Autism spectrum disorder (ASD) is a heterogeneous neurodevelopmental disorder affecting over 1% of the general population. While it is well established that ASD stems from a combination of genetic and environmental factors, there are no defined mechanisms of pathogenesis, rendering curative therapy very difficult to establish. No effective therapy exists for ASD, presenting an urgent clinical need for better control of common core pathologies of ASD regardless of its etiology. Cannabidiol (CBD), a major phytocannabinoid constituent of the cannabis plant, is gaining significant attention in both the media and in medicine for its antiepileptic, anxiolytic, and antipsychotic properties. Recent anecdotal reports, accumulating preclinical and clinical data, and mechanistic insights suggest that CBD provides a viable treatment opportunity for patients with ASD.
pathogenesis, Cannabidiol, Autism spectrum disorder (ASD)
Autism spectrum disorder (ASD), often characterized by impaired social interaction and communication, as well as repetitive and stereotypic behaviors, is one of the most common (∼1/64 at births in US) and fastest-growing neurodevelopmental disabilities. The basic symptoms of ASD are often accompanied by other neurological deficits including, but not limited to, anxiety, mood disorder, sleep disturbance, as well as seizures. These medical conditions associated with ASD interact with each other and result in difficulty of any single symptomatic relief of ASD. Many patients must undergo a series of therapies, medications, and clinical visits in an attempt to alleviate some of the symptoms, which often causes economic and personal stress on the patient and their family. For this reason there is urgent clinical need to better manage multiple neurological pathologies of ASD simultaneously, and in so doing improve the quality of life of ASD individuals and their caregivers.
Cannabis has demonstrated its potential for the therapeutic treatment of a variety of ailments and diseases. The naturally grown plant contains over 80 different biochemical compounds called cannabinoids, many of which share very similar chemical structures and active properties [1]. Cannabinoids are often classified into three sub-groups: Phytocannabinoids, endocannabinoids, and synthetic cannabinoids. Phytocannabinoids are the cannabinoids that are found naturally in the cannabis plant, of which cannabidiol (CBD) is the 2nd most naturally abundant in the plant. CBD has been gaining increasing worldwide attention because of its broad therapeutic potential for providing neuroprotective, anti-inflammatory, antipsychotic, anxiolytic, and anticonvulsant effects in both clinical and preclinical studies [2-8]. Recent anecdotal reports, accumulating animal and human research suggest that CBD provides a viable treatment opportunity for diverse developmental disorders, including ASD. Therefore, CBD holds the potential to simultaneously ameliorate seizures, anxiety, and motor and behavioral deficits that are prevalent in ASD (Figure 1). In an open-label FDA expanded access study, Anderson et al. [9] showed that CBD was not only effective in treating seizures, but also had positive effects on measures of behavior, in a cohort of children with ASD [9].
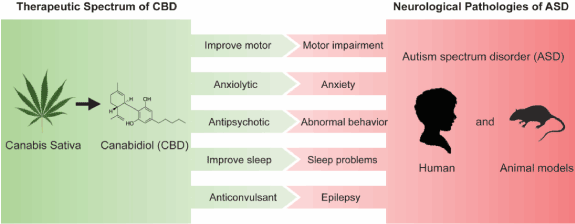
Figure 1. The beneficial spectrum of CBD provides a strategy for treating ASD pathologies.
Mechanism of action of CBD
Though the mechanism underlying the beneficial effects of CBD is still largely unknown, converging lines of evidence suggest dysregulated cannabinoid signaling likely could play a critical role in the pathophysiology of social functioning deficits in ASD [10]. Unlike psychoactive phytocannabinoid delta-9-tetrahydrocannabinol (THC), CBD displays low affinity but unexpectedly high potency as an antagonist of cannabinoid receptor 1/2 (CB1/CB2) in vitro, which likely accounts for its lack of psychotropic activity [11,12]. Contradictorily, CBD can exhibit an indirect effect on the CB1 receptors by inhibiting the degradation of the endocannabinoid anandamide [13]. However, the broad CNS benefits of CBD are likely a result of its complex pharmacological mechanisms. Besides cannabinoid receptors, CBD also interacts with many other, non-endocannabinoid signaling systems, including the equilibrative nucleoside transporter, the orphan G-protein-coupled receptor GPR55, the transient receptor potential of melastatin type 8 channels, the 5-HT1a receptor, the α3 and α1 glycine receptors, and the transient receptor potential of ankyrin type 1 channel [14]. The multiple mechanistic-targets of CBD make it a unique candidate for treating complex symptoms associated with a number of disorders, including ASD [15].
Anticonvulsant effect of CBD
Epilepsy is common (20-30%) in ASD and is more prevalent in individuals with autistic-like behavior resulting from particular genetic predisposition (e.g. Angelman syndrome, Rett syndrome, Dup15q syndrome, etc.). Animals possessing such genetic modifications also exhibit CNS hyperexcitability, enhanced seizure susceptibility and/or spontaneous recurrent seizures. CBD exhibits potent anti-epileptic features in both clinical and preclinical studies [8,16-21]. Notably, CBD significantly reduced the frequency of total seizures of all types in a recent clinical trial for the treatment of drug-resistant seizures in Dravet syndrome [8]. The antiepileptic mechanisms of CBD are still unclear, but may involve multiple targets [14], which renders it an appealing candidate for complex and refractory epilepsy in ASD. Importantly, the anticonvulsive effects of CBD may be useful in suppressing explosive rage and calming severe anxiety. It is hypothesized that unrecognized partial complex seizures, which cause changes in consciousness but not muscular convulsions, are one source of autistic behavior disturbances [22].
Anxiolytic effect of CBD
Of children with ASD, 40% had at least one comorbid diagnosed anxiety disorder [23], which interacts with and exacerbates the other symptoms of ASD. Anxiety negatively impact quality of life of ASD individuals and is associated with higher health care usage and other social costs [24]. Studies using a wide range of animal models of anxiety suggest an anxiolytic-like effect of CBD [2,4]. Interestingly, CBD reduced general anxiety in a bell-shaped dose-response curve, with anxiolytic effects observed at moderate but not higher doses [25-27]. CBD also alleviated stress-induced anxieties in mice. Acute administration of CBD reduces anxiety induced by restraint, re-exposure to a context that previously paired with foot shocks or predator stress [28-31]. Evidence from human studies also strongly supports the potential for CBD as a treatment for anxiety disorders. CBD reduced anxiety in a group of social anxiety disorder patients and was shown to regulate cerebral blood flow in limbic and paralimbic brain areas [4]. CBD also reduced anxiety induced by simulated public speaking in both healthy volunteers and patients with social anxiety disorders [32,33]. Consistent with animal studies, acute administration of CBD induced anxiolytic effects in healthy subjects with moderate (300 mg), but not lower (100 mg) or higher (900 mg) doses [34]. CBD can also attenuate anxiety related sleep problems. Animal studies revealed microinjection of CBD into the central nucleus of the amygdala blocks repeated combination tests-induced rapid eye movement sleep suppression, probably through its anxiolytic effect [35]. A case study reported CBD decreased anxiety and improved the quality and quantity of sleep of a ten-year-old girl diagnosed with post-traumatic stress disorder (PTSD) [36]. The broad anxiolytic effects of CBD provide a unique treatment opportunity for the diverse anxiety phenotypes in ASD.
Wide-spectrum behavioral benefits of CBD
Animal studies suggest that CBD has a broad spectrum of behavioral benefits. CBD administration was found to rescue reserpine-induced motor and cognitive impairments, normalize MK-801-induced social deficits and hyperactivity, and attenuate THC-induced reduction in social interaction in rats [37-40]. Acute CBD treatment also protected against hippocampal cell death and improved cognition in mouse models of ischemia and cerebral malaria [41,42]. CBD can also reduce learned fear by decreasing fear expression, disrupting memory reconsolidation, and enhancing extinction in contextual fear memory paradigm as shown in a study involving mice [43]. These findings suggest CBD holds therapeutic potentials for ASD-related behavioral abnormalities.
Concluding remarks
Individuals with ASD typically face many challenges that worsen their quality of life, create a host of social and behavioral problems, can force their families into a reduced socioeconomic lifestyle, and potentially increase their risk of death. These individuals and their families currently have no available options to alleviate the devastating toll that this experience can have on them. It is for these reasons that there is a great need to find effective treatment options and explore the safety, efficacy and tolerability, of this promising drug, CBD, for these desperate patients. Establishing, in a preclinical and clinical setting, the quality of life and other daily function changes that CBD could be causing in social behavior, cognition, or motor skills is important. Introduction of CBD into a regular treatment regimen could lead to significant reductions in their otherwise untreatable symptoms. The evaluation of changes examined in sophisticated studies while participants are receiving CBD treatment is an important step in therapeutic testing of the drug and in understanding its potential.
Collectively, the pharmacologically broad-spectrum of CBD covers diverse neurological pathologies of ASD, thereby providing a unique and viable therapeutic opportunity to alleviate multiple core ASD symptoms simultaneously. The increasing interest and attention of its beneficial effects in controlling seizures, behavioral and mood abnormalities in ASD call for systematic preclinical and clinical studies to reveal the mechanisms, efficacy, therapeutic spectrum and safety of CBD in ASD.
I thank Christopher L. Anderson (Department of Biomedical Engineering, University of Florida, United States) for critical discussions and reading of the manuscript.
- Mechoulam R, Carlini EA (1978) Toward drugs derived from cannabis. Naturwissenschaften 65: 174-179. [Crossref]
- Blessing EM, Steenkamp MM, Manzanares J, Marmar CR (2015) Cannabidiol as a Potential Treatment for Anxiety Disorders. Neurotherapeutics 12: 825-836. [Crossref]
- Campos AC, Fogaça MV, Sonegoa AB, Guimarães FS (2016) Cannabidiol, neuroprotection and neuropsychiatric disorders. Pharmacol Res 112: 119-127. [Crossref]
- Crippa JA, Derenusson GN, Ferrari TB, Wichert-Ana L, Duran FL, et al. (2011) Neural basis of anxiolytic effects of cannabidiol (CBD) in generalized social anxiety disorder: a preliminary report. J Psychopharmacol 25: 121-130. [Crossref]
- Izzo AA, Borrelli F, Capasso R, Di Marzo V, Mechoulam R (2009) Non-psychotropic plant cannabinoids: new therapeutic opportunities from an ancient herb. Trends Pharmacol Sci 30: 515-527. [Crossref]
- Zhornitsky S, Potvin S (2012) Cannabidiol in humans-the quest for therapeutic targets. Pharmaceuticals 5: 529-552. [Crossref]
- Zuardi AW, Crippa JA, Hallak JEC, Bhattacharyya S, Atakan Z, et al. (2012) A critical review of the antipsychotic effects of cannabidiol: 30 years of a translational investigation. Curr Pharm Des 18: 5131-5140. [Crossref]
- Devinsky O, Cross JH, Laux L, Marsh E, Miller I, et al. (2017) Trial of Cannabidiol for Drug-Resistant Seizures in the Dravet Syndrome. N Engl J Med 376: 2011-2020. [Crossref]
- Anderson CL, Evans VF, DeMarse TB, Febo M, Johnson CR, et al. (2017) Cannabidiol for the Treatment of Drug-Resistant Epilepsy in Children: Current State of Research. J Pediatr Neurol 2017(EFirst). [Crossref]
- Karhson DS, Hardan AY, Parker KJ (2016) Endocannabinoid signaling in social functioning: an RDoC perspective. Transl Psychiatry 6: e905. [Crossref]
- Pertwee RG, The diverse CB1 and CB2 receptor pharmacology of three plant cannabinoids: delta9-tetrahydrocannabinol, cannabidiol and delta9-tetrahydrocannabivarin. Br J Pharmacol 153: 199-215. [Crossref]
- Thomas A, Baillie GL, Phillips AM, Razdan RK, Ross RA, et al. (2007) Cannabidiol displays unexpectedly high potency as an antagonist of CB1 and CB2 receptor agonists in vitro. Br J Pharmacol 150: 613-623. [Crossref]
- Leweke FM, Piomelli D, Pahlisch F, Muhl D, Gerth CW, et al. (2012) Cannabidiol enhances anandamide signaling and alleviates psychotic symptoms of schizophrenia. Transl Psychiatry 2: e94. [Crossref]
- Devinsky O, Cilio MR, Cross H, Fernandez-Ruiz J, French J, et al. (2014) Cannabidiol: pharmacology and potential therapeutic role in epilepsy and other neuropsychiatric disorders. Epilepsia 55: 791-802. [Crossref]
- Ibeas Bih C, Chen T, Nunn AVW, Bazelot M, Dallas M, et al. (2015) Molecular Targets of Cannabidiol in Neurological Disorders. Neurotherapeutics 12: 699-730. [Crossref]
- Devinsky O, Marsh E, Friedman D, Thiele E, Laux L, et al. (2016) Cannabidiol in patients with treatment-resistant epilepsy: an open-label interventional trial. Lancet Neurol 15: 270-278. [Crossref]
- Filloux FM (2015) Cannabinoids for pediatric epilepsy? Up in smoke or real science? Transl Pediatr 4: 271-282. [Crossref]
- Friedman D, Devinsky O (2015) Cannabinoids in the Treatment of Epilepsy. N Engl J Med 373: 1048-1058. [Crossref]
- Jones NA, Glyn SE, Akiyama S, Hill TDM, Hill AJ, et al. (2012) Cannabidiol exerts anti-convulsant effects in animal models of temporal lobe and partial seizures. Seizure 21: 344-352. [Crossref]
- Rosenberg EC, Tsien RW, Whalley BJ, Devinsky O (2015) Cannabinoids and Epilepsy. Neurotherapeutics 12: 747-768. [Crossref]
- Verrotti A, Castagnino M, Maccarrone M, Fezza F (2016) Plant-Derived and Endogenous Cannabinoids in Epilepsy. Clin Drug Investig 36: 331-340. [Crossref]
- Siri k, Lyons T (2012) Cutting-Edge Therapies for Autism2011-2012. Skyhorse Pub. pp: 82-88. [Crossref]
- van Steensel FJ, Bogels SM, Perrin S (2011) Anxiety disorders in children and adolescents with autistic spectrum disorders: a meta-analysis. Clin Child Fam Psychol Rev 14: 302-317. [Crossref]
- Bodden DH, Bögels SM, Nauta MH, De Haan E, Ringrose J, et al. (2008) Child versus family cognitive-behavioral therapy in clinically anxious youth: an efficacy and partial effectiveness study. J Am Acad Child Adolesc Psychiatry 47: 1384-1394. [Crossref]
- Guimaraes FS, Chiaretti TM, Graeff FG, Zuardi AW (1990) Antianxiety effect of cannabidiol in the elevated plus-maze. Psychopharmacology (Berl) 100: 558-559. [Crossref]
- Onaivi ES, Green MR, Martin BR (1990) Pharmacological characterization of cannabinoids in the elevated plus maze. J Pharmacol Exp Ther 253: 1002-1009. [Crossref]
- Zuardi AW, Finkelfarb E , Bueno OF , Musty RE , Karniol IG (1981) Characteristics of the stimulus produced by the mixture of cannabidiol with delta 9-tetrahydrocannabinol. Arch Int Pharmacodyn Ther 249: 137-146. [Crossref]
- Campos AC, Ferreira FR, Guimaraes FS (2012) Cannabidiol blocks long-lasting behavioral consequences of predator threat stress: possible involvement of 5HT1A receptors. J Psychiatr Res 46: 1501-1510. [Crossref]
- Granjeiro ÉM, Gomes FV, Guimar&atil2021 Copyright OAT. All rights reservBM (2011) Effects of intracisternal administration of cannabidiol on the cardiovascular and behavioral responses to acute restraint stress. Pharmacology Biochemistry and Behavior 99: 743-748. [Crossref]
- Lemos JI, Resstel LB, Guimaraes FS (2010) Involvement of the prelimbic prefrontal cortex on cannabidiol-induced attenuation of contextual conditioned fear in rats. Behav Brain Res 207: 105-111. [Crossref]
- Resstel LB, Tavares RF, Lisboa SF, Joca SR, Corrêa FM, et al. (2009) 5-HT1A receptors are involved in the cannabidiol-induced attenuation of behavioural and cardiovascular responses to acute restraint stress in rats. Br J Pharmacol 156: 181-188. [Crossref]
- Bergamaschi MM, Queiroz RHC (2011) Cannabidiol reduces the anxiety induced by simulated public speaking in treatment-naive social phobia patients. Neuropsychopharmacology 36: 1219-1226. [Crossref]
- Zuardi AW, Cosme RA, Graeff FG, Guimarães FS (1993) Effects of ipsapirone and cannabidiol on human experimental anxiety. J Psychopharmacol 7: 82-88. [Crossref]
- Zuardi AW, Rodrigues NP, Silva AL, Bernardo SA, Hallak JEC, et al. (2017) Inverted U-Shaped Dose-Response Curve of the Anxiolytic Effect of Cannabidiol during Public Speaking in Real Life. Front Pharmacol 8: 259. [Crossref]
- Hsiao YT, Yi PL, Li CL, Chang FC (2012) Effect of cannabidiol on sleep disruption induced by the repeated combination tests consisting of open field and elevated plus-maze in rats. Neuropharmacology 62: 373-384. [Crossref]
- Shannon S, Opila-Lehman J (2016) Effectiveness of Cannabidiol Oil for Pediatric Anxiety and Insomnia as Part of Posttraumatic Stress Disorder: A Case Report. Perm J 20: 108-111. [Crossref]
- Almeida V, Levin R, Peres FF, Niigaki ST, Calzavara MB, et al. (2013) Cannabidiol exhibits anxiolytic but not antipsychotic property evaluated in the social interaction test. Prog Neuropsychopharmacol Biol Psychiatry 41: 30-35. [Crossref]
- Gururajan A, Taylor DA, Malone DT (2012) Cannabidiol and clozapine reverse MK-801-induced deficits in social interaction and hyperactivity in Sprague-Dawley rats. J Psychopharmacol 26: 1317-1332. [Crossref]
- Malone DT, Jongejan D, Taylor DA Cannabidiol reverses the reduction in social interaction produced by low dose Delta(9)-tetrahydrocannabinol in rats. Pharmacol Biochem Behav 93: 91-96. [Crossref]
- Peres FF, Levin R, Suiama MA, Diana MC, Gouvêa DA, et al. (2016) Cannabidiol Prevents Motor and Cognitive Impairments Induced by Reserpine in Rats. Front Pharmacol 7: 343. [Crossref]
- Campos AC, Brant F, Miranda AS, Machado FS, Teixeira AL (2015) Cannabidiol increases survival and promotes rescue of cognitive function in a murine model of cerebral malaria. Neuroscience 289: 166-180. [Crossref]
- Schiavon AP, Soares LM, Bonato JM, Milani H, Guimarães FS, et al. (2014) Protective effects of cannabidiol against hippocampal cell death and cognitive impairment induced by bilateral common carotid artery occlusion in mice. Neurotox Res26: 307-316. [Crossref]
- Jurkus R, Day HLL, Guimarães FS, Lee JLC, Bertoglio LJ, et al. (2016) Cannabidiol Regulation of Learned Fear: Implications for Treating Anxiety-Related Disorders. Front Pharmacol 7: 454. [Crossref]

