Extensive resection is the only curative treatment for a giant retroperitoneal sarcoma. We present an “anterior medial approach” for managing this tumor as an aggressive strategy. Case1: A 49-year-old female-patient had a left-sided giant retroperitoneal tumor. Tumor resection along with resection of the left kidney, ureter, adrenal gland, left colon, and left diaphragm was performed through the left anterior medial approach. Pancreatectomy was not required in this case. Case 2: A 66-year-old female-patient had a right-sided giant retroperitoneal tumor. Tumor resection with combined resection of the right kidney, ureter, adrenal gland, right colon, and duodenum was performed through the right anterior medial approach, after neoadjuvant chemotherapy. This patient could also avoid pancreaticoduodenectomy. A negative tumor margin was achieved, and there was no morbidity or mortality in both cases. The “anterior medial approach” adopted for the complete resection of giant retroperitoneal sarcomas was extremely effective for both, left- and right-sided tumors from the perspective of creating a successful operating field, reducing the extent of hemorrhage, and ensuring a negative surgical margin.
anterior medial approach, extensive resection, giant retroperitoneal sarcoma, neoadjuvant chemotherapy, pancreatectomy
Retroperitoneal sarcoma is a rare malignancy, with 0.5 to 1.0 cases per 100,000 people worldwide. Surgical resection is the only curative treatment for giant retroperitoneal sarcomas, with an extensive invasion of the surrounding organs [1,2]. In such cases, it is also essential to perform extensive resections including resection of the surrounding organs to secure a negative resection margin [3]. However, no standard procedure has been established for these surgeries [4]. We adopted an “anterior medial approach” for excising this tumor, and report here, two cases of giant retroperitoneal sarcoma on the right and left side, respectively. These were managed surgically using the anterior medial approach and favorable resection results were achieved. We present the detailed surgical procedures in this report.
Case 1: Left-sided giant retroperitoneal sarcoma
A 49-year-old female-patient had a left-sided retroperitoneal tumor (22 × 19 × 15 cm) that was discovered during an examination conducted to detect the cause of general malaise (Figures 1A-B). Although the diagnosis of liposarcoma was confirmed through a preoperative biopsy, the patient had a history of pancytopenia due to systemic lupus erythematosus (SLE), and preoperative chemotherapy was difficult. Therefore, surgery was planned. Left retroperitoneal tumor resection along with resection of the left kidney, ureter, adrenal gland, left colon, and left diaphragm was performed through a left anterior medial approach (Figure 1C).
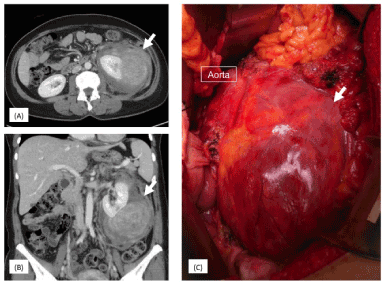
Figure 1. A, B: Contrast-enhanced CT of left-sided giant retroperitoneal sarcoma with an invasion of the left kidney. C: Intraoperative findings of left-sided giant retroperitoneal sarcoma
First, the ligament of Treitz was released, through which the surgeon advanced to the retroperitoneum. The left renal vein was secured, the left margin of the abdominal aorta behind it was identified, and the left renal artery was ligated/disconnected (Figure 2A). The retroperitoneal adipose tissue along the left margin of the abdominal aorta was detached in the downward dorsal direction, and the presence of the iliopsoas muscle was confirmed on the dorsal side. The inferior mesenteric artery was separated at the base, and the ureter and ovarian vein were ligated/disconnected at the height of the left iliac artery and vein. The transverse colon, descending colon, and their mesentery falling within the range of the tumor invasion were separated. In this case, no direct invasion of the tumor into the pancreas and spleen was observed on the cranial side; hence, these organs were preserved. The omental sac was opened, and the transverse mesocolon at the lower margin of the pancreatic body was separated. Combined resection of the Gerota fascia was performed, and the peeling was continued to resect the surrounding pancreatic adipose tissue as much as possible, ultimately reaching the dorsal side of the diaphragm on the cranial end. The major blood vessels supplying the tumor were blocked through the above procedures, and the renal vein was ligated/separated. Finally, the dorsal side of the tumor was peeled off, and the tumor was extracted. In this patient, a portion of the diaphragm revealed tumor invasion. Hence, a combined resection was performed there as well, and the defective part of the diaphragm was closed by direct sutures (Figure 2B).
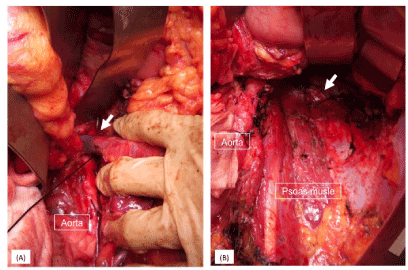
Figure 2. A, B: Intraoperative findings through the left side anterior medial approach. A: The left renal vein was secured (arrow), and the left renal artery was ligated. B: Portion of the diaphragm revealed tumor invasion. Hence, a combined resection was performed, and the defective part of the diaphragm was directly sutured closed (arrow)
The pathological tissue was a dedifferentiated liposarcoma with an invasion of the kidney, and a negative margin was observed. The patient was discharged from the hospital 14 days after the surgery, uneventfully. Additional radiotherapy was administered, and no tumor recurrence has been observed 21 months after the surgery.
Case 2: Right-sided giant retroperitoneal sarcoma
A 66-year-old female-patient had a right-sided retroperitoneal tumor (28 × 18 × 16 cm), which was discovered during an examination conducted for establishing the cause of anorexia. It was accompanied by duodenal stenosis (Figure 3A-B). A diagnosis of leiomyosarcoma was confirmed by preoperative duodenal biopsy. The tumor was massive, and preoperative chemotherapy was administered considering the possibility of a need for pancreaticoduodenectomy during curative resection. Four cycles of doxorubicin and ifosfamide were administered, following which the tumor shrank by approximately 10% (Figure 3C-D). Subsequently, curative resection was performed. Right retroperitoneal tumor resection along with combined resection of the right kidney, ureter, adrenal gland, right colon, and part of the duodenum was performed through the right anterior medial approach.
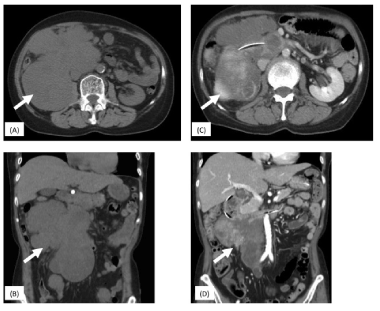
Figure 3. A, B: Plain CT of the right-sided giant retroperitoneal sarcoma (arrow) with right kidney and duodenum invasion before neoadjuvant chemotherapy. Contrast media could not be used because of acute renal failure caused due to ureteral invasion by the tumor (hydronephrosis). C, D: Contrast-enhanced CT after neoadjuvant chemotherapy. About 10% of tumor reduction was achieved (arrow)
First, the base of the ileocecal artery was ligated/separated, and the right margin of the superior mesenteric vein was separated to reach the retroperitoneum (Figure 4A). The pancreatic head, duodenum, and inferior vena cava were identified by this procedure. Partial Kocherization of the duodenum was performed from the cranial side, to identify the inferior vena cava and secure the right renal vein, following which the right renal artery was ligated/separated. The terminal ileum and its mesentery were separated. The ureter and the ovarian vein were identified and ligated/separated at the height of the iliac artery. On the cranial side, the retroperitoneum on the lower surface of the liver was released, the retroperitoneal adipose tissue and right adrenal gland were attached to the resection side, and the diaphragm was identified on the dorsal side. Both, the transverse colon and its mesentery were separated according to the area of tumor invasion. The major blood vessels supplying the tumor were blocked through the above-mentioned procedures, and the renal vein was ligated/separated. The tumor in this patient was thought to have originated from the right ovarian vein, and direct invasion of the duodenum was also observed. Hence, the tumor was detached from the dorsal side so that only these two sites were connected (Figure 4B). Finally, an adequate resection margin was included, the ovarian vein and duodenum were resected, and the tumor was extracted (Figure 5A). The defective part of the duodenum was closed using the Heineke–Mikulicz procedure (Figure 5B).
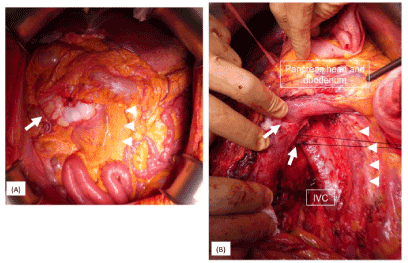
Figure 4. A, B: Intraoperative findings through the right side anterior medial approach. A: Right-sided giant retroperitoneal sarcoma (arrow). The right side on the SMV and SMA is the entry point of this approach (arrowhead). B: The tumor invaded the duodenum and right gonadal vein (arrow)
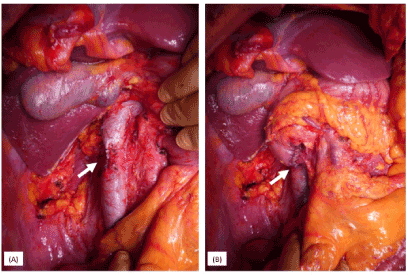
Figure 5. A, B: Intraoperative findings after tumor resection. A: IVC of the gonadal vein orifice was partially resected to secure the surgical margin (arrow). B: The defective part of the duodenum was closed using the Heineke–Mikulicz procedure (arrow)
The pathological tissue was a leiomyosarcoma with an invasion of the duodenum and ovarian vein. A negative margin was observed. The patient was discharged from the hospital 23 days after the surgery. Multiple liver metastases appeared 13 months after the surgery, and chemotherapy was re-initiated.
The “anterior medial approach” for the complete resection of giant retroperitoneal sarcomas was extremely effective for both, left- and right-sided tumors from the perspective of creating a successful operating field, reducing the extent of hemorrhage, and ensuring a negative surgical margin.
Surgical resection is the main curative treatment for retroperitoneal sarcomas, and the surgical procedure is determined based on the size, location, relationship with the surrounding organs, and tissue type of the sarcoma [4]. An extensive resection is recommended in cases where the tissue type is a liposarcoma due to its tendency for extensive invasion, whereas preservation of the surrounding organs might be acceptable in cases of leiomyosarcomas [5,6]. Combined resection of the surrounding organs is often necessary for giant retroperitoneal sarcomas that exceed 20 cm, due to the possibility of tumor invasion and other anatomical concerns. However, there are few reports that describe the details and innovations in the surgical technique for retroperitoneal sarcomas [4], and the “anterior medial approach” described in this study has only been reported by Wang et al, only for left-sided tumors [7].
The “anterior medial approach” does not involve mobilization from the lateral side, instead, it approaches the retroperitoneal muscles from the paravertebral region, using the abdominal aorta and inferior vena cava as indicators from the anterior side. This enables the complete resection of not only the retroperitoneal organs but also the retroperitoneal adipose tissue, in a single surgery [7]. To begin with, resection using the standard lateral mobilization technique is often difficult to perform in cases with massive retroperitoneal sarcomas. Furthermore, almost all giant tumors involve invasion of the surrounding organs or adipose tissue [2,4,6]. Thus, separation from the primary organs adjacent to the tumor, to secure a negative histological margin, is not advisable. Furthermore, the feeding artery of this tumor is often the renal or mesenteric artery, and the drainage is often by the renal or gonadal vein [4]. Therefore, this surgical procedure, which blocks these arterial systems and ultimately separates the draining vein, is thought to be extremely effective since it reduces congestion in the vicinity of the tumor, allows a favorable surgical field, and reduces the extent of hemorrhage. We believe that complete combined resection of at least the kidney, ureter, and colonic mesentery on the side of the tumor invasion is essential for giant retroperitoneal sarcomas exceeding 20 cm in size, regardless of their tissue type. The details of the surgical procedure are discussed below.
The entry point of the “anterior medial approach” is from the left side of the ligament of Treitz for left-sided tumors, and through the ileocecal arteries and veins on the right side of the superior mesenteric arteries and veins for right-sided tumors. Appropriately opening them up allows separation of the mesocolon and rapid entry into the retroperitoneum. Subsequently, the left margin of the abdominal aorta is exposed from the left renal vein on the left side, while the inferior vena cava is exposed from the pancreatic head and duodenum on the right side. Tracing them directly underneath, allows them to be easily separated from the paravertebral column to the retroperitoneal muscle group. The subsequent surgical steps at the caudal end are easily performed. The iliac arteries and veins are identified, and depending on the extent of tumor invasion, the ureter and gonadal veins are separated, the iliopsoas muscle is identified on the dorsal side, and the previously peeled surface is attached.
Meanwhile, on the cranial side, the distal pancreatic body and the spleen are present on the left side, while the pancreatic head and duodenum are on the right side, and the decision regarding their separation line must be considered carefully. Wang et al. performed a distal pancreatectomy on the left side in all 25 patients included in the study and observed invasion of the pancreas and its surrounding adipose tissue in 15 patients (60%), and strongly advocated the necessity of a combined pancreatectomy [7]. The patient with the left-sided tumor in this study showed no direct tumor invasion of the pancreas during the preoperative and perioperative findings. Therefore, the pancreatic parenchyma was preserved, and separation was performed accordingly. Negative margins were observed in the postoperative pathological sample. However, as the tumor cell invasion had extended up to the peri-pancreatic area, postoperative radiation treatment was also administered. Furthermore, as a direct invasion of the duodenum was clearly revealed preoperatively in the patient with the right-sided tumor, a pancreaticoduodenectomy was considered necessary for the curative treatment. We were successful in shrinking the tumor by administering preoperative chemotherapy, and eventually, successfully completed the procedure by performing only a partial duodenectomy without the need for a pancreatectomy. Pancreatectomy combined with retroperitoneal resection of the sarcoma, particularly along with pancreaticoduodenectomy has become relatively safe; however, its morbidity rate is still 34%, and the postoperative mortality and recurrence rates are still relatively high at 3.4% and 66%, respectively [8]. Considering the delicate balance between surgical invasion and cure, we believe that combined pancreatectomy should be avoided as much as possible if there is no direct invasion.
Resections combining the wall of the body cavity for diaphragmatic and iliopsoas muscle invasions are necessary for dorsal and lateral resections. We performed a combined diaphragmatic resection in the patient with a left-sided tumor in this study, but no direct tumor invasion was observed in the postoperative pathological specimen. It is difficult to accurately predict muscle layer invasions preoperatively, and currently, the only way to ensure a negative margin is by conducting extensive resections according to standard surgical procedures for soft tissue sarcomas [9]. In our institutional policy, we prefer to aggressively perform extensive resections keeping in mind diaphragmatic and abdominal wall reconstruction if the major nerves such as the femoral nerve are intact.
Finally, there is no consensus on combined resections for large blood vessel invasions, and high rates of morbidity and tumor recurrence similar to those observed in combined pancreatectomies have been reported [3,8]. Considering the balance between the extent of surgical invasion and the prognosis, we currently perform combined resections of the inferior vena cava and the iliac arteries and veins. Using various reports as reference [10,11], we believe that cases with an invasion of the abdominal aorta, celiac artery, and superior mesenteric artery are not eligible for surgery.
Pre- and postoperative chemotherapy and radiation therapy are expected to act as supplements to overcome the limitations of aggressive and extensive resections; however, their effectiveness is yet to be established [12,13]. As mentioned previously, the patient with the left-sided tumor underwent postoperative radiation therapy, and the patient with the right-sided tumor underwent preoperative chemotherapy which avoided a pancreatectomy and achieved a favorable course. However, there are no guidelines to determine the appropriate combination of these procedures, and it is important to make a comprehensive decision based on the patient’s background, the extent of tumor invasion, experience and/or skill of the operator, and/or the facility as well as the implementation of multidisciplinary treatment, which combines chemotherapy and radiation therapy based on the needs of the individual case.
The “anterior medial approach” adopted for the complete resection of giant retroperitoneal sarcomas was found to be an extremely effective approach for both, left- and right-sided tumors from the perspective of creating a successful operating field, reducing the extent of hemorrhage, and ensuring a negative surgical margin. We plan to conduct further studies once we have accumulated additional cases with a greater number of established this procedure.
None.
Funding: The funding source had no role in the design, practice, or analysis of this study.
Conflicts of interest: Authors declare no Conflict of Interest for this article.
Author contribution: Study conception and design: TM; Acquisition of data: MK, MI; Analysis and interpretation of data and Drafting of the manuscript: TM, TK; All authors read and approved the final manuscript.
- Gatta G, Capocaccia R, Botta L (2017) Burden and centralized treatment in Europe of rare tumor: result of RARECAREnet-a population based study. Lancet Oncol 18: 1022-39.
- Mullinax JE, Zager JS, Gionzalez RJ (2011) Current diagnosis and management of retroperitoneal sarcoma. Cancer Control 18(3): 177-87.
- MacNeill AJ, Gronch A, Miceli R (2018) Postoperative morbidity after radical resection of primary retroperitoneal sarcoma: A report from the Transatlantic RPS working group. Ann Surg 267(5): 959-64. [Crossref]
- Dumitra S, Gronch A (2018) The diagnosis and management of retroperitoneal sarcoma. Oncology 32(9): 464-9. [Crossref]
- Trans-Atlantic RPS Working Group (2015) Management of primary retroperitoneal sarcoma in the adult: a consensus approach from the Trans-Atlantic RPS Working Group. Ann Surg Oncol 22: 256-63. [Crossref]
- Tan MC, Brennan MF, Kuk D (2016) Histology-based classification predicts pattern of recurrence and improve risk stratification in primary retroperitoneal sarcoma. Ann Surg 263: 593-600. [Crossref]
- Wang Z, Wu JH, Lv A (2018) Anterior approach to en bloc resection in left-sided retroperitoneal sarcoma with adjacent organ involvement: A study of 25 patients in a single center. Med Sci Monit 24: 961-9. [Crossref]
- Trans-Atlantic RPS Working Group (2018) Pancreaticoduodenectomy in surgical management of primary retroperitoneal sarcoma. Eur J Surg Oncol 44(6): 810-815. [Crossref]
- Stojadinovic A, Yeh A, Brennan MF (2002) Completely resected recurrent soft tissue sarcoma: primary anatomic site governs outcomes. J Am Coll Surg 194: 436-447. [Crossref]
- Schwarzbach MH, Hormann Y, Hinz U (2006) Clinical results of surgery for retroperitoneal sarcoma with major blood vessels involvement. J Vasc Surg 44(1): 46-55.
- Blair AB, Reames BN, Ahuja N (2018) Resection of retroperitoneal sarcoma en-bloc with inferior vena cava: 20 years outcomes of a single institution. J Surg Oncol 18(1): 127-137.
- Almond LM, Gronchi A, Strauss D (2018) Neoadjuvant and adjuvant strategies in retroperitoneal sarcoma. Eur J Surg Oncol 44: 571-579. [Crossref]
- Cheng H, Miura M, Lalehzari M (2016) Neoadjuvant radiotherapy for retroperitoneal sarcoma: A systematic review. J Surg Oncol 113(6): 628-634. [Crossref]





