Aim of the study: The aim of the study is to analyze correlation between level of troponin T in fetal blood with NIHF and the course of pregnancy.
Material and method: The analysis of troponin T level in fetuses blood was conducted in 22 cases of cordocentesis carried out at the Department of Gynecology, Fertility and Fetal Therapy between 2014-2016. There were 15 cordocentesis performed in cases of NIHF undergoing intrauterine therapy and 7 cordocentesis performed in cases of other pathology that have been assigned to the control group.
Results: In the study group there were 9 results obtained over a standard 0.308 ng/ml and 6 results below this value. In the control group 5 results were normal and 2 were above norm. In the group of fetuses with NIHF, 89% of cases with elevated levels of troponin T were unsuccessful, while in the group with normal levels 83% of the cases had a successful course. In the control group 100% of normal levels of troponin T had a successful course, and there were 2 cases of elevated levels of troponin T in 1 newborn rated at 3 points in the Apgar score because of respiratory disorders.
Conclusions: Based on the analysis, it appears that the level of troponin T in fetal blood can be a marker providing information on the prognosis and, therefore, the level of cTnT in fetal blood should be measured to determine the degree of heart failure in conjunction with the echocardiography study.
non-immune hydrops fetalis, intrauterine therapy, cordocentesis, troponin T, NIHF
Non-immune fetal edema is a condition in which fluid accumulates in at least two areas of the body, including pleural cavity (hydrothotax), pericardial cavity, peritoneal cavity, and subcutaneous tissue, often accompanied by polyhydramnios and the edema bearing [1,2]. Isolated ascites and isolated hydrothorax is usually considered as part of non-immune hydrops fetalis, wherein the severity of edema is minimal, therefore ascites or hydrothorax is the dominant clinical manifestation [3-6].
Non-immune hydrops fetalis was a frequently occurring pathology before the introduction of the prevention of the serological conflict in the 1970s when the swelling was caused mainly by erythroblastosis being a result of alloimmunisation Rh. Currently, over 90% of cases of fetal edema is nonimmunologic hydrops. The incidence of NIHF is 1:2500-3500 newborns and 1:1600-7000 fetuses [4-6], but the precise incidence of edema is difficult to determine because many cases are not detected prior to intrauterine fetal death, and in the course of some pregnancies swelling may disappear by itself in the period of gestation.
The causes of edema are numerous, their number is estimated at 120. These are all the pathologies affecting vascular function disorder in the fetus, disrupting the tissue-fluid in the fetus.
The causes of edema was systematized by Bellini et al. [2] after analyzing more than 6,000 cases of non-immunological fetal edema , and in 2015 NIHF causes were systemized once again, also taking into account the study from 2009 [7]. These causes are divided into the following groups:
- Cardiovascular causes (23,3%)
- Hematologic causes (9,0%)
- Chromosomal causes (9,3%)
- Defect syndrome (5,5%)
- Lymphatic dysplasias (15%)
- Congenital metabolic syndromes (1,3%)
- Infections (7,0%)
- Irregularities in the precinct of the chest (2,3%)
- Urinary tract defects (0,9%)
- Tumour coating (0,7%)
- TTTS and TRAP (4,1%)
- Defects in the gastrointestinal tract (1.3%)
- Other unclassified causes (3,6%)
- idiopathic (20,1%)
Analysis of more than 6,000 cases of nonimmune hydrops fetalis was enabled by Bellini et al. ([2,7] to distinguish the three most common causes. Cardiovascular (23.3%), idiopathic (20.1%) causes and genetic disorders (9.3%) are the most common vehicles leading to edema. In northern China, however, the most common NIHF cause is an alpha-talesemia, representing more than 50% of the causes of edema [8].
Intrauterine therapy in the case of NIHF includes: cordocentesis with blood transfusion and/or administration of albumin, decompression of body cavities, shunt implantation between the cavities of the body and the amnion, amnioreduction. Intrauterine therapy in NIHF can be implemented in case of exclusion of severe pathology. Differential diagnosis should focus primarily on a detailed ultrasound examination with cardiovascular assessment, infectious screening and fetal karyotype assessment. Due to the fact that the most common cause of NIHF are cardiovascular causes diagnosis of fetal echocardiography is necessary and evaluation of cardiovascular flows in peripheral vessels in the Doppler examination. Cordocentesis is one of the elements of intrauterine fetal NIHF therapy. Cordocentesis is carried out in order to determine fetal blood count, the albumin level as well as karyotype, blood gas analysis, virological and biochemical screening and possibly the assessment of the pressure in the umbilical vein. Blood transfusion is carried out in case of anemia in the fetus. Anemia can be determined on the basis of blood counts, as well as on the basis of the flows in the middle cerebral artery - the risk of anemia is higher in fetuses in whom MCA PSV (cm/s) is 1.5 times higher than the median. Blood can be administered to the umbilical vein or hepatic vein. Albumin is administered to increase oncotic pressure, which leads to a fetal hydrops reduction .Currently, 10% of human albumin solution is administered in an amount adjusted for gestational age .
In the literature there are reports on the determination of different kinds of NIHF markers to determine the etiology of this pathology or determine the prognostic factor for fetuses with edema. Troponin T is one such marker. Troponin T is a protein regulating the contraction of striated muscle and cardiac muscle, and the determination of tropinin protein isoforms is primarily used in the diagnosis of myocardial infarction in adults. TnC increase in the value above the 99. percentile of the reference control group (above the upper limit of the reference range) in combination with acute ischemia symptoms permits myocardial infarction diagnosis. CTnT is the most sensitive biochemical marker of myocardial damage, and increasing its value may indicate worse outcomes. The range of cTnT standards in fetuses differs from the level for adults and newborns. The level of troponin T in fetuses was described by Fortunato et al. [9] using the immune electrochemiluminescence method (Elecsys 1010/2010 analyzer system, Roche, Mannheim, Germany) at a temperature of 20-25 degrees centigrade. Fortunato et al., established the standard for the level of troponin T in fetal blood based on the examination 109 healthy fetuses with a maximum of 0,308 ng/ml [9].
The aim of the study is to analyze the correlation between the level of troponin T in fetal blood in cases of NIHF and the course of pregnancy and the postnatal newborn state.
The Department of Gynecology, Fertility and Fetal Therapy at the Institute of Polish Mothers Memorial Hospital Centre in Lodz has, since 2014, been conducting a study involving the examination of troponin T levels in fetuses blood obtained from cordocentesis. Until June 2016 the troponins T level was examined in the course of all cordocentesis performed in the Department of Gynecology, Fertility and Fetal Therapy.
There were 15 cordocentesis performed in NIHF diagnosed cases undergoing intrauterine therapy in the Department of Gynecology, Fertility and Fetal Therapy and 7 cordocentesis performed in cases of other pathology that have been assigned to the control group. In control group it was 4 cases of sérology conflict, 2 cases of uropathy and 1 case of heart arythmia.
Cordocentesis was carried out in pregnant women from 22 to 33 weeks of pregnancy. The level of troponin T was determined at the Center for Medical Laboratory Diagnostics ICZMP using Troponin T hs STAT Troponin T hs STAT CalSet, Procell Elecsys, CleanCell Elecsys, Assay CUP, Assay TIP, Sys Wash reagents.
The results obtained in pg/ml units were converted into ng/ml. The Apgar score, birth weight infants, the week of termination of pregnancy and newborn survival were taken into account in the statistical analysis.
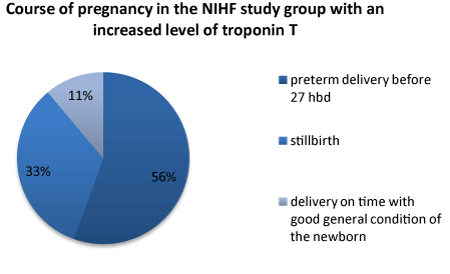
In the study group 9 results were obtained over the value of 0.308 ng/ml and 6 results were below this value. In the control group 5 results were normal, and 2 were above normal (Figure 1-6). In the NIHF group cases with increased value of troponin T were concluded as follows: 3 cases of intrauterine fetal death, 5 cases of preterm birth before 27 hbd, and 1 case of delivery on time with good general condition of the newborn. None of the infants born before 27 hbd survived 3 days. 89% of this group thus ended unsuccessfully.
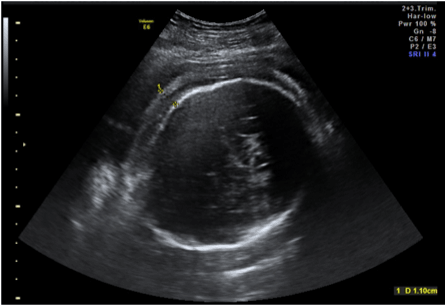
Figure 1. Hydrops of fetal head in case of NIHF.
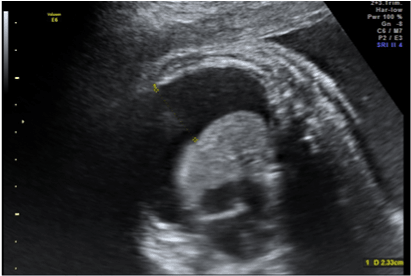
Figure 2. Fluid in fetal pleural cavity.
Figure 3. Cordocentesis.
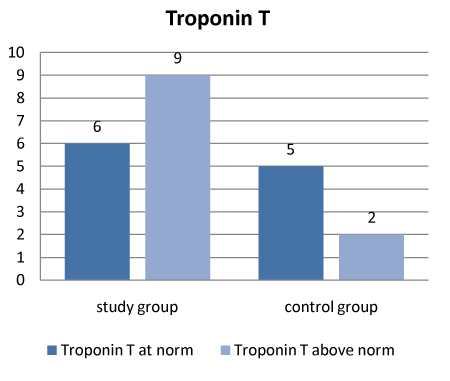
Figure 4. The level of troponin T in the test group and control group.

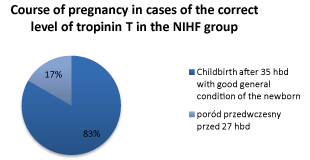
Figure 6. The course of pregnancy in cases of normal levels of troponin T in the study group.
In 6 cases in the group NIHF, where the level of troponin T was normal, 5 ended up giving birth after 35 weeks of pregnancy and a good general state of newborns, and 1 case ended with a premature birth before 27 hbd. The mean birth weight of newborns in a group where troponin T was normal was 2875 g, and the median Apgar score was 5. 83% cases had therefore a successful course. In the control group in 5 cases of normal levels of troponin T, the status of newborns was good (average body weight was 3100 g, and the median was Apgar 7), and in 2 cases of elevated troponin T 1 case ended with the delivery on time and good general condition of the newborn, and in 1 case of newborn received a rating of 3 on a Apgar scale because of respiratory distress. In cases of normal levels of troponin T, 100% of the cases had successful course.
Summing up all the 11 cases of elevated troponin T(control group + study group), in 9 of them pregnancy was unsuccessful (81%) and in all 11 cases where the level of troponin T was normal in 91% of them (10 cases), the course of pregnancy was successful. The results point to the fact that troponin T may be a good marker of pregnancy and postnatal development of newborn state, which could reflected in a possible course of therapy and intrauterine fetal condition monitoring.
Nonimmunologic Fetal edema is frequently associated with abnormal flow in the central cerebral artery and fetal anemia, however, anemia may be the result of heart failure. In cases where the ultrasound image with the Doppler study confirms the development of fetal anemia, fetal transfusions are carried out for packed red cells or albumin. Determination of the morphology of the fetus must be done with the first sample of blood taken during cordocentesis, which is an integral element of intrauterine transfusion/use of pop-analyzer which are to take place/.
Indication for the fetal blood transfusion:
- hemoglobin level lower by 3 g% from the average predicted value for a given gestational age.
- hematocrit level below 25%, regardless of the gestational age.
NKKCz is the blood used for this perinatal - it being a radiated formulation of red blood cells. It is a concentrate of blood group 0 Rh minus with a ray dose of 25 Gy. In case of perinatal transfusion, the expiry date for the formulation is 24 hours. The hemoglobin rate in NKKCZ is about 25 g/dl (10). The volume of packed red blood
cells for transfusion may be calculated by the following formula:
Vtransfused [ml] = [Vfetal-placental x (Htfinal – Ht initial)]/Ht transfused blood
The total blood volume of the fetus and placenta calculated by multiplying the approximate fetal weight calculated in ultrasound by 0.14 ml/g [11].
Currently, the amount needed for red blood cell mass can be calculated in a computer program, using the above described algorithm. Re-transfusion is planned for the moment which is expected to obtain hematocrit values between 20 and 25%. This term can be approximately determined assuming a fall in hematocrit by 1% per day. In cases of very severe form of anemia during the pause between the first and second treatment, most of the fetal erythrocytes begins to break apart. For this reason, the first break is from 7 to 14 days, while the spacing between the following transfusions is between 21 to 28 days [12].
Although intrauterine blood transfusions are able to improve fetal anemia, an accurate assessment of heart failure is indicated as this may be an important prognostic factor. The relationship between the ultrasound markers of heart failure and fetal biochemical indicators has been examined by Mäkikallio et al. [13]. He found that fetuses with elevated cTnT are able to maintain the systolic function of the heart. Authors suggest that the function of the fetal heart muscle does not change even under conditions of severe myocardial damage. However, in cases of anemia in the fetus and the resulting edema of these conditions can be significantly changed.
Renal infarction may be assessed indirectly by the Doppler venous and atrioventricular flow. Hofstaetter et al. [14] studied the cardiovascular system of the fetus in the supervision of edema. They came to the conclusion that the best predictor of fetal Doppler is the ductus venosus. If the value of the A wave in the ductus venosus dropped to zero it was a negative predictor for the fetus.
The opportunity to study the fetal heart using fetal cTnT levels could potentially improve the prognosis. Therefore, in addition to determining the value of hemoglobin in the blood of the fetus, cordocentesis may also provide useful information on fetal heart rate by analyzing fetal concentrations of cTnT.
On the basis of this analysis, it appears that the level of troponin T in fetal blood can be a marker giving information on prognosis and, therefore, the level of cTnT in fetal blood should be measured to determine the degree of heart failure in conjunction with echocardiography. Further studies are needed to confirm the usefulness of troponin T research in predicting the prognosis for the fetus after intrauterine interventions.
- Potter EL (1943) Universal edema of the fetus unassociated with erythroblastosis. Am J Obstet Gynecol 46: 130.
- Bellini C, Hennekam RC, Bonioli E (2009) A diagnostic flow chart for non-immune hydrops fetalis. Am J Med Genet A 149A: 852-853. [Crossref]
- Kaczmarek P. The importance of fetal echocardiography in non-immunological edema. Work on the degree of doctor of medical sciences
- Im SS, Rizos N, Joutsi P, Shime J, Benzie RJ (1984) Nonimmunologic hydrops fetalis. Am J Obstet Gynecol 148: 566-569. [Crossref]
- Kaczmarek P, Jaczewski B, Oszukowski P, Respondek-Liberska M (2003) Nonimmunologic fetal-predictors based on an analysis of 230 cases with regard to fetal echocardiography. Ginekol Pol 74: 1112-1117
- Rodeck M, Whittle MJ (2002) Fetal medicine: basic science and clinical practice. Churchil Livingstone Ltd, 225-247.
- Bellini C, Donarini G, Paladini D, Calevo MG, Bellini T, et al. (2015) Etiology of non-immune hydrops fetalis: An update.
Am J Med Genet A 167A: 1082-1088. [Crossref]
- Liao C, Wei J, Li Q, Li J, Li L, et al. (2007) Nonimmune hydrops fetalis diagnosed during the second half of pregnancy in Southern China. Fetal Diagn Ther 22: 302-305.
[Crossref]
- Fortunato G, Carandente Giarrusso P, Martinelli P, Sglavo G, Vassallo M, et al. (2006) Cardiac troponin T and amino-terminal pro-natriuretic peptide concentrations in fetuses in the second trimester and in healthy neonates. Clin Chem Lab Med 44: 834-836. [Crossref]
- Baranowski W (2009) Physiology and biochemistry of blood.
- Guidelines for the treatment of blood and its components and blood products in medical entities. Second edition. Warsaw Wydanie II. Warszawa .
- Guidelines for the American College of Obstetricians and Gynecologists. Proceedings in the maternal-fetal serological conflict. Educational Bulletin.
- Mäkikallio K, Vuolteenaho O, Jouppila P, Räsänen J (2002) Ultrasonographic and biochemical markers of human fetal cardiac dysfunction in placental insufficiency. Circulation 105: 2058-2063. [Crossref]
- Hofstaetter C, Hansmann M, Eik-Nes SH, Huhta JC, Luther SL (2006) A cardiovascular profile score in the surveillance of fetal hydrops. J Matern Fetal Neonatal Med 19: 407-413. [Crossref]