Background: Angiomyofibroblastoma is a rare, benign superficial vulvar lesion of the middle-aged female. Most cases in the literature are initially misdiagnosed as Bartholin gland cysts with final diagnosis ascertained via pathology at the time of surgical excision.
Case: A 45-year-old female presented with a large, painless vulvar mass. This mass had formed five years prior to presentation to our clinic, and the patient underwent a procedure to attempt to drain the mass 2-3 years following its initial formation. At her initial visit, a 15 X 10 cm mass was found on the posterior aspect of the right labia minora at the level of the vaginal vestibule. This mass extended inferiorly to the perineum. Needle aspiration was attempted in office but was unsuccessful. The mass was removed under general anesthesia in the operating room en bloc from the vulva without complication and the patient was discharged on postoperative day 0. The pathology of this mass was reported as angiomyofibroblastoma.
Conclusion: Differentiating angiomyofibroblastoma from other potential soft tissue masses of the vulva, especially from the commonly mistaken Bartholin gland cyst or rare aggressive angiomyxoma, is essential for preventing unnecessary procedural workup or treatment miss of a malignant mass. Noting the key distinguishing pathological and clinical features of the tumor and considering risk factors for malignancy will aid in correct diagnosis without exposing the patient to extraneous procedures and potential harm.
benign gynecology, angiomyofibroblastoma, vulvar mass, Bartholin cyst
Angiomyofibroblastoma (AMFB) is a rare benign tumor traditionally described as occurring in the female genital tract; it usually occurs in females between menarche and menopause and was first identified in 1992 [1-3]. It generally arises on the vulva but may also present on the vagina or cervix [3]. Rarely, it has been noted to present in the broad ligament, spermatic cord, testicles, and bladder [4-7]. It is a superficial lesion that involves the subcutaneous tissue and does not often invade the deep pelvis, though exceptions have occurred in the limited existing literature [4,7].
AMFB is known to arise from mesenchymal progenitor cells [1]. Given the rarity of the pathology, there is not a great deal known about genetic composition of the tumor. Attempts have been made to identify genetic aberrations in AMFB that have been described in angiofibromas, myofibromas, and malignant relatives of angiomyofibroblastoma, but with little consistency in results [8]. In accordance with its benign nature, the tumor is generally well-circumscribed and covered by a thin, fibrous pseudocapsule [2,3]. Histologically, there is an alternation between hypocellular and hypercellular areas with tumor cells in the hypocellular areas appearing slender and spindled with elongated nuclei; those in hypercellular areas are plumper, but remain spindled with more vesicular nuclei [2,3]. A loose, myxoid to hyalinized stroma contains a large network of vessels. Tumor cells have a preference for crowding the vessels as is typical in any neoplastic process [2,3]. No nuclear atypia or mitotic figures are usually present, though a higher-mitotic as well as a nuclear atypia variant do exist which confers no greater risk for malignant transformation according to 4-5 year follow up [9]. A lipomatous variant has also been described, which appears to differ only histologically and otherwise confers no difference in prognosis [10].
Immunohistochemical staining patterns vary in the currently available literature, but some commonalities do exist. Almost all cases of AMFB have stained strongly positive for estrogen receptors, desmin, and vimentin [2,3,8]. There is some heterogeneity in the reported staining for progesterone, smooth-muscle actin, and VEGF [8]. All AMFB do not stain for S-100, cytokeratin, and CD34 [2,8,9]. Ki-67 index is low [9].
Few radiological descriptions exist of angiomyofibroblastoma in the literature, yet imaging may aid in accurate preoperative diagnosis. Transvaginal ultrasonography is often favored for pathology of the internal female genital tract whereas either CT or MRI are generally preferred for external lesions. The limited existing reports of transvaginal sonography of AMFB note lesions to be well-circumscribed with hyperechoic areas interspersed with small, cystic hypoechoic structures [11]. Vascularity on ultrasound is not a common feature, and AMFB does not demonstrate prominent Doppler flow [11]. MR findings are nonspecific to other common benign pelvic tumors such as leiomyoma [11]. It is hypointense on T1-weighted images and hyperintense on T2 Gadolinium-weighted images which correspond to the abundant collagen stroma of the tumor [11].
A 45-year-old female presented to our clinic with what had been repeatedly treated as a Bartholin gland cyst, noted to be enlarging rapidly over the past year. She had previously undergone operative drainage of the cyst at an outside institution 2-3 years ago without placement of a Word catheter. She noted that the “fluid” had gradually reaccumulated since then with a more rapid enlargement over the few months prior to her presentation to our institution. In April 2019, she was placed on a short course of antibiotics by her outside OBGYN in efforts to decrease the size of the mass. Beginning in November 2019, the lesion grew in size and became increasingly bothersome, interfering with her ability to sit and walk. Her outside OBGYN re-evaluated her and referred her to our institution for further care. She was evaluated in our clinic in January 2020. A genital examination was performed which revealed a 15 by 10 cm mass at the right lower portion of her labia and vulva without obvious signs of infection. The mass was noted to be soft and mobile. Aspiration of the cystic-appearing mass was attempted with no return of fluid. She returned to the clinic for a preoperative appointment and was posted for a Bartholin gland cyst marsupialization versus a vulvar mass excision. The mass was successfully excised en bloc in February 2020. Intraoperatively, it was noted to be a 12 by 10 cm soft, cystic mass without signs of infection (Figure 1). Pathology returned as an angiomyofibroblastoma measuring 11.2 cm in the largest dimension (Figure 2). Stains were positive for desmin, ER, PR, and actin; the blood vessel components stained positive for CD34 though the tumor cells were not (Figure 3). She followed up in our clinic postoperatively and was recovering appropriately from her surgery.
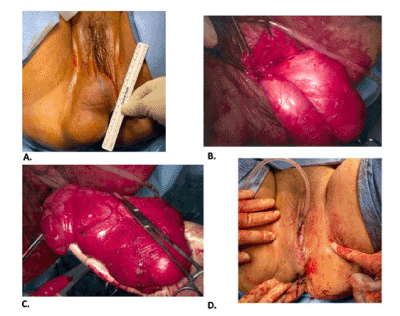
Figure 1. (A) Preoperative measurement of lesion on vulva, approximately 10 cm. (B) Intraoperative dissection of lesion in encapsulated form. (C) Complete excision of lesion. (D) Vulva status-post closure
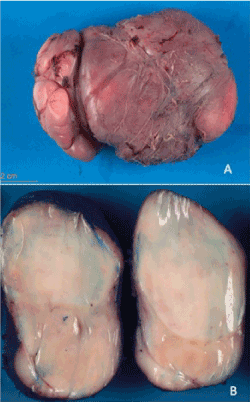
Figure 2. (A) Gross apperance of the surgical specimen. (B) Homogenous, tan-white and rubbery cut surface lacking necrosis or hemorrhage
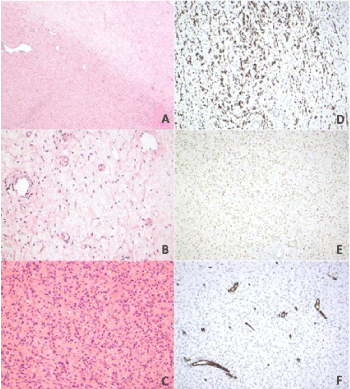
Figure 3. Histologic features of the tumor. (A) Alternating zones of cellularity in low power (4X). (B) Hypocellular area with capillary-like blood vessels. (C) Hypercellular area showed plump, ovoid to spindle shaped cells. (D) Tumor cells showed diffuse strong positivity for desmin stain. (E) Tumor cells show positive for estrogen receptor stain. (F) Tumor cells showed negative for CD34
The appearance of an enlarging soft tissue mass on the vulva presents a variety of possible diagnoses. It is important to include more common benign conditions when formulating differential diagnoses such as a Bartholin gland cyst, epidermal inclusion cyst, vulvar hematoma, and lipoma, to rarer masses, including malignancies, such as Bartholin gland carcinoma, vulvar Crohn’s disease, ectopic breast tissue, vulvar carcinoma subtypes, and dermatologic pathologies occurring on the vulva. Nearly all of the literature reporting cases of AMFB discuss the comparison to its malignant counterpart, aggressive angiomyxoma even though nearly all cases mention that the lesion was initially thought to be pathology of the Bartholin gland. This case, therefore, lends well to a comparison between AMFB and pathology of the Bartholin’s gland since it is exceedingly more common. The differences in treatment between the two conditions makes distinguishing the correct diagnosis all the more critical in order to prevent unnecessary and potentially harmful procedures. In the case of our patient, attempts at treating her AMFB as a Bartholin gland cyst resulted in multiple rounds of unnecessary antibiotics which ultimately contributes to the development of antimicrobial resistance, as well as an unnecessary in-office attempt at aspiration which increases the risk of bleeding and infection of the overlying skin.
When comparing AMFB to a Bartholin gland cyst, there are several similarities. An AMFB is often encountered in the vicinity of the Bartholin gland. Both conditions produce a mobile, generally painless mass that may interfere with walking, sitting and intercourse [12]. Both masses are well circumscribed [13]. Further clinical investigation should offer important clues, however, in differentiating the two lesions. On physical exam, an AMFB will likely lack fluctuance, which is present in a Bartholin gland cyst or abscess. In-office drainage, as in our patient, will be unsuccessful, as will be attempts at marsupialization. The histopathology of a normal Bartholin gland is primarily mucinous glands which drain via ducts lined with cuboidal epithelium [14]. Excised cysts and abscesses will have varied histology based on the etiology of the cyst with reports ranging from mucoceles, to adenomas, to nodular hyperplasia- all markedly different from angiomyofibroblastoma14.
Angiomyofibroblastoma tends toward slow growth; the propensity for malignancy is low even though it possesses similarities to its more aggressive histologic relative, aggressive angiomyxoma1. Almost all existing case reports present a comparison of AMFB to its malignant counterpart, aggressive angiomyxoma, and delineate key differences between the two. This is obviously an important distinction and there is at least one case report of an aggressive angiomyxoma being misdiagnosed as angiomyofibroblastoma due to similarities in histopathology [15]. For this reason, it may be reasonable to consider tailoring follow up to the pathology of the case, the risk factors of the patient for a malignant condition, and the extent of excision possible during removal of the mass (i.e. if excision was completed en bloc with surrounding tissue versus residual tumor left in situ). This may be achieved by encouraging yearly well woman visits to ensure there is no recurrence of symptoms. In the case of our patient, she was followed up two weeks postoperatively, and we anticipate she will return to our clinic in one year for her well woman exam.
Occam’s razor is a critically important principle when practicing medicine. Often the most common diagnosis is the most likely answer. Our case highlights, however, that good clinical intuition must be coupled with pretest probability in order to arrive correctly at proper identification and treatment of disease.
The authors have no financial disclosures or conflicts of interest.
- Fletcher CD, Tsang WY, Fisher C, Lee KC, Chan JK (1992) Angiomyofibroblastoma of the vulva. A benign neoplasm distinct from aggressive angiomyxoma. Am J Surg Pathol 16: 373-382.
- Jian W, Weiqi, S., Xiaoyu, T., Daren, S., Xiongzeng, Z., Renyuan, Z. (2000) Clinicopathologic analysis of angiomyofibroblastoma of the female genital tract. Chin Med J 113: 1036-1039. [Crossref]
- Dodd LG (2014) Atlas of soft tissue and bone pathology: with histologic, cytologic, and radiologic correlations. New York, New York: Demos Medical.
- Huang HC, Chen YR, Tsai HD, Cheng YM, Hsiao YH (2017) Angiomyofibroblastoma of the broad ligament: a case report. Int J Gynecol Pathol 36: 471-475. [Crossref]
- Tzanakis NE, Giannopoulos GA, Efstathiou SP, Rallis GE, Nikiteas NI (2010) Angiomyofibroblastoma of the spermatic cord: a case report. J Med Case Reports 4: 79. [Crossref]
- Msakni I, Ghachem D, Bani MA, Gargouri F, Mansouri N, et al. (2017) Paratesticular angiomyofibroblastoma-like tumor: unusual case of a solidocystic form. Case Reports Med.
- Deka PM, Bagawade JA, Deka P, Baruah R, Shah N (2017) A rare case of intravesical angiomyofibroblastoma. Urology 106: e15-e18. [Crossref]
- Horiguchi H, Matsui-Horiguchi M, Fujiwara M, Kaketa M, Kawano M, et al. (2003) Angiomyofibroblastoma of the vulva: report of a case with immunohistochemical and molecular analysis. Int J Gynecol Pathol 22: 277-284. [Crossref]
- Takeshima Y, Shinkoh Y, Inai K (1998) Angiomyofibroblastoma of the vulva: A mitotically active variant? Pathology Int 48: 292-296. [Crossref]
- Upreti S, Morine A, Ng D, Bigby SM (2015) Lipomatous variant of angiomyofibroblastoma: a case report and review of the literature. J Cutan Pathol 42: 222-226. [Crossref]
- Eckhardt S, Rolston R, Palmer S, Ozel B (2018) Vaginal angiomyofibroblastoma: a case report and review of diagnostic imaging. Case Rep Obstet Gynecol 2018: 7397121. [Crossref]
- Calvert H, Kapurubandara S, Nikam Y, Sharma R, Achan A (2018) A vaginal angiomyofibroblastoma as a rare cause of a prolapsing vaginal mass: a case report and review of the literature. Case Reports in Obstet Gynecol 8579026-8579026.
- Seo JW, Lee KA, Yoon NR, Lee JW, Kim BG, et al. (2013) Angiomyofibroblastoma of the vulva. Obstet Gynecol Sci 56: 349-351. [Crossref]
- Lee M, Dalpiaz A, Schwamb R, Miao Y, Waltzer W, et al. (2015) Clinical pathology of Bartholin’s glands: a review of the literature. Curr Urol 8: 22-25. [Crossref]
- Wang YF, Qian HL, Jin HM (2016) Local recurrent vaginal aggressive angiomyxoma misdiagnosed as cellular angiomyofibroblastoma: A case report. Exp Ther Med 11: 1893-1895. [Crossref]



