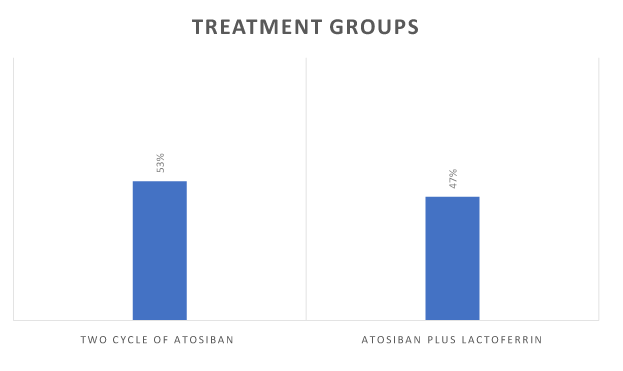Abstract
Twin gestation contributes significantly to perinatal morbidity and mortality related to high occurrence of preterm birth. We evaluated 202 women consecutively selected with twin pregnancies and threatened preterm labor. 98 women were threatened with a single cycle of Atosiban; 34% of them delivered before 34 weeks. The study group consisted of 104 patients submitted to a second cycle of Atosiban because of regular uterine contractions and or cervical length modifications occurred from 48 hours to 7 days and gestational age was prior to 32 week of gestation (group A). After the second cycle of Atosiban, 49 out of 104 patients received a treatment with a vaginal tablet of lactoferrin (group B). After the second cycle of Atosiban, 84% of patients of group A and 90% of patients of group B delivered after 34 weeks. The overall rate of delivery before 34 weeks in the studies groups was of 16%. In our experience, repeated cycles of Atosiban have shown effectiveness in delaying delivery in twin pregnancies. It seems logical to use an oxytocin receptor antagonist as first line drugs in twin pregnancies because of the increased risk of pulmonary edema.
Key words
atosiban, preterm birth, pulmonary edema, twin pregnancies
Introduction
Twin gestation contributes significantly to perinatal morbidity and mortality related to high occurrence of preterm birth. Over the last decade, the number of assisted-reproduction twins have been increasing, doubling the twining rates in Western countries [1]. In addition, twin gestations imposed financial and psychological burdens on the family and society [2].
In Campania, a Southern region of Italy, the birth rate from assisted reproduction increased from 6% in 2007 to 16,5% in 2012.
Overall, the increased risk of maternal and fetal morbidity associated with twin compared with singleton pregnancies is nowadays the strongest argument for single embriotransfer in the debate of assisted reproduction. At least 60% of all twins are born before the 37th week of gestation. In the last seven years twin gestation in our department increased from 3.5% to 7.7% in 2013. In several double blind trials Atosiban has demonstrated a tocolytic effectiveness similar to beta agonists, with the advantages of no side effects due to its specific effect on uterine tissue [3,4].
Moreover, women with multiple pregnancy have a higher risk of pulmonary edema then women with singleton pregnancy. Pulmonary edema occurs in approximately 1/400 women treated with beta-agonists [5]. In twin pregnancies, the levels of urinary aldosterone are increased. This secondary hyperaldosteronism may be further increased by beta-agonists, which rise both aldosterone and renin levels. This condition induces fluids retention and plasma volume expansion, leading to a greater risk of pulmonary edema [5].
Prevention of preterm labor is the goal of long-term tocolysis. Women with recurrent episodes of recurrent preterm labor are at risk for preterm premature rupture of membrane (pPROM), further cervical changes and preterm delivery [6].
Atosiban has been used in Reproductive Medicine Unit of the University of Naples Federico II from 2003 to date, as the first line tocolytic drug in women with threatened preterm delivery in twin pregnancy.
We have previously reported our experience in a small series of twin pregnancies submitted to a second treatment with Atosiban because the presence of symptoms and signs of preterm labor within seven days from the first treatment. This therapeutic approach showed to be relevant for those patients. On the basis of this preliminary experience, we conducted a retrospective cohort study, with the aim to evaluate the effectiveness of repeated Atosiban administration in twin pregnancies over the last ten years in our single centre.
Material and methods
This was a retrospective study of twin pregnancies with 276 consecutive women from April 2003 to December 2013 conducted at the Reproductive Medicine Unit of the University of Naples Federico II. All women with twin pregnancies with threatened preterm labor with or without contractions and reduced cervical length were eligible for the study. Exclusion criteria were maternal age <18 years, fetal death, major fetal defect, severe twin-to-twin transfusion syndrome or selective fetal growth restriction, and cervical length >20mm. On the basis of this criteria 74 patients were excluded.
Diagnosis at admission were: 113 preterm labors, 50 preterm premature ruptures of membranes, 21 incontinent cervices with cerclage, 18 vaginal bleedings (Figure 1). On the total, 172 twin pregnancies derived from reproductive assisted techniques. Preterm labor was defined as ≥4 uterine contractions / 30 min and or cervical length <20 mm with or without uterine contractions. pPROM was proven by a sterile speculum examination (RCOG guidelines N° 44/2006). Atosiban was administered according to the recommended protocol with an initial bolus of 6.75 mg in 1 minute followed by a high rate infusion of 18 mg/hour over 3 hours and a lower concentration of 6 mg/hour over a period of 45 hours.
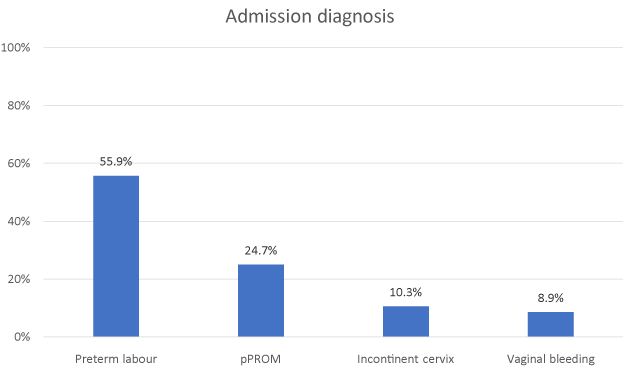
Figure 1. Study population characteristics at admission.
During the first cycle of Atosiban, all women received corticosteroids prophylaxis (antenatal administration of 12 mg of betamethasone, followed by a second dose 24 hours later) to enhance lung maturation. The second cycle of Atosiban was administered when regular uterine contractions and or cervical length modifications occurred from 48 hours to 7 days after the first cycle and when gestational age was prior to 32 weeks of gestation (group A) (Figure 2). Bacterial vaginosis was detected in 22% of women and was treated with antibiotic therapy; erythromycin (250 mg orally 6 hourly) was given for 10 days following the diagnosis of pPROM in all patients. After the second cycle of atosiban, 49 out 104 patients received a treatment with a vaginal tablet of lactoferrin (Lf) (300 mg) for 21 days (group B) (Figure 3).
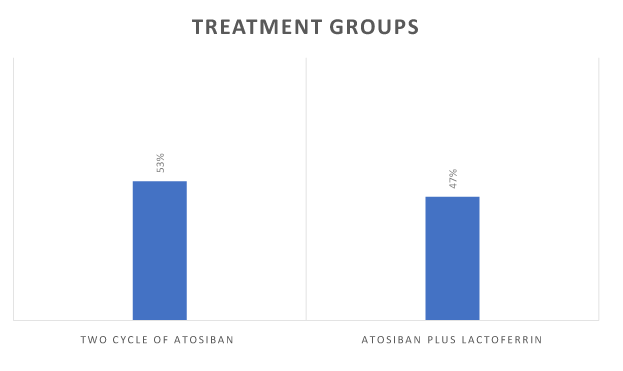
Figure 2. Two studies groups.
98 pregnant women received a single dose of Atosiban just to perform the corticosteroids prophilaxis in 48 hours. This group of patients (the control group) refers to patients evaluated before our preliminary reported experience.
Statistical analysis was performed using GRAPH Pad Prism 4.0 software (Graph-pad Software Inc, La Jolla, CA). The distribution of variables was compared with Student’s Test for paired data. Differences associated with p values lower than 0.05 were considered statistically significant. The results are reported as mean +/- standard deviation.
Results
After 48 hours from the end of the first cycle of Atosiban, 98 women didn’t show uterine contractions and/or cervical length modifications; 34% of them delivered before 34 weeks.
While 104 women had regular uterine contractions and/or cervical length modifications (group A). 22% of those patients had recurrent symptoms of preterm labor from 48 hours, whereas the remaining 78% showed symptoms within 7 days (Figure 3). After the second cycle, 84% of patients of group A and 90% of patients of group B delivered after 34 weeks (Figure 4). No difference was detected between spontaneous twin pregnancies and assisted reproduction ones.
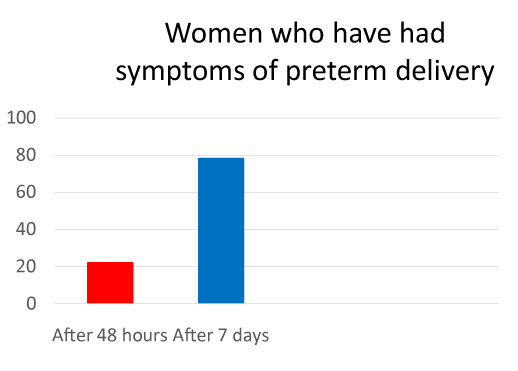
Figure 3. Percentage of patients that respectively showed symptoms of preterm delivery after the first treatment with atosiban.
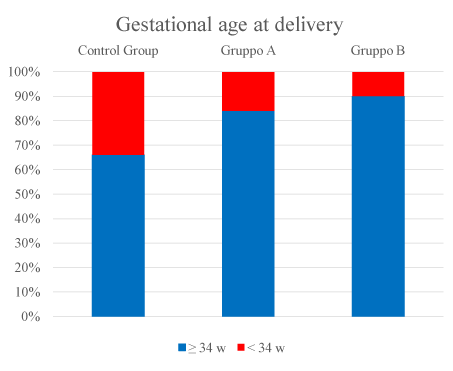
Figure 4. Percentage of patients who delivered <34 weeks in the study groups.
The overall mean numbe2021 Copyright OAT. All rights reservocolysis was 38 in the group of preterm labor, whereas in the subgroup of women with pPROM the interval until delivery was 19 days; in the group B the days gained were 52. After initial bolus administration, only 5% of women presented drug related side effects such as flushing for 1-5 minutes. During infusion, 3% of women experienced side effects possibly related to Atosiban such as nausea and vertigo. The overall rate of delivery before 34 weeks in the studies groups was of 16%.
Discussion
In this retrospective study repeated Atosiban administration resulted effective in prolonging twin pregnancies. In multiple pregnancies, over distension of the uterus with stretching of fetal membranes and or cervical ripening is a more likely mechanism for its release than infection or inflammation.
The uterine over distension in twin pregnancies may cause a relative decidua ischemia with release of lysosomal enzymes. These enzymes include several phospholipases which initiate the prostaglandins cascade by releasing arachidonic acid, the critical substrate for their synthesis. The increased release of prostaglandins affects collagen integrity altering the consistence and distensibility of the cervix [7].
There is an inverse relationship between cervical length and the likelihood ratio for preterm birth. Atosiban acts by blocking oxytocin receptors. Oxytocin seems to initiate myometrial contractility by both a direct and an indirect mechanism. It directly interacts with membrane receptors leading to an increase in intracellular calcium. The indirect effect of oxytocin is the stimulation of prostaglandins release from decidua and fetal membranes; the prostaglandins are strictly involved in cervical ripening [8].
Recently a Cochrane Database Systematic Review on oxytocin receptor antagonists for inhibiting preterm labor underlines the importance of well studies about different tocolytic strategies at different gestational ages in order to optimize safety and efficacy [9].
Furthermore, Atosiban determined fewer maternal side effects in comparison with other tocolytics [9]. As we previously demonstrated in a selected group of pregnant from ART, the use of oxytocin receptor antagonist in the mid second trimester would appear to be an useful tool to delay preterm labour when prostaglandins release activates oxytocin receptors [3].
Multiple pregnancies are also associated with a major maternal plasma volume expansion and secondary hyperaldosteronism [5]. This conditions increase the risk of pulmonary edema, particularly when tocolytic therapy with beta agonists combined with fluid therapy is administered. Beta mimetics increase both aldosterone and renin levels in twin pregnancies and may potentiate the risk of pulmonary edema [5,10].
Moreover, same studies revealed that Atosiban is cost-saving versus beta mimetics, due to its superior safety profile [4,11]. In our experience, repeated cycles of Atosiban have shown effectiveness in delaying the interval to delivery, in twin pregnancies. The rate of preterm birth < 34week of gestation with single cycle of Atosiban was 34% (n=33) compared with the overall rate of the studies groups of 16%. It seems logical to use an oxytocin receptor antagonist as first line drugs in twin pregnancies because of the increased risk of pulmonary edema.
In addition, the use of vaginal lactoferrin (Lf), an approximately 80-KDa iron-binding glycoprotein of the transferrin family with bacteriostatic and bactericidal properties, was usefull in the maintenance therapy, in our experience. Several studies [8,12,13] suggest that Lf plays an important role against cervicovaginal inflammation and or infection by decreasing levels of interleukin (IL)-6 and increasing cervical length. Both drugs, Atosiban and Lf, seem able in preventing preterm delivery caused by cervical ripening, blocking prostaglandins release.
Twin pregnancies are at an increased risk of stillbirth, antenatal morbidity and infant death. This is largely due to preterm delivery and complications of prematurity and low birth weight are the primary risk factors for infant mortality. The NICU admission rate was lowest following delivery at 38 weeks’ gestation, and the NICU admission rate decreased significantly with each additional week of pregnancy from 32 through 38 weeks’ gestational age. We found an increased risk of mortality associated with delivery at 34 weeks’ [14-16]. We hope that these data provide information to assist providers for the management of threatened preterm labor in twin pregnancies.
Based on this consideration and the safety profile of Atosiban, two cycles are better than one in symptomatic patients before 32 weeks of gestation, in our experience, even if corticosteroid prophylaxis is already performed This management is used to reduce perinatal morbidity/mortality due to extremely prematurity in those twin pregnancies with no additional complications that may indicate earlier delivery. In addition maintenance therapy using vaginal Lf improve gestational age delivery as showed in the study group B.
Declaration of interest
The authors report no declarations of interest
References
- Ananth CV, Chauhan SP (2012) Epidemiology of twinning in developed countries. Semin Perinatol 36: 156-161. [Crossref]
- Luke B, Brown MB, Alexandre PK, Kinoshi T, O'Sullivan MJ, et al. (2005) The cost of twin pregnancy: maternal and neonatal factors. Am J Obstet Gynecol 192: 909-915. [Crossref]
- Locci M, Nazzaro G, Merenda A, Pisaturo ML, Laviscio P, et al. (2006) Atobisan versus ritodrine used prophylactically in ICSI pregnancy to prevent pre-term birth in women identified as being at high risk on the basis of transvaginal ultrasound scan. J Obstet Gynecol 26: 396-401.
- Shim JY, Park YW, Yoon BH, Cho YK, Yang JH, et al. (2006) Multicentre, parallel group, randomized, single blind study of the safety and efficacy of atosiban versus ritidrine in the treatment of acute preterm labour in Korean women. BJOG 113: 1228-1234. [Crossref]
- Lamont RF (2000) The pathophysiology of pulmonary oedema with the use of beta-agonists. BJOG 107: 439-444. [Crossref]
- Wu MY, Chen SU, Lee CN, Ho HN, Yang YS (2010) Use of atosiban in a twin pregnancy with extremely preterm premature rupture in the membrane of one twin: a case report and literature review. Taiwan J Obstet Gynecol 49: 495-499. [Crossref]
- Romero R, Espinoza J, Kusanovic JP, Gotsch F, Hassan S, et al. (2008) The preterm parturition syndrome. BJOG 113: 17-42.
- Locci M, Nazzaro G, Miranda M, Salzano E, Montagnani S, et al. (2013) Vaginal lactoferrin in asymptomatic patients at low risk for pre-term labour for shortened cervix: cervical length and interleukin-6 changes. J Obstet Gynaecol 33: 144-148. [Crossref]
- Flenady V, Reinebrant HE, Liley HG, Tambimuttu EG, Papatsonis DN (2014) Oxytocin receptor antagonists for inhibiting preterm labour. Cochrane Database Syst Rev: CD004452. [Crossref]
- Dudenhausen J (2006) 'Normal' pregnancy with adverse events on initial tocolytic treatment. BJOG 113 Suppl 3: 116-117. [Crossref]
- Wex J, Abou-Setta AM, Clerici G, Di Renzo GC (2011) Atosiban versus betamimetics in the treatment of preterm labour in Italy: clinical and economic importance of side-effects. Eur J Obstet Gynecol Reprod Biol 157: 128-135. [Crossref]
- Giunta G, Giuffrida L, Mangano K, Fagone P, Cianci A (2012) Influence of lactoferrin in preventing preterm delivery: a pilot study. Mol Med Rep 5: 162-166. [Crossref]
- Poppiti R, Locci M, Nazzaro G (2009) Prevention of preterm delivery in twin pregnancies with atosiban. J Matern Fetal Neonatal Med 92: 61.
- Page JM, Pilliod RA, Snowden JM, Caughey AB (2015) The risk of stillbirth and infant death by each additional week of expectant management in twin pregnancies. Am J Obstet Gynecol 212: 630. [Crossref]
- Conde-Agudelo A, Romero R (2014) Prediction of preterm birth in twin gestations using biophysical and biochemical tests. Am J Obstet Gynecol 211: 583-595. [Crossref]
- Liem SM, van de Mheen L, Bekedam DJ, van Pampus MG, Opmeer BC, et al. (2013) Cervical length measurement of the prediction of pre-term birth in symptomatic women with a twin pregnancy: a systematic review and metanalysis. Obstet Gynecol Int: 125897. [Crossref]