increase of cIMT [63], so last but not least, myointimal hyperplasia resulting from iatrogenic (injury) causes such as surgical injury may increase cIMT [64].
Conclusion
Relative to the other easily obtainable and established risk factors, use of cIMT as a screening and classification tool for atherosclerosis in general and in RA patients, would need further assessment in a large population. In addition, studies of these measurement in different populations, geographical locations and ethnic groups may be of importance. Also, possibly having a specific population cIMT measurement as a reference is of vital importance for futures studies, both in clinical uses and in research tools. Multiple measurements and views of the obtained image is important to the accuracy of the obtained value. None the less, cIMT is a valuable non-invasive early manifestation of atherosclerosis. Hence, it is important to pursue, especially as it has been found that a single cIMT measurement is of equivalent importance to the commonly used risk factors for the prediction of coronary heart disease and cerebrovascular disease.
Abbreviations
CCA: Common carotid artery; CHD: Coronary heart disease, cIMT: Carotid intima-media thickness, CRP: C-reactive protein; CV: Cardiovascular; CVD: Cardiovascular disease; ED: Endothelial dysfunction; IMT: Intima-media thickness; RA: Rheumatoid arthritis
Competing interests
The authors declare that they have no competing interests.
Authors’ contribution
HS wrote the manuscript. HH and AI edited the manuscript. All authors read and approved the final manuscript.
References
- Wolfe F, Mitchell DM, Sibley JT, Fries JF, Bloch DA, et al. (1994) The mortality of rheumatoid arthritis. Arthritis Rheum 37: 481-494. [crossref]
- Carroll L, Hannawi S, Marwick T, Thomas R (2006) Rheumatoid arthritis: links with cardiovascular disease and the receptor for advanced glycation end products. Wien Med Wochenschr 156: 42-52. [crossref]
- Van Doornum S, Jennings GL, Wicks IP (2006) Reducing the cardiovascular disease burden in rheumatoid arthritis. Med J Aust 184: 287-290. [crossref]
- Pignoli P, Tremoli E, Poli A, Oreste P, Paoletti R (1986) Intimal plus medial thickness of the arterial wall: a direct measurement with ultrasound imaging. Circulation 74: 1399-1406. [crossref]
- Lorenz MW, Markus HS, Bots ML, Rosvall M, Sitzer M (2007) Prediction of clinical cardiovascular events with carotid intima-media thickness: a systematic review and meta-analysis. Circulation 115: 459-467. [crossref]
- Hannawi S, AlSalmi I, Moller I, Naredo E (2017) Uric acid is independent cardiovascular risk factor, as manifested by increased carotid intima-media thickness in rheumatoid arthritis patients. Clin Rheumatol 36: 1897-1902. [crossref]
- del Sol AI, Moons KG, Hollander M, Hofman A, Koudstaal PJ, et al. (2001) Is carotid intima-media thickness useful in cardiovascular disease risk assessment? The Rotterdam Study. Stroke 32: 1532-1538. [crossref]
- Jonsson SW, Backman C, Johnson O, Karp K, Lundström E, et al. (2001) Increased prevalence of atherosclerosis in patients with medium term rheumatoid arthritis. J Rheumatol 28: 2597-2602. [crossref]
- Bergholm R, Leirisalo-Repo M, Vehkavaara S, Mäkimattila S, Taskinen MR, et al. (2002) Impaired responsiveness to NO in newly diagnosed patients with rheumatoid arthritis. Arterioscler Thromb Vasc Biol 22: 1637-1641. [crossref]
- Hürlimann D, Forster A, Noll G, Enseleit F, Chenevard R, et al. (2002) Anti-tumor necrosis factor-alpha treatment improves endothelial function in patients with rheumatoid arthritis. Circulation 106: 2184-2187. [crossref]
- Kumeda Y, Inaba M, Goto H, Nagata M, Henmi Y, et al. (2002) Increased thickness of the arterial intima-media detected by ultrasonography in patients with rheumatoid arthritis. Arthritis Rheum 46: 1489-1497. [crossref]
- Park YB, Ahn CW, Choi HK, Lee SH, In BH, et al. (2002) Atherosclerosis in rheumatoid arthritis: morphologic evidence obtained by carotid ultrasound. Arthritis Rheum 46: 1714-1719. [crossref]
- Gonzalez-Juanatey C, Llorca J, Testa A, Revuelta J, Garcia-Porrua C, et al. (2003) Increased prevalence of severe subclinical atherosclerotic findings in long-term treated rheumatoid arthritis patients without clinically evident atherosclerotic disease. Medicine (Baltimore) 82: 407-413. [crossref]
- Hänsel S, Lässig G, Pistrosch F, Passauer J (2003) Endothelial dysfunction in young patients with long-term rheumatoid arthritis and low disease activity. Atherosclerosis 170: 177-180. [crossref]
- Cuom G, Di Micco P, Niglio A, La Montagna G, Valentini G (2004) Atherosclerosis and rheumatoid arthritis: relationships between intima-media thickness of the common carotid arteries and disease activity and disability. Reumatismo 56: 242-246.
- Gonzalez-Juanatey C, Testa A, Garcia-Castelo A, Garcia-Porrua C, Llorca J, et al. (2004) Active but transient improvement of endothelial function in rheumatoid arthritis patients undergoing long-term treatment with anti-tumor necrosis factor alpha antibody. Arthritis Rheum 51: 447-450. [crossref]
- Vaudo G, Marchesi S, Gerli R, Allegrucci R, Giordano A, et al. (2004) Endothelial dysfunction in young patients with rheumatoid arthritis and low disease activity. Ann Rheum Dis 63: 331-335. [crossref]
- Cardillo C, Schinzari F, Mores N, Mettimano M, Melina D, et al. (2006) Intravascular tumor necrosis factor alpha blockade reverses endothelial dysfunction in rheumatoid arthritis. Clin Pharmacol Ther 80: 275-281. [crossref]
- Gonzalez-Juanatey C, Llorca J, Sanchez-Andrade A, Garcia-Porrua C, Martin J, et al. (2006) Short-term adalimumab therapy improves endo-thelial function in patients with rheumatoid arthritis refractory to infliximab. Clin Exp Rheumatol 24: 309-312. [crossref]
- Roman MJ, Moeller E, Davis A, Paget SA, Crow MK, et al. (2006) Preclinical carotid atherosclerosis in patients with rheumatoid arthritis. Ann Intern Med 144: 249-256. [crossref]
2021 Copyright OAT. All rights reserv
- Hannawi S, Haluska B, Marwick TH, Thomas R (2007) Atherosclerotic disease is increased in recent onset rheumatoid arthritis: a critical role for inflammation. Arthritis Res Ther 9: R116. [crossref]
- Hannawi S, Marwick TH, Thomas R (2009) Thomas, Inflammation predicts accelerated brachial arterial wall changes in patients with recent-onset rheumatoid arthritis. Arthritis Res Ther 11: R51. [crossref]
- Salonen JT, Salonen R (1993) Ultrasound B-mode imaging in observational studies of atherosclerotic progression. Circulation 87: II56-65. [crossref]
- Alkaabi JK, Ho M, Levison R, Pullar T, Belch JJ (2003) Rheumatoid arthritis and macrovascular disease. Rheumatology (Oxford) 42: 292-297. [crossref]
- Seçil M, Altay C, Gülcü A, Ceçe H, Göktay AY, et al. (2005) Automated measurement of intima-media thickness of carotid arteries in ultrasonography by computer software. Diagn Interv Radiol 11: 105-108. [crossref]
- Crouse JR, Byington RP, Bond MG, Espeland MA, Sprinkle JW, et al. (1992) Pravastatin, lipids, and atherosclerosis in the carotid arteries: design features of a clinical trial with carotid atherosclerosis outcome. Control Clin Trials 13: 495-506. [crossref]
- Crouse JR, Harpold GH, Kahl FR, Toole JF, McKinney WM (1986) Evaluation of a scoring system for extracranial carotid atherosclerosis extent with B-mode ultrasound. Stroke 17: 270-275. [crossref]
- Strandness DE Jr (1986) Ultrasound in the study of atherosclerosis. Ultrasound Med Biol 12: 453-464. [crossref]
- Poli A, Tremoli E, Colombo A, Sirtori M, Pignoli P, et al. (1988) Ultrasonographic measurement of the common carotid artery wall thickness in hypercholesterolemic patients. A new model for the quantitation and follow-up of preclinical atherosclerosis in living human subjects. Atherosclerosis 70: 253-261. [crossref]
- Salonen R, Seppänen K, Rauramaa R, Salonen JT (1988) Prevalence of carotid atherosclerosis and serum cholesterol levels in eastern Finland. Arteriosclerosis 8: 788-792. [crossref]
- Bond MG, Wilmoth SK, Enevold GL, Strickland HL (1989) Detection and monitoring of asymptomatic atherosclerosis in clinical trials. Am J Med 86: 33-36.
- Bots ML, Hoes AW, Koudstaal PJ, Hofman A, Grobbee DE (1997) Common carotid intima-media thickness and risk of stroke and myocardial infarction: the Rotterdam Study. Circulation 96: 1432-1437. [crossref]
- Chambless LE, Folsom AR, Clegg LX, Sharrett AR, Shahar E, et al. (2000) Carotid wall thickness is predictive of incident clinical stroke: the Atherosclerosis Risk in Communities (ARIC) study. Am J Epidemiol 151: 478-487. [crossref]
- O'Leary DH, Polak JF, Kronmal RA, Manolio TA, Burke G, et al. (1999) Carotid-artery intima and media thickness as a risk factor for myocardial infarction and stroke in older adults. Cardiovascular Health Study Collaborative Research Group. N Engl J Med 340: 14-22. [crossref]
- Hofman A, Grobbee DE, de Jong PT, van den Ouweland FA (1991) Determinants of disease and disability in the elderly: the Rotterdam Elderly Study. Eur J Epidemiol 7: 403-422. [crossref]
- Fan AZ, Paul-Labrador M, Merz CN, Iribarren C, Dwyer JH (2006) Smoking status and common carotid artery intima-medial thickness among middle-aged men and women based on ultrasound measurement: a cohort study. BMC Cardiovasc Disord 6: 42. [crossref]
- Johnson HM, Douglas PS, Srinivasan SR, Bond MG, Tang R, et al. (2007) Predictors of carotid intima-media thickness progression in young adults: the Bogalusa Heart Study. Stroke 38: 900-905. [crossref]
- Wendelhag I, Gustavsson T, Suurküla M, Berglund G, Wikstrand J (1991) Ultrasound measurement of wall thickness in the carotid artery: fundamental principles and description of a computerized analysing system. Clin Physiol 11: 565-77. [crossref]
- Salonen JT, Salonen R (1991) Ultrasonographically assessed carotid morphology and the risk of coronary heart disease. Arterioscler Thromb 11: 1245-1249. [crossref]
- Sidhu PS, Desai SR (1997) A simple and reproducible method for assessing intimal-medial thickness of the common carotid artery. Br J Radiol 70: 85-89. [crossref]
- Stensland-Bugge E, Bønaa KH, Joakimsen O (1997) Reproducibility of ultrasonographically determined intima-media thickness is dependent on arterial wall thickness. The Tromso Study. Stroke 28: 1972-1980. [crossref]
- Kanters SD, Algra A, van Leeuwen MS, Banga JD (1997) Reproducibility of in vivo carotid intima-media thickness measurements: a review. Stroke 28: 665-671. [crossref]
- Montauban van Swijndregt AD, De Lange EE, De Groot E, Ackerstaff RG (1999) An in vivo evaluation of the reproducibility of intima-media thickness measurements of the carotid artery segments using B-mode ultrasound. Ultrasound Med Biol 25: 323-330. [crossref]
- Salonen R, Salonen JT (1991) Determinants of carotid intima-media thickness: a population-based ultrasonography study in eastern Finnish men. J Intern Med 229: 225-231. [crossref]
- Spence JD (2002) Ultrasound measurement of carotid plaque as a surrogate outcome for coronary artery disease. Am J Cardiol 89: 10B-15B. [crossref]
- Adams MR, Nakagomi A, Keech A, Robinson J, McCredie R, et al. (1995) Carotid intima-media thickness is only weakly correlated with the extent and severity of coronary artery disease. Circulation 92: 2127-2134. [crossref]
- Stensland-Bugge E, Bønaa KH, Joakimsen O (2001) Age and sex differences in the relationship between inherited and lifestyle risk factors and subclinical carotid atherosclerosis: the Tromso study. Atherosclerosis 154: 437-48. [crossref]
- Zureik M, Touboul PJ, Bonithon-Kopp C, Courbon D, Berr C, et al. (1999) Cross-sectional and 4-year longitudinal associations between brachial pulse pressure and common carotid intima-media thickness in a general population. The EVA study. Stroke 30: 550-555. [crossref]
- Bots ML, Hofman A, Grobbee DE (1997) Increased common carotid intima-media thickness. Adaptive response or a reflection of atherosclerosis? Findings from the Rotterdam Study. Stroke 28: 2442-2447. [crossref]
- Bots ML, Hofman A, de Bruyn AM, de Jong PT, Grobbee DE (1993) Isolated systolic hypertension and vessel wall thickness of the carotid artery. The Rotterdam Elderly Study. Arterioscler Thromb 13: 64-69. [crossref]
- Wikstrand J, Wendelhag I (1994) Methodological considerations of ultrasound investigation of intima-media thickness and lumen diameter. J Intern Med 236: 555-559. [crossref]
- Raninen RO (1998) Ultrasound imaging for arterial wall thickness measurement: an in vitro study with stereomicroscopic correlation. Ultrasound Med Biol 24: 833-839. [crossref]
- Joakimsen O, Bønaa KH, Stensland-Bugge E (1997) Reproducibility of ultrasound assessment of carotid plaque occurrence, thickness, and morphology. The Tromso Study. Stroke 28: 2201-2207. [crossref]
- Espeland MA, Tang R, Terry JG, Davis DH, Mercuri M, et al. (1999) Associations of risk factors with segment-specific intimal-medial thickness of the extracranial carotid artery. Stroke 30: 1047-1055. [crossref]
- Espeland MA, Craven TE, Riley WA, Corson J, Romont A, et al. (1996) Reliability of longitudinal ultrasonographic measurements of carotid intimal-medial thicknesses. Asymptomatic Carotid Artery Progression Study Research Group. Stroke 27: 480-485. [crossref]
- Glagov S, Zarins C, Giddens DP, Ku DN (1988) Hemodynamics and atherosclerosis. Insights and perspectives gained from studies of human arteries. Arch Pathol Lab Med 112: 1018-1031. [crossref]
- Schwartz CJ, Mitchell JR (1962) The morphology, terminology and pathogenesis of arterial plaques. Postgrad Med J 38: 25-34. [crossref]
- Baldassarre D, Werba JP, Tremoli E, Poli A, Pazzucconi F, et al. (1994) Common carotid intima-media thickness measurement. A method to improve accuracy and precision. Stroke 25: 1588-1592. [crossref]
- Hulthe J, Wikstrand J, Emanuelsson H, Wiklund O, de Feyter PJ, et al. (1997) Atherosclerotic changes in the carotid artery bulb as measured by B-mode ultrasound are associated with the extent of coronary atherosclerosis. Stroke 28: 1189-1194. [crossref]
- Wong M, Edelstein J, Wollman J, Bond MG (1993) Ultrasonic-pathological comparison of the human arterial wall. Verification of intima-media thickness. Arterioscler Thromb 13: 482-486. [crossref]
- Gamble G, Beaumont B, Smith H, Zorn J, Sanders G, et al. (1993) B-mode ultrasound images of the carotid artery wall: correlation of ultrasound with histological measurements. Atherosclerosis 102: 163-173. [crossref]
- Bots ML, Mulder PG, Hofman A, van Es GA, Grobbee DE (1994) Reproducibility of carotid vessel wall thickness measurements. The Rotterdam Study. J Clin Epidemiol 47: 921-930. [crossref]
- Zarins CK, Giddens DP, Bharadvaj BK, Sottiurai VS, Mabon RF, et al. (1983) Carotid bifurcation atherosclerosis. Quantitative correlation of plaque localization with flow velocity profiles and wall shear stress. Circ Res 53: 502-514. [crossref]
- Healy DA, Clowes AW, Zierler RE, Nicholls SC, Bergelin RO, et al. (1989) Immediate and long-term results of carotid endarterectomy. Stroke 20: 1138-1142. [crossref]




