Abstract
Tirzepatide, known commercially as Mounjaro, is a Glucagon-like peptide 1/glucose-dependent insulinotropic peptide (GIP) receptor agonist that has been recently popularised as a novel weight loss agent owing to its effects of delaying gastric emptying, increasing satiety and reducing overall food intake. The drug has been approved for use as in weight loss management by NICE within the UK and internationally. Adverse reactions from Tirzepatide use most commonly include nausea, vomiting and diarrhoea. Liver injury secondary to Mounjaro is currently poorly recognised and a literature search revealed only three previously published case reports of Tirzepatide-induced liver injury.
In this case, a 42-year-old female patient presented with a four-week history of pruritus, nausea, fatigue and dark urine. There was no history of travel, allergen exposure, recreational drug use, oral contraceptive therapy or excessive alcohol intake. The patient had a three-month history of Mounjaro use, which patient had stopped a few weeks prior to her attendance at the emergency department.
Serological tests found deranged liver function tests, including a serum AST of 1155 U/L and AST measuring 697U/L. An autoantibody screen and virology screen were negative. Ultrasound and MRCP both showed no abnormalities. A core biopsy of the liver showed mixed pattern of inflammation characterised by canalicular cholestasis, periportal chronic inflammation, focal interface activity and scattered lobular inflammation, consistent with drug-induced liver injury (DILI).
The mechanism of hepatic injury secondary to Mounjaro is unclear. Previously published case reports have highlighted the 5 day half-life and slow metabolism of the drug, which may explain the latent onset of symptoms. Ongoing monitoring of Tirzepatide-associated adverse effects is recommended.
Keywords
tirzepatide, mounjaro, liver, injury, hepatotoxicity, DILI
Introduction
Tirzepatide (also known commercially as Mounjaro), a Glucagon-like peptide 1(GLP-1) and glucose-dependent insulinotropic peptide (GIP) receptor agonist, is a drug historically used for management of triple therapy-resistant type two diabetes mellitus. GLP-1, synthesized by L-cells, functions like incretin by stimulating insulin secretion and inhibiting glucagon release, thus accounting for the role of GLP-1 receptor agonists in management in Type 2 Diabetes Mellitus [1]. GIP, like GLP-1, stimulates insulin secretion secondary to hyperglycaemia and additional, increases sensitivity of adipose tissue to insulin [2].
The extra-pancreatic effects of GLP-1 agonists, including delayed gastric emptying, increased satiety and reduced overall food intake, accounts for its promising new role in weight loss management [2]. Following publication of a technology appraisal publication by the National Institute for Health and Care Excellence (NICE) in December 2024 [3], Tirzepatide has been approved as a novel agent for weight loss management within the NHS (NICE, 2025). High popularity of the drug as a weight loss medication has led to rapid expansion of its use both within the UK and internationally. The SURPASS-5 trial, which published its results in 2022, assessed the efficacy of Tirzepatide in addition to insulin Glargine on type 2 diabetic patients with poor glycaemic control [4]. The results demonstrated that 68.1% to 78.3% of tirzepatide-treated patients experienced adverse events, the most common being gastrointestinal symptoms including diarrhoea and nausea [4]. Other less frequent complications included hypersensitivity reactions (6.9%) and injection site reactions (3.4%), and overall, no deaths occurred in this trial [4]. However, due to the swiftly expanding use of Tirzepatide as a weight loss agent, surveillance for newly reported adverse events should be carefully considered and reviewed.
This report discusses a case of acute liver injury likely secondary to use of Mounjaro, a rare complication which has only been reported in a small handful of published case reports.
Case presentation
The patient is a middle-aged female who presented to the emergency department with complaints of pruritus, nausea and fatigue for four weeks. There was no history of excessive alcohol intake, travel, allergen exposure, oral contraceptive use or recreational drug use. She reported use of 3-month history of Mounjaro use for weight loss, which she stopped a several weeks prior to her attendance.
Investigations performed at the initial presentation demonstrated deranged liver function tests, including elevated ALT of 1155 U/L and AST measuring 697 U/L. The total bilirubin measured 64 umol/L and the alkaline phosphatase was noted at 213 U/L. Serum IgG levels were elevated, however an autoantibody panel and virology screen were both negative. An abdominal ultrasound and MRCP showed no evidence of abnormalities.
A core biopsy taken from the right hepatic lobe showed periportal chronic inflammation without a prominent plasma cell component, and focal interface activity (Figure 1). Mild bile ductular proliferation and mild periportal fibrosis were noted and highlighted with CK7 immunohistochemistry (Figures 2 and 3). There was no evidence of bile duct injury or ductopenia. The hepatic lobules contained scattered chronic inflammatory infiltrate and mild canalicular cholestasis (Figures 4 and 5). Steatosis, confluent necrosis, Mallory-Denk bodies and granulomata were not identified.

Figure 1. Periportal chronic inflammation devoid of plasma cells, with focal interface activity
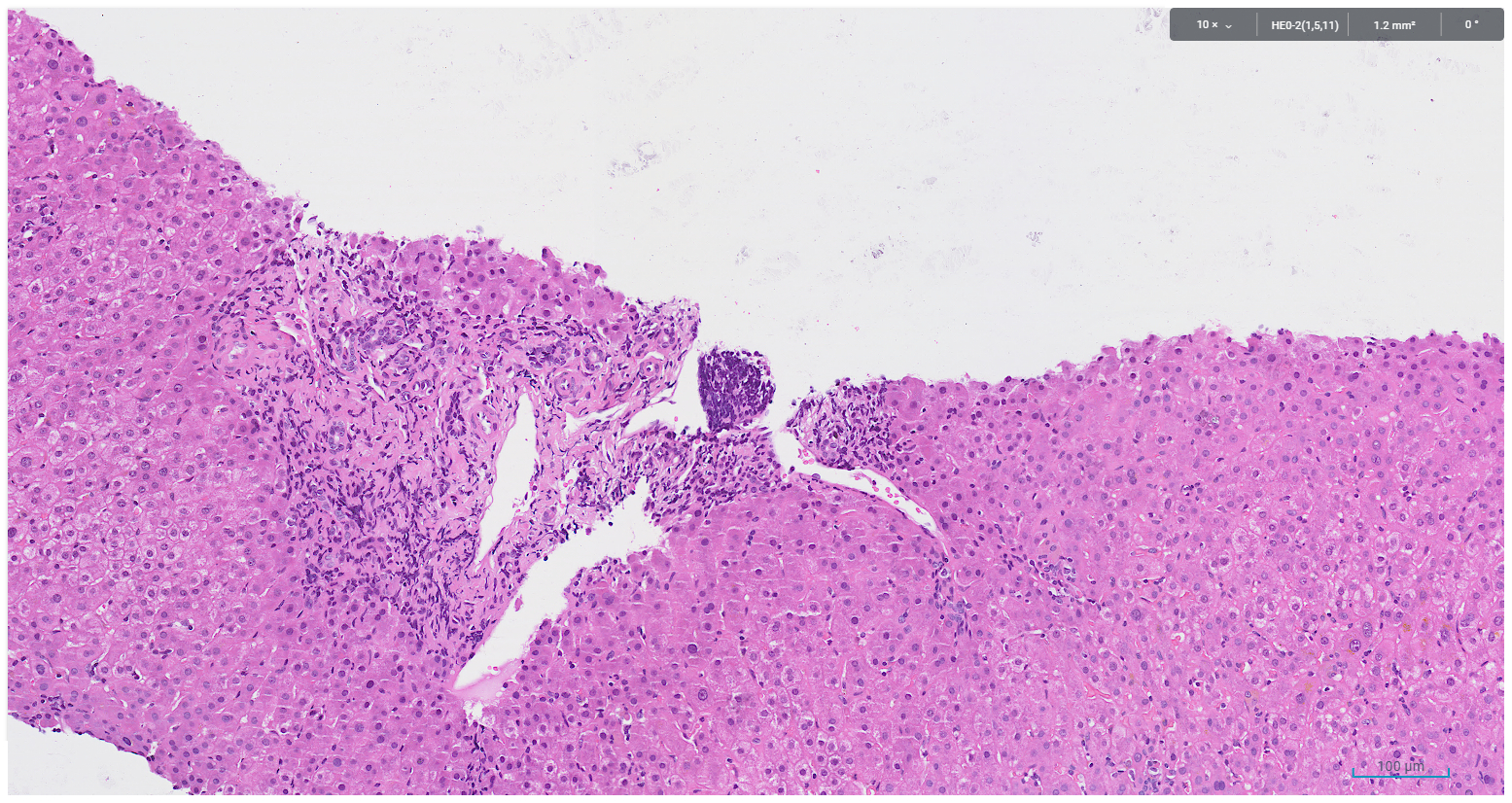
Figure 2. Bile ductular reaction, periportal fibrosis and chronic inflammation devoid of plasma cell infiltrate

Figure 3. legend: CK7 immunohistochemistry highlights bile ductular reaction at the corresponding site of figure 2

Figure 4. Canalicular cholestasis and scattered lobular lymphocytic infiltrate
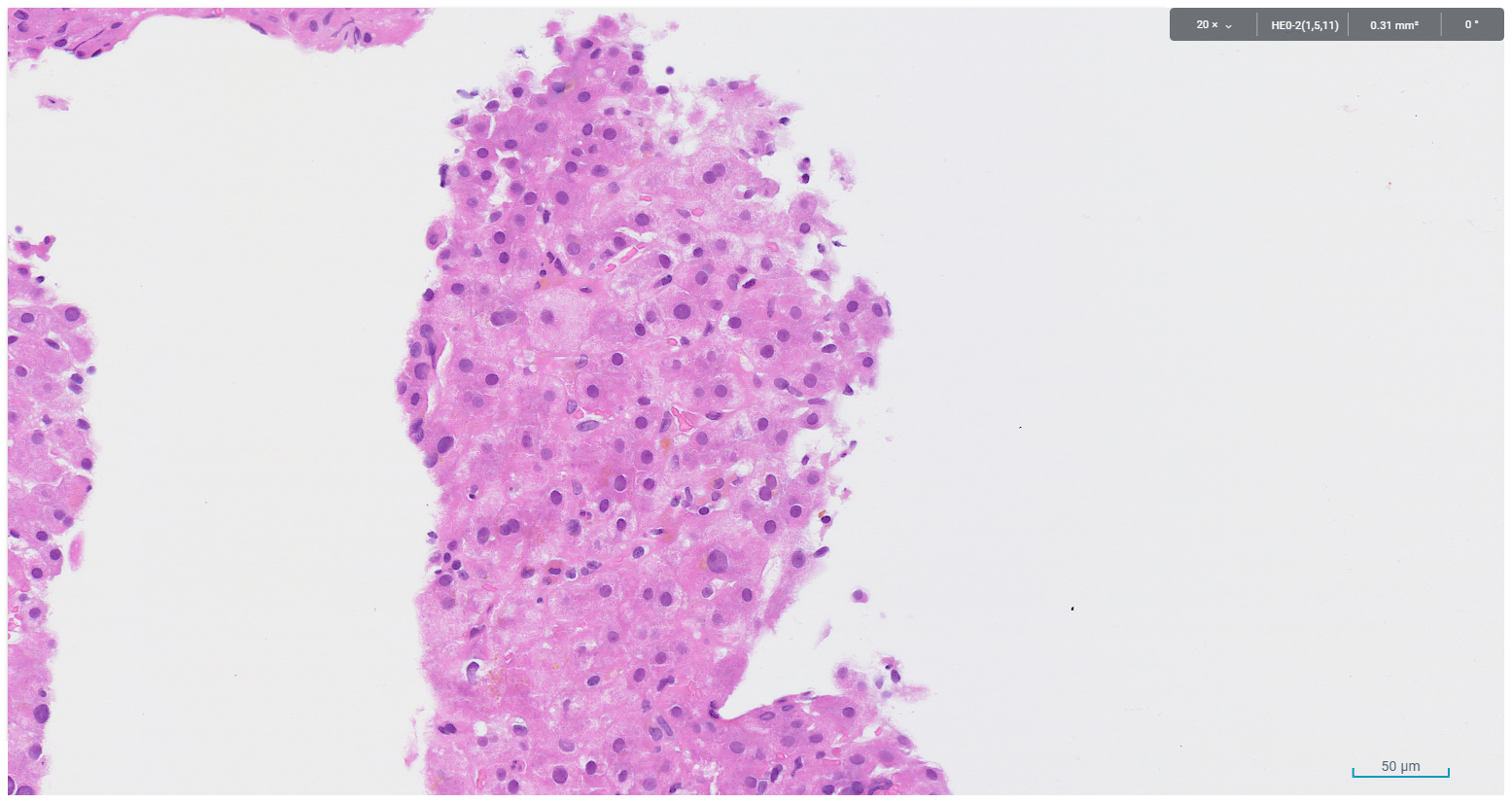
Figure 5. Canalicular cholestasis and scattered lobular lymphocytic infiltrate
PAS diastase stain highlighted ceroid pigment in Kupffer cells, a feature that can indicate non-specific hepatocellular injury. Features of haemochromatosis, autoimmune hepatitis, viral hepatitis, alpha-1 antitrypsin deficiency and cirrhosis were not identified. The histological findings were those of mixed pattern of chronic hepatitis with cholestasis and mild periportal fibrosis, features consistent with drug-induced liver injury.
Over the next several weeks, the patient’s liver function drastically improved, with the ALT measuring 825 U/L within one week and 416 U/L within one month. Overall, the clinical impression, serological findings and histological features were consistent with drug-induced liver injury likely secondary to Mounjaro.
Discussion
In this case, the R-ratio, used to ascertain the serological pattern of liver injury, was calculated as greater than 5, indicating a serologically hepatocellular pattern of injury, although histopathological findings showed mixed features. The Roussel Uclaf Causality Assessment Method (RUCAM) scale is a well-established tool utilised to qualitatively assess causality in cases of drug-induced liver injury [5]. Here, the RACEM scale was calculated as 6, indicating a probable drug-induced liver injury (DILI) secondary to Mounjaro (Table 1). Also favouring DILI, there was marked improvement in hepatic function tests over the next month, with the serum ALT reducing by 29% from its peak over the next week and 64% within one month. Additionally, other causes of acute liver injury, including autoimmune hepatitis, viral hepatitis, biliary obstruction, alcoholic liver disease and ischaemic injury were excluded.
Table 1. Roussel Uclaf Causality Assessment Method (RUCAM) scoring for evaluation of drug-induced liver injury in the present case (Total score = 6; probable causality)
Roussel Uclaf Causality Assessment Method (RUCAM) scale |
Factor |
Case |
Score |
Time of onset from the beginning of drug use |
Time of onset from cessation of the drug >15 days (slowly metabolised) |
+1 |
Change in ALT levels between peak and stopping drug use |
>50% decrease from peak value within 30 days |
+2 |
Risk factors (Age>55 years, alcohol, pregnancy) |
None |
0 |
Concomitant drug use |
None |
0 |
Exclusion of other causes |
Group 1 and group 2 causes excluded |
+2 |
Previous information on hepatotoxicity |
The drug is not labelled for hepatotoxicity, but published case reports exist |
+1 |
Response to readministration |
Rechallenge not performed |
0 |
Total score |
Probable |
6 |
A literature search identified only three published case reports of Tirzepatide-induced liver injury. Like this case, a delayed onset of toxicity was also noted in other reported cases. In a case reported by Sohal, et al. [6], the patient was of a similar age group and gender and presented 2.5 months after initiating treatment with Tirzepatide [6]. Another case showed liver injury presenting 7 months after onset of treatment [7]. Interestingly, the histological findings noted in the case by Sohal, et al. [6] showed a hepatocellular pattern of injury, with no features of cholestasis, as seen in our case [6]. A third case report of Tirzepatide-induced liver injury by Phox, et al. describes biopsy features of a mixed pattern of injury involving hepatic lobules and portal tracts, with a corresponding R-factor consistent with mixed hepatocellular and cholestatic injury [8].
At present, the mechanism of hepatic injury secondary to Tirzepatide is unclear and is currently only documented in a handful of published case reports. The histological findings showed only focal interface activity with no evidence of a plasma cell- predominant infiltrate, making drug-induced autoimmune hepatitis an unfavourable differential. There was also no evidence of steatosis in this case. Tirzepatide is noted to have a prolonged half-life of approximately 5 days and strong affinity to albumin [9]. A study performed by Martin, et al. assessed drug distribution on rats using [14C]-radiolabeled tirzepatide and found that after reaching peak concentrations, radioactivity declined in most tissues by 336 hours post dose, apart from arterial walls, dose site and intervertebral ligaments, where this figure was 672 hours post dose [10]. These findings highlight the slow metabolism of the drug and may explain the delayed onset of symptoms in the few cases of liver injury that have been documented [11]. Sohal, et al. [6] hypothesized that Tirzepatide-induced liver injury may be secondary to rapid mobilisation of fat from the liver, resulting in elevated liver enzymes, although the lack of steatosis in this case makes this hypothesis questionable.
Conclusion
Overall, drug-induced liver injury, whilst a common adverse effect of many medications, is a phenomenon that remains poorly understood in the context of Mounjaro therapy. Documented cases of Tirzepatide-induced liver injury suggest that these are best categorised as idiosyncratic reactions, which follows an unpredictable course with variable latency of onset. Future complications of Tirzepatide therapy must continue to be documented and consideration may be given to monitoring of liver function during and after therapy.
Authors’ contributions
Radhika Sudhir, MBBS: Manuscript drafting. Manal Atwan, MD: Manuscript drafting and supervision.
Conflicts of interest
The authors of this manuscript have no relationship or financial interests with companies related to the findings of the study. The authors confirm that there are no known conflicts of interests
References
- Sillassen CD, Petersen JJ, Kamp CB, Grand J, Dominguez H, et al. (2025) Adverse effects with tirzepatide: A protocol for a systematic review with meta-analysis and trial sequential analysis. BMJ Open 15: e094947. [Crossref]
- Mishra R, Raj R, Elshimy G, Zapata I, Kannan L, et al. (2023) Adverse events related to tirzepatide. J Endocr Soc 7: bvad016. [Crossref]
- National Institute for Health and Care Excellence (2024) Tirzepatide for managing overweight and obesity. England: National institute for health and care excellence.
- Dahl D, Onishi Y, Norwood P, Huh R, Bray R, et al. (2022) Effect of subcutaneous tirzepatide vs placebo added to titrated insulin glargine on glycemic control in patients with type 2 diabetes: The SURPASS-5 randomized clinical trial. JAMA 327: 534-545. [Crossref]
- Danan G, Teschke R (2015) RUCAM in drug and herb induced liver injury: The update. Int J Mol Sci 17: 14. [Crossref]
- Sohal A, Casanova L, Kowdley KV (2024) A rare case of tirzepatide-induced hepatotoxicity. ACG Case Rep J 11: e01484. [Crossref]
- Abdullah I, El-Ghousain H, Alenezi M (2024) Tirzepatide-related acute liver injury. Eur J Case Rep Intern Med 11: 004813. [Crossref]
- Phox M, Thesing J, Kilgore III WR, Alderson J (2025) Tirzepatide-Induced liver injury: A rare medication side effect. ACG Case Rep J 12: e01661. [Crossref]
- Chavda VP, Ajabiya J, Teli D, Bojarska J, Apostolopoulos V (2022) Tirzepatide, a new era of dual-targeted treatment for diabetes and obesity: A mini-review. Molecules 27: 4315. [Crossref]
- Martin JA, Czeskis B, Urva S, Cassidy KC (2024) Absorption, distribution, metabolism, and excretion of tirzepatide in humans, rats, and monkeys. Eur J Pharm Sci 202: 106895. [Crossref]
- Hosack T, Damry D, Biswas S (2023) Drug-induced liver injury: A comprehensive review. Therap Adv Gastroenterol 16:17562848231163410. [Crossref]





