MACI® (autologous cultured chondrocytes on porcine collagen membrane), an autologous cellularized scaffold product, obtained United States Food and Drug Administration (FDA) approval in 2016 for the repair of symptomatic, single or multiple full-thickness cartilage defects of the knee with or without bone involvement in adults. The approval was supported by the pivotal 2‑year SUMMIT (Superiority of MACI Implant Versus Microfracture Treatment) study, which was a prospective Phase 3 multicenter, randomized, open‑label, parallel group clinical study conducted in 144 adult patients (72 MACI and 72 microfracture). The study demonstrated superior efficacy of MACI compared with arthroscopic microfracture in the treatment of patients, ages 18 to 54 years old, with at least 1 symptomatic Outerbridge Grade III or IV focal cartilage defect of the knee.
In accordance with the Pediatric Research Equity Act, Vericel and FDA agreed upon an initial pediatric study plan to conduct a study in patients aged 10 to 17 years; the plan was submitted with the MACI Biologics License Application. The ongoing post-marketing study, which was required as a condition of FDA approval, “PEdiatric Autologous cultured chondrocytes treatment of cartilage defects in the Knee” (PEAK) is a 2‑year prospective, multicenter, randomized, open-label, parallel group clinical trial being conducted at 10 sites in the US (ClinicalTrials.gov identifier NCT03588975). A total of 45 patients, ages 10 to 17 years, will be randomized to receive a one‑time treatment with MACI or microfracture (2:1, 30 MACI:15 microfracture). The primary efficacy endpoint is the percentage of patients who respond to study treatment after 2 years, defined as patients who have ≥10‑point improvement on both the pain and function (sports and recreational activities) subscales of the Knee Injury and Osteoarthritis Outcome Score (KOOS)-Child from baseline to Year 2. The PEAK study is the only ongoing randomized controlled trial studying chondral and osteochondral defect treatment options in children and adolescents to date which incorporates the FDA’s feedback on post-marketing study requirements for the treatment of cartilage defects in pediatric patients.
Autologous Chondrocyte Implantation (ACI), cartilage, Osteochondritis Dissecans (OCD), pediatric research, randomized controlled trial
Chondral and osteochondral defects of the knee in children are commonly due to acute trauma and unsalvageable osteochondritis dissecans (OCD) lesions. Either etiology leads to the issue of a focal articular cartilage defect in the knee, similar to a “pothole on a good road,” in the setting of an active child with a long life ahead. In children, articular cartilage injuries that do not heal properly can result in long-term morbidity [1] and can consume considerable healthcare resources [2].
The natural history of focal articular cartilage defects left empty is known to be suboptimal. Anderson et al first reported on 19 young patients (mean age 21 years) with 20 OCD lesions that were evaluated a mean of 9 years after fragment excision alone [3]. When considered on the International Knee Documentation Committee (IKDC) scale, 2 knees were rated normal, 6 nearly normal, 4 abnormal, and 8 severely abnormal. The authors concluded that the “long-term results are extremely poor” [3]. Similarly, Wright et al studied 17 patients that were evaluated a mean of 9 years after fragment excision alone and found that 11 of 17 patients (65%) had a fair or poor result [4]. Sanders et al., reported on 134 OCD patients that were evaluated a mean of 17 years after fragment excision and found that twenty patients (~15%) underwent knee arthroplasty [5].
Several approaches exist to fill and repair the focal articular cartilage defects. These include marrow stimulation techniques including microfracture, autologous chondrocyte implantation (ACI), osteochondral autograft transfer (OAT), and fresh osteochondral allograft (OCA) transplantation [6]. Few studies have focused on observing outcomes for these treatments in the pediatric population.
In fact, we are aware of only one publication of a prospective randomized clinical trial for treating focal articular cartilage injury in children [7]. This study evaluated OAT versus microfracture and showed comparable clinical benefit of both treatments, although the microfracture group showed diminished benefit over the 4.2‑year follow-up [7]. There have been no randomized controlled trials in children comparing ACI to OAT, OCA, or microfracture.
Rationale for a pediatric study
Four large case series evaluating ACI in pediatrics have been reported to date with ACI Carticel [8-10] and ACI/MACI [2]. In addition, an industry-sponsored prospective Registry-based study included a total of 37 pediatric patients treated with Carticel [11], and an industry-sponsored retrospective cohort study included 29 adolescents treated with third-generation ACI using spheroids [12]. Carticel treatment was associated with improved functional outcomes in the Mithöfer (2005) case series of 20 patients ≤18 years of age (mean age 15.9 years) with a mean follow-up of 47 months [8], and in the Rush University case series of 37 patients <18 years of age (mean age 16.7 years) with a mean follow-up of 55 months [9]. Results from the Mithöfer case series were similar to those seen in adults, where greater functional improvement post-Carticel treatment correlated with shorter duration of symptoms before implantation and a lower number of prior operations. Patients with a shorter duration of preoperative symptoms also saw greater clinical improvement in a systematic review of clinical outcomes following ACI in adolescent knees [13]. Long-term outcomes were also reported in a case series of 27 pediatric patients (13 – 17 years of age, mean age 15.9 years) treated with ACI [10]. Approximately 90% rated knee-specific outcomes as good or excellent and were satisfied with the procedure over a mean 9.6-year follow-up period [10]. In the Macmull (2011) case series evaluating 35 patients 14 to 18 years of age (mean 16.3 years), ACI and MACI treatment resulted in significant improvements in function and subjective pain scores over a mean 66-month follow-up period [2]. Higher success rates were attained among pediatric patients in this study than in studies of adult patients.
The Micheli study of 37 adolescents was part of a voluntary Registry-based study designed in collaboration with FDA to follow patients who received a Carticel implant [11]. Patients 11 to 17 years of age (mean age 16 years) reported significant improvements in their overall condition and marked reduction in pain and swelling. These results are similar to those observed in adults. The retrospective cohort study of chondrospheres in 28 adolescents 15 – 17 years of age compared patient reported outcomes to outcomes from 42 young adults 18 – 34 years of age and found that both groups had improved pain and functional scores but that there were not statistically significant differences between the two age groups [12]. A systematic review of the literature for articular cartilage repair in pediatric and adolescent patients showed that ACI, as well as other available cartilage repair procedures, provides postoperative improvement above published minimal clinically important difference [13]. No randomized controlled trials have been conducted to date evaluating the safety or efficacy of ACI or MACI in pediatric patients.
In accordance with the Pediatric Research Equity Act, which gives FDA the authority to require the conduct of pediatric studies for certain products when these drugs are not adequately labeled for children [14], MACI manufacturer Vericel and FDA agreed upon an initial pediatric study plan as part of the MACI Biologics License Application (BLA). As MACI is unlikely to be used in children under the age of 10 years, Vericel submitted a request for a partial pediatric waiver for children less than 10 years of age. Vericel also submitted a request for a deferral to conduct a postmarketing study to evaluate the safety and efficacy of MACI in patients aged 10 to 17 years. The FDA Pediatric Review Committee (PeRC) recommended a partial waiver in patients ages birth through 9 years because knee cartilage defects are rare or do not exist in children in this age group. The PeRC also recommended a deferral of a postmarketing study in patients ages 10 to 17 years. The agreement included a commitment to complete the study by June 2025. Upon MACI BLA approval, completion of the study in children and adolescents became a postmarketing requirement. The agreed-upon objective of the study is to compare the efficacy and safety of MACI vs arthroscopic microfracture in the treatment of patients aged 10 to 17 years with symptomatic articular chondral or osteochondral defects of the knee.
Treatments to be studied
Microfracture is considered by some to be the standard of care for the treatment of chondral defects and many cartilage repair studies in adults include microfracture treatment arm as an active control [15]; it is considered the first-line therapy for smaller defects [16]. The few small studies (10 to 26 patients) of microfracture in adolescent patients (growth plate status not reported) generally demonstrated improved function and activity [7,17,18].
ACI is a 2‑step procedure process that involves first arthroscopically harvesting a biopsy of hyaline cartilage from non-weight-bearing areas of the knee, culturing chondrocytes from the biopsied tissue ex vivo, and then implanting the cultured chondrocytes into the cartilage defect in a second surgical procedure. Carticel was the only ACI product available in the United States (US) until 2016 and had been used in the US for the treatment of chondral defects in patients aged 10 to 18 years since 1995. In 2016, FDA approved MACI® (autologous cultured chondrocytes on porcine collagen membrane), an autologous cellularized scaffold product, for the repair of symptomatic, single or multiple full-thickness cartilage defects of the knee with or without bone involvement in adults. FDA approval was based on the pivotal 2‑year SUMMIT study which was a prospective Phase 3 multicenter, randomized, open‑label, parallel group clinical study conducted in 144 adult patients (72 MACI and 72 microfracture). The study demonstrated superior efficacy of MACI compared with arthroscopic microfracture in the treatment of patients, ages 18 to 54 years old, with at least 1 symptomatic Outerbridge Grade III or IV focal cartilage defect of the knee [19,20].
Study design
The Vericel-sponsored PEdiatric Autologous cultured chondrocytes treatment of cartilage defects in the Knee (PEAK) Study, which began enrollment in October 2018, is an ongoing prospective, multicenter, randomized, open-label, parallel group clinical trial being conducted at 10 sites in the US (ClinicalTrials.gov identifier NCT03588975). A total of 45 patients, ages 10 to 17 years, will be randomized to receive a 1‑time treatment with MACI or microfracture (2:1, 30 MACI:15 microfracture, Figure 1).
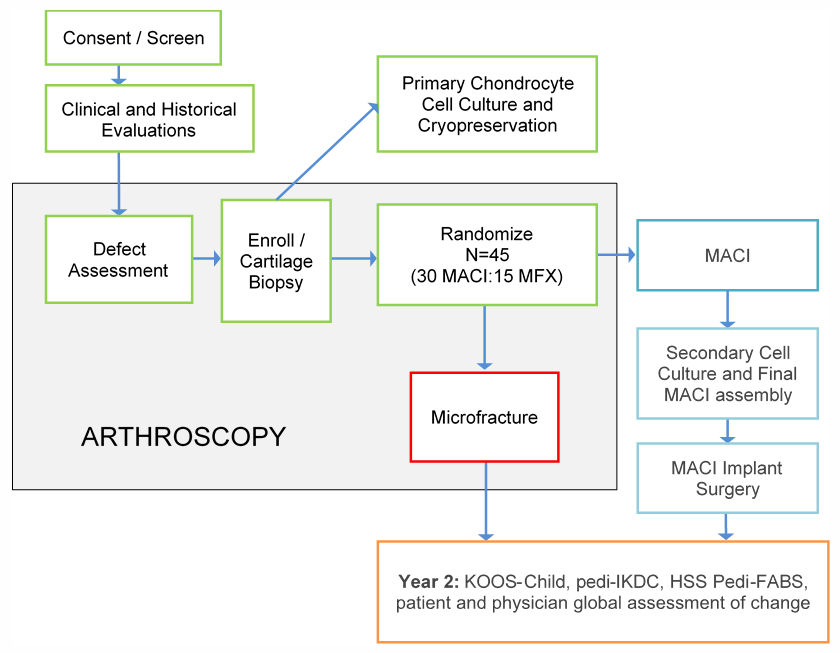
Figure 1. Study design.
Abbreviations: HSS Pedi-FABS = Hospital for Special Surgery Pediatric Functional Activity Brief Scale; IKDC= International Knee Documentation Committee; KOOS= Knee Injury and Osteoarthritis Outcome Score.
The primary efficacy analysis will be performed 2 years following administration of study treatment. The choice of microfracture as a comparator is consistent with FDA guidance for the design of studies in cartilage repair of the knee [21]. Due to differences in surgical techniques between MACI and microfracture, the study design is, by necessity, open label.
The study is being conducted in accordance with the International Conference on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use Good Clinical Practice guidelines and with the approval of an institutional review board at each site. All legal guardians of the study participants are required to sign an informed consent prior to study.
enrollment. Site locations in the US can be found on ClinicalTrials.gov identifier NCT03588975.
An overview of participant eligibility criteria is provided in Table 1 and complies with the current clinical algorithm for the treatment of OCD in the pediatric patient [22]. Although there is no upper limit of defect size specified in the protocol, surgeons will likely limit the upper defect size treated given the randomization to microfracture or MACI. In addition, bone autografting for osteochondral defects with less than 6 mm of bone loss is not considered necessary for successful treatment [23].
Table 1. Eligibility criteria.
Inclusion criteria |
Exclusion criteria |
- Symptomatic cartilage or osteochondral defects as a result of acute trauma or osteochondritis dissecans (OCD)
|
- Any surgery on the target knee joint within 6 months prior to screening (not including diagnostic arthroscopy)
|
- One or more International Cartilage Repair Society (ICRS) Grade III or IV chondral or unsalvageable osteochondral defects located on the femoral condyles and/or trochlea amenable to treatment with the surgical procedure determined at randomization (MACI or microfracture)
|
- ICRS Grade III or IV chondral or unsalvageable osteochondral defects located on the patella or tibia or any lesion that is bipolar to the index lesion.
|
- At least 1 defect size ≥2.0 cm2 on the femoral condyles and/or the trochlea; defects include OCD lesions with a bone lesion depth of ≤6 mm and does not require a bone graft
|
Concomitant inflammatory disease or other conditions that affects the joints. |
- Stable target knee (ie, anterior and posterior cruciate ligaments should be free of laxity as well as stable and intact). Ligament repair or reconstruction procedures are allowed a minimum of 6 months prior to screening arthroscopy.
|
- Known history of septic arthritis in the index knee joint.
|
- Intact meniscus or partial meniscus (at least 50% of functional meniscus remaining) in the target knee.
|
- Known history of hypersensitivity to gentamicin, other aminoglycosides, or products of porcine or bovine origin
|
|
- Females who are pregnant or lactating
|
After meeting screening criteria at the initial visit, all patients will have a screening arthroscopy to further assess study eligibility. During the screening arthroscopy, patients will be further evaluated against entry criteria. Cartilage lesion size will be measured prior to any cartilage repair procedure and randomization. All patients who meet the eligibility criteria and are considered suitable for treatment in the study will have a cartilage biopsy taken prior to randomization to study treatment. Eligible patients will be randomized during the screening arthroscopy procedure to receive either MACI or microfracture treatment. Randomization will be conducted via a centralized web-based interactive response technology. Patients randomized to microfracture will undergo the procedure during the screening arthroscopy.
All biopsied tissue will be sent to the Vericel manufacturing facility in Cambridge, Massachusetts, where the sample will be accessioned, chondrocytes cultured, and cryopreserved. Cells from patients randomized to the MACI group will be used in the preparation of the MACI implant; cells from patients randomized to the microfracture group will be stored in case of future use. Cryopreserved cells maintain viability for a minimum of 5 years. Upon MACI order receipt, cells are thawed and undergo secondary cell culture expansion to produce an adequate number of cells for MACI assembly. MACI is tested and released prior to shipment for surgery. MACI implant is available as a cellular sheet, 3 x 5 cm, with a 0.5-cm2 section removed from the lower left-hand corner. It consists of autologous cultured chondrocytes on a resorbable Type I/III collagen membrane at a density of at least 500,000 cells per cm2. Rigorous assay testing (cell viability, minimum cell number, chondrocyte identity, potency, sterility, endotoxin and mycoplasma) is performed on each MACI lot prior to product release.
Patients randomized to treatment with MACI will return within 12 weeks of the screening arthroscopy to undergo the MACI implantation procedure via arthrotomy. Patients are to follow a physician-prescribed postoperative rehabilitation program. The rehabilitation program will be the based on the treatment received (MACI or microfracture). Guidance for rehabilitation following MACI is based on Ebert 2017 publication [24] and that for microfracture is based on Steadman 2001 [25]. Both programs are staged to promote a progressive return to full joint range of motion and weight bearing as well as muscle strengthening and conditioning. Compliance with the rehabilitation schedule will be monitored. Patients will be assessed post-study treatment at Weeks 6 and 12 for safety and at Weeks 12, 24, 52, and 104 for safety and efficacy.
The primary efficacy endpoint is the percentage of patients who respond to study treatment after 2 years (“responders”), ie, patients who have ≥10‑point improvement on both the pain and function (sports and recreational activities [SRA]) subscales of the Knee Injury and Osteoarthritis Outcome Score (KOOS)-Child [26,27] from baseline to Week 104. Secondary efficacy variables include the change from baseline in KOOS-Child pain and function (SRA) subscales, other KOOS subscales, the pedi- IKDC subjective knee evaluation form, Hospital for Special Surgery Pediatric Functional Activity Brief Scale (HSS Pedi-FABS), and physician and patient global assessment of change.
Statistical considerations
Sample size is selected to provide a reasonably precise estimate of the difference in responder rates between MACI and microfracture as measured by the width of a two-sided 90% confidence interval. No hypothesis testing is planned. Assuming underlying responder rates of 88% and 68% for MACI and microfracture, respectively, a total of 45 patients at a 2:1 allocation (MACI: microfracture) will permit estimation of the difference in responder rates with 90% confidence interval limits ±22% of the point estimate.
Simulations of study results
Because the study sample size is limited, simulations were performed to assess the probability that the responder rate in the 30 MACI patients exceeds that of 15 microfracture patients based on the assumption of an 88% responder rate in MACI and a 68% responder rate for microfracture as observed in the adult study [19]. The underlying statistical concept for the analysis of responder rates is the inference of proportions and binomial distribution. It concerns n independent “experiments” with a probability of p for success and 1-p for failure. Thus, n is the number of patients randomized to the clinical trial, p is the probability of response and 1-p is the probability of non-response. The simulation used a binomial random number generator performed in SAS 9.4 RAND function for the number of successes for each group based on these two rates. One million sets of simulated response rates (1 million simulated studies of 45 patients) were generated for assumed response rates for the two arms based on the adult data. In addition, the responder rate for microfracture was increased to assess the probability of MACI responder rate being greater than that of microfracture. The responder rate for MACI patients exceeded that of microfracture in 91.7% of the simulations using the same responder analysis observed for adults. Additional simulations varying the responder rate in the microfracture group showed that there was a >80% probability that MACI responder rate exceeds that of microfracture as long as microfracture responder rate is 75% or less.
Data extrapolation
Because MACI will be used in the pediatric population for the same indication as approved in adults, the disease process is similar in adults and pediatric patients, and the clinical outcomes of therapy is likely to be similar, extrapolation from adult efficacy data is planned. Change from baseline in KOOS pain and function subscores for adults will be compared to results obtained in the pediatric study, limiting the patients to those with closed growth plates. Although no patients <18 years old were included in the SUMMIT study (6 patients were 18 to <22 years old), the change from baseline in KOOS pain and function scores by patient age was evaluated by linear regression. Among the 72 patients (age range 18 to 54) treated with MACI in SUMMIT, no statistically significant association was observed between patient age and improvement in KOOS pain or function scores 2 years following MACI treatment (Figure 2).
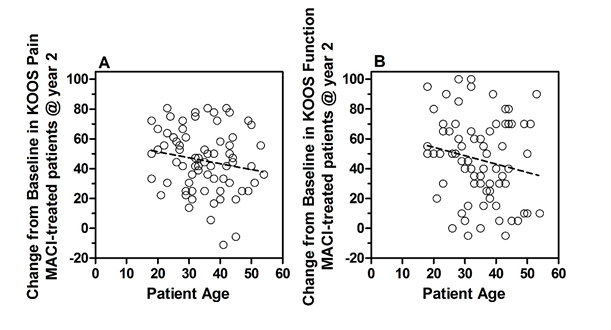
Figure 2. Change from baseline in KOOS Pain and function in SUMMIT study patients treated with MACI.
Regression analyses were performed on the change from baseline in KOOS Pain (A) and KOOS Function Sports and Recreational activities (B). The mean change in KOOS pain at 2 years was 45 points and linear regression was not statistically significantly different from zero (p = 0.145; r2 = 0.03). The mean change in KOOS function at 2 years was 46 points and linear regression was not statistically significantly different from zero (p = 0.126; r2 = 0.03).
These results, along with the literature data, suggest that the response of younger patients to MACI implantation is no worse or may be better than that of older patients.
Study status
As of December 12, 2020, a total of 18/45 (40%) patients have been randomized and treated since October 2018 in the PEAK study. A majority of the enrolled patients are male (73%) and 80% are 13-16 years age. All patients randomized to date have a single defect; of the patients treated (n=18), the average defect size was 3.75 cm2 and 75% of the treated patients had unsalvageable OCD lesions.
Oversight
The MACI PEAK Steering Committee, comprised of the Vericel Chief Medical Officer and manuscript authors, was established to provide medical and scientific expertise. The Committee contributed to the development of the clinical trial protocol and amendments, provided guidance on the selection of clinical trial sites, and is providing ongoing oversight of the trial implementation to ensure study quality and integrity. A Safety Monitoring Plan is in place to ensure periodic review of any potential adverse events as well as evaluate study conduct. Periodic study status reports are also provided to FDA.
The PEAK study is the only ongoing randomized controlled trial studying chondral and osteochondral defect treatment options in children and adolescents to date and reflects the current thinking of the FDA on post marketing study requirements for the treatment of cartilage defects in pediatric patients. Strengths of the study design include randomization, multicenter nature of the study, as well as the use of age-validated disease appropriate patient reported outcomes measures for primary and secondary efficacy measures.
Although randomized controlled trials provide the highest level evaluation of clinical effectiveness, challenges exist regarding the design and conduct of randomized controlled trials in pediatric patients in general and for an orthopedic indication specifically where underpowered studies are not uncommon [28,29]. The ethical feasibility of conducting studies in pediatric patients <18 years has been called into question by the ICRS due to the legal and practical implications related to the consent process and the ethical treatment of minors [15].
Because the incidence of symptomatic cartilage lesions in the 10 to 17 years pediatric age group requiring surgical restoration is quite rare [6,11], significant challenges are anticipated to enroll an adequate number of pediatric patients. Thus, the clinical study is limited in scope (45 patients planned to be treated and no hypothesis testing) with a 4.5 year enrollment period. Limitations of the PEAK study include the relatively small sample size which limits the power to perform statistical testing between treatment groups. The validity of the results of this study is not negated by the small sample size given the rigorous study design and conduct, multiple secondary endpoint assessments to support the primary efficacy analysis, and the availability of adult data for extrapolation.
Additional limitations of the study design include lack of stratification for OCD vs chondral defects because of the limited sample size. However, in an attempt to minimize confounders, the study limits defect location to the femur and excludes subjects requiring concomitant ligament repair. The sponsor and FDA aimed to design a study that could be completed in the proposed timeframe, thereby limiting the sample size.
Slow completion rates for pediatric postmarketing studies have been reported [30], although these slow rates do not necessarily reflect noncompliance with FDA mandated postmarketing requirements. A majority (82%) of open BLA postmarketing requirements are considered on-schedule by FDA and only 7% of open PREA BLA postmarketing requirements are delayed [31]. In certain situations, the original postmarketing requirement schedules were adjusted for unanticipated delays in the progress of the clinical trial. These data suggest FDA’s acceptance of sponsor’s slow but potentially more realistic timelines for study completion.
Continued research is needed for the optimal management of OCD lesions in pediatric patients [32]. The American Academy of Orthopaedic Surgeons (AAOS) Clinical Practice Guideline on OCD of the Knee [33] acknowledged that the field lacked high quality research. Results from randomized controlled studies such as this designed after the pivotal study in adults [19], will contribute to our understanding of optimal treatments and result in better outcomes for this patient population.
All 3 authors made substantial contributions to the clinical trial design, approved the final draft of the manuscript, and guarantee the accuracy; AR drafted the manuscript and JC and TG revised it for important intellectual content.
The authors thank John Ilgenfritz for help with simulations, and Fang Dong and Hongbo Li of Vericel Corporation for clinical programming and contribution to the simulations of study results.
- Sanders TL, Pareek A, Obey MR, Johnson JR, Carey JL, et al. (2017a) High Rate of Osteoarthritis After Osteochondritis Dissecans Fragment Excision Compared with Surgical Restoration at a Mean 16-Year Follow-up. Am J Sports Med 45: 1799-1805. [Crossref]
- Macmull S, Parratt MTR, Bentley G, Skinner JA, Carrington RWJ, et al. (2011) Autologous chondrocyte implantation in the adolescent knee. Am J Sports Med 39: 1723‑1730. [Crossref]
- Anderson AF, Pagnani MJ (1997) Osteochondritis Dissecans of the Femoral Condyles Long-term Results of Excision of the Fragment. Am J Sports Med 25: 830-834. [Crossref]
- Wright RW, McLean M, Matava MJ, Shively RA (2004) Osteochondritis Dissecans of the Knee Long-term results of excision of the fragment. Clin Ortho Relat Res 424: 239-243. [Crossref]
- Sanders TL, Pareek A, Johnson NR, Carey JL, Maak TG, et al. (2017b) Nonoperative management of osteochondritis dissecans of the knee: progression to osteoarthritis and arthroplasty at mean 13-year follow-up. Orthop J Sports Med 5: 2325967117704644. [Crossref]
- McCormick F, Harris JD, Abrams GD, Frank R, Gupta A, Hussey K, et al. (2014) Trends in the surgical treatment of articular cartilage lesions in the United States: an analysis of a large private-payer database over a period of 8 years. Arthroscopy 30: 222-226. [Crossref]
- Gudas R, Simonaityte R, Cekanauskas E, Tamosiunas R (2009) A prospective, randomized clinical study of osteochondral autologous transplantation versus microfracture for the treatment of osteochondritis dissecans in the knee joint in children. J Pediatr Orthop 29: 741‑748. [Crossref]
- Mithöfer K, Minas T, Peterson L, Yeon H, Micheli LJ (2005) Functional outcome of knee articular cartilage repair in adolescent athletes. Am J Sports Med 33: 1147‑1153. [Crossref]
- Cvetanovich GL, Riboh JC, Tilton AK, BJ Cole (2017) Autologous Chondrocyte Implantation Improves Knee-Specific Functional Outcomes and Health-Related Quality of Life in Adolescent Patients. Am J Sports Med 45: 70-76. [Crossref]
- Ogura T, Bryant T, Minas T (2017) Long-term outcomes of autologous chondrocyte implantation in adolescent patients. Am J Sports Med 45: 1066-1074. [Crossref]
- Micheli LJ, Moseley JB, Anderson AF, Browne JE, Erggelet C, et al. (2006) Articular cartilage defects of the distal femur in children and adolescents: treatment with autologous chondrocyte implantation. J Pediatr Orthop 26: 455‑460. [Crossref]
- Hoburg A, Loer I, Korsmeier K, Siebold R, Niemeyer P, et al. (2019) Matrix-Associated Autologous Chondrocyte Implantation Is an Effective Treatment at Midterm Follow-up in Adolescents and Young Adults. Orthop J Sports Med 7: 2325967119841077. [Crossref]
- DiBartola AC, Wright BM, Magnussen RA, DC Flanigan (2016) Clinical Outcomes After Autologous Chondrocyte Implantation in Adolescents' Knees: A Systematic Review. Arthroscopy 32: 1905-1916. [Crossref]
- Penkov D, Tomasi P, Eichler I, Murphy D, Yao LP, et al. (2017) Pediatric Medicine Development: an overview and comparison of regulatory processes in the European Union and United States. Ther Innov Regul Sci 51: 360-371. [Crossref]
- Mithoefer K, Saris DBF, Farr J, Kon E, Zaslav K, et al. (2011) Guidelines for the design and conduct of clinical studies in knee articular cartilage repair: International Cartilage Repair Society recommendations based on current scientific evidence and standards of clinical care. Cartilage 2: 100‑121. [Crossref]
- Oussedik S, Tsitskaris K, Parker D (2015) Treatment of articular cartilage lesions of the knee by microfracture or autologous chondrocyte implantation: a systematic review. Arthroscopy. 2015; 31:732‑744. [Crossref]
- Salzmann GM, Sah B-R, Schmal H, Niemeyer P, Sudkamp NP (2012) Microfracture for treatment of knee cartilage defects in children and adolescents. Pediatr Rep 4: e21. [Crossref]
- Steadman JR, Briggs KK, Matheny LM, Guillet A, Hanson CM, et al. (2015) Outcomes following microfracture of full-thickness articular cartilage lesions of the knee in adolescent patients. J Knee Surg 28: 145‑150. [Crossref]
- Saris D, Price A, Widuchowski W, Bertrand-Marchand M, Caron J, et al. (2014) Matrix-applied characterized autologous cultured chondrocytes versus microfracture two-year follow-up of a prospective randomized trial. Am J Sports Med 42: 1384‑1394. [Crossref]
- Brittberg M, Recker D, Ilgenfritz J, Saris DBF, SUMMIT Extension Study Group (2018) Matrix-applied characterized autologous cultured chondrocytes versus microfracture five-year follow-up of a prospective randomized trial. Am J Sports Med 46: 1343-1351. [Crossref]
- FDA Guidance for Industry (2011) Preparation of IDEs and INDs for Products Intended to Repair or Replace Knee Cartilage. December 2011.
- Ganley TJ, Flynn JM (2011) Osteochondritis dissecans. In: Scott WN, editor. Insall & Scott – Surgery of the knee. 5th ed. Philadelphia: Churchill Livingstone. Chapter 87.
- Minas T, Ogura T, Headrick J, T Bryant (2018) Autologous Chondrocyte Implantation ‘‘Sandwich’’ Technique Compared with Autologous Bone Grafting for Deep Osteochondral Lesions in the Knee. Am J Sports Med 46: 322-332. [Crossref]
- Ebert JR, Edwards KP, Fallon M, Ackland TR, Janes GC, et al. (2016) Two-Year Outcomes of a Randomized Trial Investigating a 6-Week Return to Full Weightbearing After Matrix-Induced Autologous Chondrocyte Implantation. Am J Sports Med 45: 838-848. [Crossref]
- Steadman JR, Rodkey WG, Rodrigo JJ (2001) Microfracture: Surgical Technique and Rehabilitation to Treat Chondral Defects. Clin Orthop Relat Res 391S: S362-S369. [Crossref]
- Ortqvist M, Roos EM, Brostron EW, Janarv P-M, Iverson MD (2012) Development of the Knee Injury and Osteoarthritis Outcome Score for Children (KOOS-Child) comprehensibility and content validity. Acta Orthopaedica 83: 666‑673. [Crossref]
- Ortqvist M, Iversen MD, Janarv P-M, Brostron EW, Roos EM (2014) Psychometric properties of the Knee injury and Osteoarthritis Outcome Score for Children (KOOS-Child) in children with knee disorders. Br J Sports Med 48: 1437-1446. [Crossref]
- Dodwell E. Dua S, Dulai SK, Astone K, K Mulpuri (2015) The Quality of Randomized Controlled Trials in Pediatric Orthopaedics: Are We Improving? J Pediatr Orthop 35: 536-545. [Crossref]
- Lyman S, Nakamura N, Cole BJ, Erggelet C, Gomoll AH, et al. (2016) Cartilage-Repair Innovation at a Standstill: Methodologic and Regulatory Pathways to Breaking Free. J Bone Joint Surg Am 98: e63. [Crossref]
- Hwang T, Orenstein L, Kesselheim AS, Bourgeois FT (2019) Completion rate and reporting of mandatory pediatric postmarketing studies under the US pediatric research equity act. JAMA Pediatr 173: 68-74. [Crossref]
- Food and Drug Administration, Department of Health and Human Services. Report on the Performance of Drugs and Biologics Firms in Conducting Post marketing Requirements and Commitments Fiscal Year 2019. November 2020. (https://www.fda.gov/media/140827/download) accessed December 10, 2020.
- Cruz Jr AE, Shea KG, Ganley TJ (2016) Pediatric knee osteochondritis dissecans lesions. Orthop Clin N Am 47: 763-775. [Crossref]
- Chambers HG, Shea KG, Carey JL (2011) American Academy of Orthopaedic Surgeons Clinical Practice Guideline: diagnosis and treatment of osteochondritis dissecans. J Am Acad Orthop Surg 19: 307-309. [Crossref]
Editorial Information
Editor-in-Chief
Michael A. Portman
University of Washington, USA
Article type
Research Article
Publication History
Received: January 15, 2021
Accepted: January 24, 2021
Published: January 29, 2021
Copyright
©2021 Carey JL. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation
Carey JL, Remmers AE, Ganley TJ (2020) A study of MACI in patients aged 10 to 17 years with symptomatic chondral or osteochondral defects of the knee: Design and rationale. Pediatr Dimensions 6: DOI: 10.15761/PD.1000212.


