Although rolling Nagoya mice carrying a mutation in the a1 subunit of the Cav2.1 channel demonstrate severe ataxia, heterozygous rolling Nagoya (rol/+) mice seem no apparent behavioral abnormalities. However, in our previous study, aged rol/+ mice show deficits with regard to spatial short-term memory in the object location test. These results indicate that onset of phenotypes in rol/+ mice is age dependence. However, it is not yet clear whether nociceptive transmission deficits accompany accelerated aging in rol/+ mice. To assess whether rol/+ mice exhibit an age-related abnormality of nociceptive transmission, we examined a battery of tests using the von Frey test for mechanically induced response, the hot plate test for thermally induced response, and the formalin paw test for chemically induced response. In the von Frey test and the hot plate test, although 2-month-old rol/+ and +/+ mice had similar responses, 22-month-old rol/+ mice showed nociceptive transmission deficits. In the formalin paw test, 2-month-old rol/+ and +/+ mice had similar responses, while 22-month-old rol/+ mice exhibited attenuated Phase 2 response, but normal Phase 1 response. These findings indicate that the rol/+ mice could be useful to delineate the involvement of age-related nociceptive mechanisms.
aging, Cav2.1, nociceptive behavior, Rolling Nagoya mice
Neuronal voltage-dependent Ca2+ channels (VDCCs) including Cav2.1 (P/Q-type), Cav2.2 (N-type), and Cav2.3 (R-type) channels mediate a number of neuronal functions including neuronal excitation, neurite outgrowth, synaptogenesis, neurotransmitter release, neuronal survival, differentiation, plasticity, and regulation of gene expression [1-3]. The VDCC is a molecular complex comprised of several subunits: a1, a2-d, b, and g [1]. The a1 subunit is essential for channel functioning and determines fundamental channel properties [1]. The a1 subunit of the Cav2.1 channel (Cav2.1a1) broadly expresses in the presynaptic terminals [1]. Thus, alterations in the Ca2+ current through Cav2.1a1 would induce neuronal and circuit dysfunction.
Rolling Nagoya (rol/rol) mice carrying a mutation in the Cav2.1a1 demonstrate severe ataxia [4,5]. It has been reported that Ca2+ influx through Cav2.1 decreases with aging [6]. In a previous study, we examined age-related cognitive alterations [7]. Although we found no difference between 2-month-old heterozygous rolling Nagoya (rol/+) and wild-type (+/+) mice, 22-month-old rol/+ mice showed decreased spatial learning compared with age-matched +/+ mice.
The aging brain shows age-associated abnormalities in peptidergic, cholinergic, monoaminergic, and amino acid neurotransmitter release [8-10], suggesting that changes in these domains might contribute to deficits in neuronal functions. The nervous systems express conspicuous signs of aging in the form of memory dementia, which commonly affect the elderly human population. Elderly people, especially those over eighty years of age, are the fastest growing segment of the population throughout the world, and this trend has important implications for the study of pain. It is important to understand the relationship between aging and pain, since it has been reported that the pain threshold is significantly increased in aged human subjects [11]. Several studies have examined age differences in nociceptive behaviors in the rat, and the results showed that older animals are less responsive than younger animals [12,13]. There are two different types of nociception: phasic and tonic nociception. Phasic nociception refers to a brief transient pain elicited by a brief noxious stimulus, and standard tests include the von Frey and hind paw pressure tests for mechanically induced response and the tail flick and hot plate tests for thermally induced response. Tonic nociception refers to continuous pain elicited by inflammation, and the standard test is the formalin paw test for chemically induced response. It was reported that 24-month-old rats were less sensitive than 3-month-old rats in terms of pain responsiveness in the hind paw pressure test, tail flick test, hot plate test [12], and formalin paw test [13]. The expression level of Cav2.1a1 in synapses decreases with aging [14]. Given that the precise regulation of Ca2+ signaling is important for functions of neurons and circuits, alterations in Ca2+ current through Cav2.1 in aged rol/+ mice would affect nociceptive behaviors. However, it is not yet clear whether nociceptive transmission deficits accompany accelerated aging in rol/+ mice.
In this study, we examined age-related nociceptive responses of rol/+ mice by using the von Frey test for mechanically induced response, the hot plate test for thermally induced response, and the formalin paw test for chemically induced response.
Mice
All animal procedures were approved by the Animal Experiments Committee of Shanghai Jiao Tong University and RIKEN. The Rolling Nagoya strain, with a mixed C57BL/6J and SIII genetic background, was provided by the RIKEN BRC with the support of the National BioResource Project of the Ministry of Education, Culture, Sports, Science, and Technology in Japan. Rolling Nagoya was backcrossed with C57BL/6J mice for 12 generations. Male rol/+ and +/+ F1 progeny were derived from a cross between rol/+ mice and genotyped by PCR using tail DNA [7]. The mice were given free access to water and food pellets (CRF-1, Oriental Yeast Co., Ltd., Tokyo, Japan) and were kept under a 12/12-h light/dark cycle (lights on from 08:00 to 20:00) at 23 ± 1°C and 55 ± 5% humidity. We used 2-month-old (young) and 22-month-old (aged) rol/+ and +/+ mice and all tests were performed during the light phase on male mice only.
Histologic examination
Sections of the dorsal root ganglion (DRG), lumbar level of the spinal cord, and thalamus from 2-month-old (rol/+, +/+: n=6, 5) and 22-month-old (rol/+, +/+: n=5, 5) were fixed in 4% buffered formalin and embedded in paraffin. Paraffin sections 4 m thick were stained with hematoxylin and eosin (HE).
Assessment of nociceptive behavior
Experiments including the von Frey test, the hot plate test, and the formalin paw test were performed according to our previous report [15]. We used separate groups of mice for each of three behavioral tests. The von Frey test and the hot plate test were conducted between 10:00 AM and 4:00 PM by a well-trained experimenter who was blinded as to the mouse strains. The formalin paw test was conducted between 10:00 AM and 12:00 PM by a well-trained experimenter who was blinded as to the mouse strains, because it has been reported that there is a marked decrease in tonic pain response in the afternoon [16].
In the von Frey test, 2-month-old (rol/+, +/+: n=12, 12) and 22-month-old ((rol/+, +/+: n=8, 8) were placed individually in plastic cages (11 x 18 x 15 cm) with a wire mesh bottom. After an acclimation period of at least 60 min, von Frey hairs (Touch Test Sensory Evaluator, North Coast Medical, San Jose, CA, USA) of different bending forces (0.02, 0.04, 0.07, 0.16, 0.40, 0.60, 1.00, 1.40, 2.00, 4.00 g) were pressed perpendicularly against the plantar skin until they slightly buckled. Enough pressure to just bend the hair was used and maintained for 6 sec on the left hind paw. If the mouse withdrew its foot, the next lowest force hair was tested. If the mouse did not withdrew its foot, the next highest force hair was tested. Once the mouse responded, a total of six stimulations of the same intensity was applied to the left hind paw at intervals of 6 sec. The minimum force (g) at which mice exposed six times to the same stimulus showed at least four instances of paw-lifting was recorded.
In the hot plate test, 2-month-old (rol/+, +/+: n=12, 10) and 22-month-old (rol/+, +/+: n=8, 8) were placed on a hot plate (Hot plate Analgesia Meter, OTS40, Medizintechnik, Burgdorf, Germany) heated to 50 °C. The cut-off time was 30 sec. The latency until mice showed the first sign of discomfort (forepaw or hind paw licking/biting) was recorded.
In the formalin paw test, 20 ml of 3% formalin solution was injected subcutaneously under the dorsal surface of the left hind paw of 2-month-old (rol/+, +/+: n=10, 10) and 22-month-old (rol/+, +/+: n=8, 8). The animals' responses, such as licking or biting the left hind paw, were recorded between 0 and 15 min (phase 1) and 15 and 60 min (phase 2) after the injection.
Statistical analyses
Data are presented as the means ± SEM. Statistical analysis was conducted by using Excel Statistics 2006 (SSRI, Tokyo, Japan). Data were analyzed using Dunnett’s test or by analysis of variance (ANOVA) followed by Bonferroni’s correction for multiple comparisons between groups, if appropriate The results were considered significant if the probability of error was 5% or less.
There was no apparent morphological abnormality of the DRG, lumbar spinal dorsal horn and laternal ventroposterior nucleus of 2- and 22-month-old rol/+ mice on HE staining (data not shown).
The nociceptive responses to mechanical stimuli were measured using the von Frey test. No significant difference was observed between 2-month-old rol/+ (0.7 ± 0.08 g) and +/+ (0.6 ± 0.09 g) mice (F(1,22)=0.025, P>0.05). On the other hand, there was significant difference between 22-month-old rol/+ (1.6 ± 0.11 g) and +/+ (1.0 ± 0.17 g) mice (F(1,14)=5.232, P<0.05).
Thresholds for noxious heat stimuli were measured using the hot plate test. Although no significant difference was observed between 2-month-old rol/+ (14.3 ± 0.94 s) and +/+ (13.9 ± 0.67 s) mice (F(1,22)=0.108, P>0.05) at 50°C, thermal nociceptive threshold was significant difference between 22-month-old rol/+ (29.7 ± 1.81 s) mice and +/+ (22.1 ± 1.73 s) mice of the same age (F(1,14)=5.561, P<0.05).
Nociceptive responses to chemical stimuli were measured using the formalin paw test in the 2-month-old (Figure 1A) and 22-month-old (Figure 1B) mice. Saline injection in the formalin test did not induce a pain response in any of the mice (data not shown). Subcutaneous injection of formalin produced a characteristic biphasic licking/biting response consisting of an initial, rapidly decaying acute phase (phase 1, 0–15 min after injection) and a long-lived tonic phase (phase 2, 15–60 min after injection). In phase 1, injected formalin directly stimulated the peripheral nociceptors and elicited pain responses, while in phase 2, inflammation induced by formalin injection elicited the response. There was no significant difference in licking/biting time in phase 1 between 2-month-old rol/+ and +/+ mice (F(1,18)=0.017, P>0.05) or between 22-month-old rol/+ and +/+ mice (F(1,18)=0.016, P>0.05). On the other hand, although the total licking/biting times of phase 2 responses in 2-month-old rol/+ and +/+ mice were similar (F(1,18)=0.014, P>0.05), the total licking/biting time of phase 2 responses in 22-month-old rol/+ mice was significantly decreased compared with that of age-matched +/+ mice (F(1,14)=8.657, P<0.01).
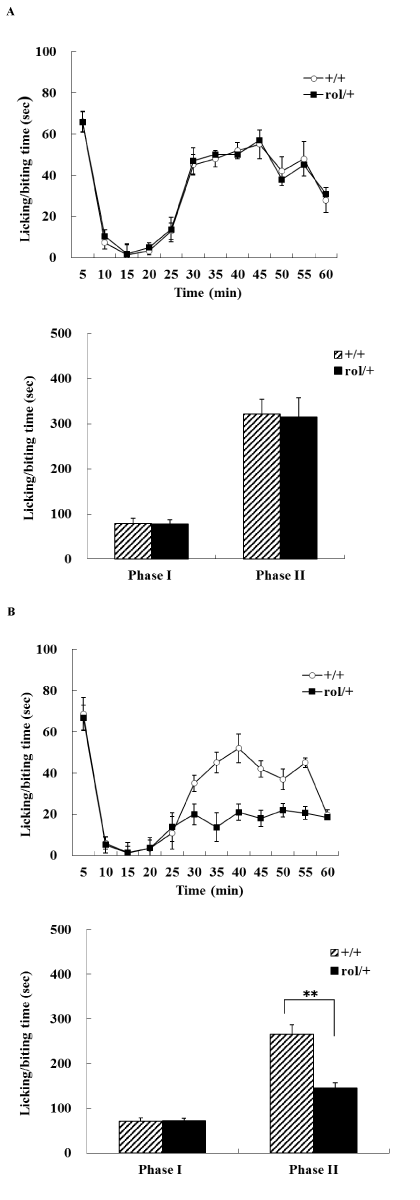
Figure 1.Chemically evoked nociceptive responses. Time–effect curve of nociceptive responses (licking/biting) of rol/+ and +/+ mice at the age of 2 month (A, upper) and 22 months (B, upper) in the formalin test. Licking/biting time in every 5 min period was plotted. Histograms show the licking/biting time during phase 1 and phase 2 of rol/+ and +/+ mice at the age of 2 month (A, lower) and 22 months (B, lower). Asterisks indicate a significant difference from age-matched +/+ mice (**, P<0.01).
In this study, nociceptive responses were examined using the von Frey test for mechanically induced response, the hot plate test for thermally induced response, and the formalin paw test for chemically induced response in rol/+ mice of different ages.
The pain signals produced by the von Frey test enter DRG where the primary afferents are located, leading to excitation of the lumbar levels of the spinal cord [17]. The pain signals produced by the hot plate test and formalin paw test stimulate the DRG and then this information is conveyed to the lumbar levels of the spinal cord and supraspinal levels (e.g., thalamus) [17]. Therefore, we examined the structure of the DRG, lumbar spinal dorsal horn and laternal ventroposterior nucleus of 2- and 22-month-old rol/+ mice. There was no apparent morphological abnormality on HE staining. It is therefore unlikely that morphological changes during development led to impairment of nociceptive response.
In the von Frey test, significant difference was observed between 22-month-old rol/+ and +/+ mice. In the hot plate test, significant difference was also observed between 22-month-old rol/+ and +/+ mice. Nociceptive information from mechanical stimuli is modulated by a simple spinal reflex and by descending pathways [17,18]. Thermally induced pain behavior is known to be associated with both spinal and supraspinal involvement in nociception [17, 19]. The rol/+ mice showed age-related nociceptive transmission deficits in the von Frey test and the hot plate test. These results indicate that alterations in Ca2+ current through Cav2.1 in aged rol/+ mice would affect inputs via primary afferents from mechanical and thermal stimulus, or spinal and supraspinal reflexes.
In the formalin paw test, rol/+ mice at the age of 22 months exhibited attenuated phase 2 response, but normal phase 1 response. It has been reported that Cav2.1 channel plays a role in the regulation of glutamate release [20,21]. Gene knockdown with intrathecal small interfering RNA of N-methyl-D-aspartate (NMDA) receptor NR2B subunit inhibits phase 2 of formalin-induced nociception [22]. These results indicate that aged rol/+ mice would affect glutamate release leading to nociceptive nociceptive transmission deficits.
The decrease in pain response could be related to motor deficits rather than pain response. However, because phase 1 of formalin paw test were normal in rol/+ mice at 22 months of age, this could be considered as evidence of intact motor response in the nociceptive behavioral tests. Although we would need to study age-related functional changes by means of electrophysiological analysis to examine the relationship between the Cav2.1 channel and neurotransmitter release in rol/+ mice, the present results indicate that age-related change in nociceptive response to mechanical, thermal, and chemical stimuli in rol/+ might arise from alterations of neurotransmitter mechanisms underlying the deterioration of depolarization-induced Ca2+ influx through the Cav2.1 channel.
This work was supported by China 973 project (2010CB529604) and National Scientific Foundation of China (81271511 and 30900432).
The authors declare no competing interests.
WL and ET designed and supervised the research, and wrote the manuscript. YZ and KN performed the surgeries and behavioral experiments. All authors read and approved the final version of the manuscript.
- Catterall WA, Few AP (2008) Calcium channel regulation and presynaptic plasticity. Neuron 59: 882-901. [Crossref]
- Evans RM, Zamponi GW (2006) Presynaptic Ca2+ channels--integration centers for neuronal signaling pathways. Trends Neurosci 29: 617-624. [Crossref]
- Jarvis SE, Zamponi GW (2007) Trafficking and regulation of neuronal voltage-gated calcium channels. Curr Opin Cell Biol 19: 474-482. [Crossref]
- Mori Y, Wakamori M, Oda S, Fletcher CF, Sekiguchi N, et al. (2000) Reduced voltage sensitivity of activation of P/Q-type Ca2+ channels is associated with the ataxic mouse mutation rolling Nagoya (tg(rol)). J Neurosci 20: 5654-5662. [Crossref]
- Oda S (1973) The observation of rolling mouse Nagoya (rol), a new neurological mutant, and its maintenance (author's transl). Jikken Dobutsu 22: 281-288. [Crossref]
- Tanaka Y, Ando S (2001) Age-related changes in the subtypes of voltage-dependent calcium channels in rat brain cortical synapses. Neurosci Res 39: 213-220. [Crossref]
- Takahashi E, Niimi K (2009) Spatial learning deficit in aged heterozygous Cav2.1 channel mutant mice, rolling mouse Nagoya. Exp Gerontol 44: 274-279. [Crossref]
- Dickstein DL, Kabaso D, Rocher AB, Luebke JI, Wearne SL, et al. (2007) Changes in the structural complexity of the aged brain. Aging Cell 6: 275-284. [Crossref]
- Foster TC (2007) Calcium homeostasis and modulation of synaptic plasticity in the aged brain. Aging Cell 6: 319-325. [Crossref]
- Mora F, Segovia G, del Arco A (2007) Aging, plasticity and environmental enrichment: structural changes and neurotransmitter dynamics in several areas of the brain. Brain Res Rev 55: 78-88. [Crossref]
- Harkins SW, Price DD, Martelli M (1986) Effects of age on pain perception: thermonociception. J Gerontol 41: 58-63. [Crossref]
- Akunne HC, Soliman KF (1994) Serotonin modulation of pain responsiveness in the aged rat. Pharmacol Biochem Behav 48: 411-416. [Crossref]
- Gagliese L, Melzack R (1999) Age differences in the response to the formalin test in rats. Neurobiol Aging 20: 699-707. [Crossref]
- Iwamoto M, Hagishita T, Shoji-Kasai Y, Ando S, Tanaka Y (2004) Age-related changes in the levels of voltage-dependent calcium channels and other synaptic proteins in rat brain cortices. Neurosci Lett 66: 277–281.
- Takahashi E, Sagane K, Nagasu T, Kuromitsu J (2006) Altered nociceptive response in ADAM11-deficient mice. Brain Res 1097: 39-42. [Crossref]
- Meunier CJ, Burton J, Cumps J, Verbeeck RK (1998) Evaluation of the formalin test to assess the analgesic activity of diflunisal in the rat. Eur J Pharm Sci 6: 311-316. [Crossref]
- Chapman CR, Casey KL, Dubner R, Foley KM, Gracely RH, et al. (1985) Pain measurement: an overview. Pain 22: 1-31. [Crossref]
- Garcia-Nicas E, Laird JM, Cervero F (2001) Vasodilatation in hyperalgesic rat skin evoked by stimulation of afferent A beta-fibers: further evidence for a role of dorsal root reflexes in allodynia. Pain 94: 283-291. [Crossref]
- Wall PD, Melzack R (1999) Assessing transient and persistent pain in animals, In: R. Dubner and K. Ren (Eds.), Textbook of Pain, Churchill Livingstone, Edinburgh 360–362.
- Kimura M, Katayama K, Nishizawa Y (1999) Role of glutamate receptors and voltage-dependent calcium channels in glutamate toxicity in energy-compromised cortical neurons. Jpn J Pharmacol 80: 351-358. [Crossref]
- Lee CY, Chen CC. Liou HH (2009) Levetiracetam inhibits glutamate transmission through presynaptic P/Q-type calcium channels on the granule cells of the dentate gyrus. Br J Pharmacol 158: 1753–1762. [Crossref]
- Tan PH, Yang LC, Shih HC, Lan KC, Cheng JT (2005) Gene knockdown with intrathecal siRNA of NMDA receptor NR2B subunit reduces formalin-induced nociception in the rat. Gene Ther 12: 59-66. [Crossref]

