It was Claude Bernard, a French physician and physiologist, who first correlated pain with sympathetic nervous system in the mid-1800’s [1]. The first detailed descriptions of sympathetically-mediated pain came from one of his students, Silas Weir-Mitchell, in his book “Gunshot Wounds and other Injuries of Nerves”, published in 1864 [2]. During the American Civil War, Mitchell noted that some soldiers developed a particularly painful state after bullet injuries. In his reports, he described soldiers who experienced burning pain that never affected the trunk, rarely affected the thighs and arms, but often involved the legs and forearms, and mainly involved the hands and feet. The patients described burning pain, which became intensely hyperalgesic. At some point, they avoided even contact with the air, which now we know as allodynia. As time went by, the patient’s face showed evidence of suffering, they developed sleep disturbances, and finally, they became hysterical. A few years later, Mitchell coined the term “causalgia” (from Greek kausos fever + algia pain) to describe this condition [3].
Since these early reports, many terms such as Sudeck’s atrophy, sympathalgia and reflex sympathetic dystrophy have been used to describe this clinical scenario. In 1994, the Committee of Classification of Chronic Pain of the International Association for the Study of Pain (IASP) coined the term “Complex Regional Pain Syndrome” (CRPS) as a definitive nomenclature, with its typical subtypes: CRPS type I (where there is no obvious nerve damage) and CRPS type II (where there is identifiable nerve damage).
Symptoms of CRPS-I (which used to be called reflex sympathetic dystrophy) include “burning” pain (a term that usually used to describe superficial structures) and “aching” pain (describing pain in the deep tissues) as well as a variety of stimulus-evoked pain sensations, including hyperalgesia and allodynia (with mechanical, cold, and sometimes heat sensitivity) [4]. Other symptoms include vasomotor and sudomotor dysregulation; trophic changes in the skin, hair, nails, and bone; and dystonia or other motor abnormalities [4,5]. The clinical presentation of CRPS-II (which used to be called causalgia) is the same in all respects, except that a verifiable nerve injury is present.
Coderre and Bennett [4] have developed an animal model of chronic post-ischemia pain that creates CRPS-I-like symptoms. These finding, coupled with recent histological [6] and animal [7] data, suggest that some form of initial nerve trauma is “an important trigger for the cascade of events leading to CRPS” [8]. Therefore, the distinction between the pathogenesis of CRPS-I (“non-nerve”) and that of CRPS-II (“defined nerve”) may be a matter of degree and not of mechanism [9].
The first set of CRPS clinical criteria that was developed is known as the Orlando or IASP criteria, and its validation had been based on experience from other syndrome-defined diagnoses such as headaches and psychiatric disorders [10]. The original diagnostic criteria are described below (Table 1). Later, concerned by the low specificity of IASP/Orlando criteria [11], researchers proposed the Budapest criteria, which had been divided in the clinical criteria (Table 2) and the research criteria (Table 3) [12,13]. The only difference between the two criteria is the requirement of at least one symptom in three of the four categories in the clinical criteria and at least one symptom in all of the four categories in the research criteria. The primary aim of the research criteria is to guarantee a high specificity rate (Table 4), avoiding the recruitment of other neuropathic pain conditions on research scenario.
Table 1. IASP criteria for Complex Regional Pain Syndrome
1. |
The presence of an initiating noxious event or a cause of immobilization. |
2. |
Continuing pain, allodynia, or hyperalgesia with which the pain is disproportionate to any inciting event. |
3. |
Evidence at some time of edema, changes in skin blood flow, or abnormal sudomotor activity in the region of pain. |
4. |
This diagnosis is excluded by the existence of conditions that would otherwise account for the degree of pain and dysfunction. |
Table 2. Budapest clinical criteria for Complex Regional Pain Syndrome
1. |
Continuing pain, which is disproportionate to any inciting event. |
2. |
Must report at least one symptom in three of the four following categories:
Sensory: Reports of hyperalgesia and/or allodynia
Vasomotor: Reports of temperature asymmetry and/or skin color changes and/or skin color asymmetry
Sudomotor/Edema: Reports of edema and/or sweating changes and/or sweating asymmetry
Motor/Trophic: Reports of decreased range of motion and/or motor dysfunction (weakness, tremor, dystonia) and/or trophic changes (hair, nail, skin) |
3. |
Must display at least one sign* at time of evaluation in two or more of the following categories:
Sensory: Evidence of hyperalgesia (to pinprick) and/or allodynia (to light touch and/or deep somatic pressure and/or joint movement)
Vasomotor: Evidence of temperature asymmetry and/or skin color changes and/or asymmetry
Sudomotor/Edema: Evidence of edema and/or sweating changes and/or sweating asymmetry
Motor/Trophic: Evidence of decreased range of motion and/or motor dysfunction (weakness, tremor, dystonia) and/or trophic changes (hair, nail, skin) |
4. |
There is no other diagnosis that better explains the signs and symptoms. |
* A sign is counted only if it is observed at time of diagnosis.
Table 3. Budapest research criteria for Complex Regional Pain Syndrome.
1. |
Continuing pain, which is disproportionate to any inciting event. |
2. |
Must report at least one symptom in each of the four following categories:
Sensory: Reports of hyperalgesia and/or allodynia
Vasomotor: Reports of temperature asymmetry and/or skin color changes and/or skin color asymmetry
Sudomotor/Edema: Reports of edema and/or sweating changes and/or sweating asymmetry
Motor/Trophic: Reports of decreased range of motion and/or motor dysfunction (weakness, tremor, dystonia) and/or trophic changes (hair, nail, skin) |
3. |
Must display at least one sign* at time of evaluation in two or more of the following categories:
Sensory: Evidence of hyperalgesia (to pinprick) and/or allodynia (to light touch and/or deep somatic pressure and/or joint movement)
Vasomotor: Evidence of temperature asymmetry and/or skin color changes and/or asymmetry
Sudomotor/Edema: Evidence of edema and/or sweating changes and/or sweating asymmetry
Motor/Trophic: Evidence of decreased range of motion and/or motor dysfunction (weakness, tremor, dystonia) and/or trophic changes (hair, nail, skin) |
4. |
There is no other diagnosis that better explains the signs and symptoms. |
* A sign is counted only if it is observed at time of diagnosis.
Table 4. Sensitivity and specificity of clinical and research Budapest criteria.
Criteria type |
Sensibility |
Specificity |
Clinical |
0.85 |
0.69 |
Research |
0.69 |
0.96 |
The incidence and prevalence of CRPS varies widely, depending on the diagnostic criteria and the population studied. In a Dutch population, de Mos et al. [14] found an incidence of 26.2 cases/100,000 population/year when the diagnosis was made by non-specialists, and 19.5/100,000/year when specialists were in charge. This is more than four times higher than reported by Sandroni et al. [15] in Olmsted County, USA, who adopted the IASP criteria as primary endpoint and found a 5.46/100.000/year incidence of CRPS. The mean age in the Dutch study was 52.7 ± 2.20 years, with more cases identified in upper extremity (59.2%) than lower extremity (39.1%). Females were more affected than males on a 3.4:1 proportion ratio. The main drawback of this retrospective epidemiological study is that there were no definitive diagnostic criteria established [14]. Regarding the precipitating event, fractures were the most common (44.1%), followed by strains (17.6%), elective surgery (12.2%), tendon injury (5.5%) and others (8.8%). In 1.7% of cases, no precipitation factor was identified [14]. Jellad et al. [16] found an incidence of CRPS as high as 32.2% after wrist fractures, with a mean delay of 21.7 ± 23.7 days after cast removal before the onset of symptoms, with the incidence increased in women who described severe pain and inference with activities of daily living.
There are several studies that tried to identify patients at risk for developing CRPS. Geertzen et al. [17] in a case-control study, found that 79.2% of patients with CRPS were able to relate a major stressful event at least 3 months prior to the disease versus 21.4% of the control group. Dilek et al. [18] also showed that high anxiety was correlated with a higher incidence of CRPS after distal radial fracture. However, others have failed to link anxiety and stress to CRPS [19,20].
It is well accepted that high levels of pain in the early phases of trauma is strongly related to higher incidence of CRPS, suggesting a central sensitization as a background for the disease [16,21,22]. Female gender can be considered a risk factor for CRPS [15,18-20,23-26] and, since there is a higher incidence in older patients, the postmenopausal status is also related higher incidence of CRPS [14-16,21], although this association could be an epiphenomena linked to higher incidence of fractures and others traumas in this population. However, the common factor in many of these risks may be nerve entrapment.
The nerve has its own blood flow (vaso neurvorum) as well as a variety of accompanying vascular structures. An entrapment neuropathy is defined as a pressure-induced segmental injury to a peripheral nerve from an anatomic or pathologic process [27]. Entrapments of a nerve can occur anywhere along the path of that nerve, but they primarily occur at anatomic locations within fibro-osseous tunnels (such as seen in the carpal tunnel) or when the nerve passes through a fascial opening [28]. Increased pressure on the nerve can occur from trauma, compression, stretching, or edema. Within even minutes of the compression, there is a decreased intraneural blood flow, with inhibition of axonal transportation of nutrients, and an increase in intraneural edema [29]. Some people may be particularly susceptible to certain entrapment neuropathies because of a congenital narrowing of a tunnel or because of thickening of a fascia or an aberrant location of a fascial structure. Some patients with a systemic disorder such as diabetes mellitus (DM) show entrapment signs and symptoms much more frequently than non-diabetics [30,31], presumably because of the already compromised nerve blood flow.
Graded experimental compression results in profound short- and long-term effects on in vivo blood flow. Mild compression (20-30 mm Hg) decreases venous flow; moderate compression decreases capillary and arterial flow; and pressures of 60-80 mmHg cause frank ischemia [32]. These pressures correspond to those that have been measured clinically in the tarsal tunnel [33], carpal tunnel [34] and cubital tunnel [35]. Axonal transport of nutrients is blocked by pressures >50 mm Hg [36], and nerve impulse conduction is blocked after less than an hour of compression >70 mm Hg [37].
There are therefore clinically several stages of entrapment [29]. Stage I patients feel pain and paresthesias intermittantly, increased at night (presumably because daytime activity promotes microvascular perfusion). Stage II patients have persistent paraesthesias, numbness, burning pain, and occationally muscle weakness. Prolonged compression leads to neural edema, which can progress to epineural fibrosis and scarring, which further thickens the nerve and worsens the entrapment. Damage to the myelin sheath and subsequent axonal disruption (Stage III) are the end stages of chronic compression, resulting in irreversible damage [38].
It is important to note that there may be more than one site of entrapment. Upton and McComas observed that 81/115 (70%) patients with carpal or cubital tunnel syndrome also had electrophysiological evidence of a nerve injury in the neck [39]. They named this phenomenon the “double crush syndrome” (DCS), postulating that the presence of a more proximal lesion renders the distal nerve trunk particularly vulnerable to compression, with a degree of pain and dysfunction greater than that expected from either entrapment alone. DCS appears to be due to the effect of compression on anterograde axoplasmic flow, decreasing blood flow and nutrients “downstream”, which would put the distal section of the nerve at greater risk of ischemia [40].
Stretch injury may also have a significant role in the pain after joint injuries, such as sprains or DJD. As an example, inversion injuries of the ankle (lateral ankle sprains) can cause injury to the superficial peroneal nerve, resulting in persistent pain and CRPS symptoms [41]. Pain from degenerative joint disease (DJD) may not be purely due to intra-articular pathology therapies [42], as evidenced by the knee pain relief seen with injection and denervation of the infrapatellar saphenous nerve [43,44]. As little a 6% stretch of a nerve can cause permanent injury [45].
As described above, the animal model findings of Coderre and Bennett have lead them to propose that the fundamental cause of pain in CRPS is a persistent deep tissue (muscle, bone, and/or nerve) ischemia and subsequent inflammatory reaction [4]. They note that CRPS-I nearly always follows injury to the deep tissues (crush injuries, sprains, fractures), which results in nerve injury and entrapment. This injury evokes an up-regulation of alpha-adrenergic receptors on the arterial smooth muscle cells, leading to a 3-fold increase in vasoconstriction response from norepinephrine [46]. Coderre and Bennett [4] also hypothesized that the allodynia and hyperalgia seen in CRPS patients is due to central sensitization, which would explain the fluctuating skin sensitivity seen in CRPS patients [47], therefore suggesting that allodynia and hyperalgia should be considered epiphenomena, and not diagnostic criteria.
Every syndrome-defined diagnosis in medicine ends up with a clear exclusion criteria. Other causes of pain must be strictly ruled out. Although this is theoretically obvious, it’s rarely appropriately done in clinical practice, which make some CRPS diagnoses a “waste bucket diagnosis”. Strange patterns of extremity pain are frequently and quickly labeled as CRPS and this is amplified because of the “searching satisfaction beahavior”, which means that most of physicians stop looking for a medical condition once they found something that justify the patient’s symptoms.
For example, 37% of diabetic neuropathy patients would be diagnosed as CRPS if Orlando/IASP criteria is used, neglecting the glucose levels [48]. Del Pinar, in a polemic editorial, postulated that CRPS may not exist [49], since physicians are misdiagnosing entrapments, unstable fractures and even psychiatric disorders as CRPS.
It is well documented that nerve ligation can promote sympathetic fibers sprouting, which can be responsible for vasomotor symptoms [50]. Misdiagnosing nerve entrapments as CRPS is quite dangerous, because, while entrapment treatment should be focused on nerve decompression or nerve ablation (for example cryoablation), CRPS points to sympathetic blocks, spinal cord stimulation and multidisciplinary approach to treatment.
Beyound treatment differences, prognosis is quite different as well. Mislabeling someone as a “CRPS patient” can promote significant nocebo effects due to “Google medical advice”.
Treatment of nerve entrapments requires first the right diagnosis, which usually involves a diagnostic injection, using landmarks, ultrasound (US), or fluoroscopy [51]. The injection of local anesthetic provides the anesthesia, as well as local disruption of the sympathetic stimulation; depot steroids are usually also injected to address the inflammation. If there is only temporary relief, the use of hydrodissection (fluid is injected around the nerve under US guidance to lyse the adhesions around the nerve) may be of use. Cryoneuroablation, which freezes the nerve but leaves the myelin sheath intact, is a technique of nerve destruction that can be safely used on large, myelinated nerves [52]. Peripheral nerve stimulation [53] and surgical release might also offer relief [54].
There are multiple examples of nerve entrapments that likely present similarly to CRPS. Perhaps the most frequently misdiagnosed is the superficial radial nerve (SRN) entrapment, which can be triggered by repetitive pronation/supination [55], IV infiltration [56], forearm laceration [57] Collies fracture [56], handcuffs [58], injections for De Quervain’s tennsynovitis [59] and even a “trivial” injury to the radial forearm (such as banging it on a table or chair). The patient will present with poorly localized radial forearm pain and swelling (Figure 1), with allodynia and pain on movement of the wrist or finger, which can spread to the whole hand and forearm. Patients often try to protect the hand from stimulation with gloves or bandages [57].

Figure 1. Patient presentation with superficial radial nerve entrapment (Image courtesy of Andrea Trescot, MD)
The radial nerve arises from the posterior cord of the brachial plexus, spirals around the humerus, and then descends down the humerus to the elbow. At the elbow, the radial nerve divides into a superficial and deep branch. The superficial branch (SRN) travels under the brachioradialis muscle (BR) and then travels superficially along the distal radial forearm to the thumb and dorsum of the hand (Figure 2) [60].
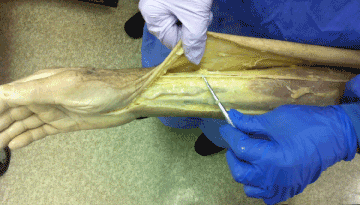
Figure 2. Dissection of the superficial radial nerve (Image courtesy of Andrea Trescot, MD
Splinting, changing work ergonomics, and diagnostic/therapeutic injections are first line treatments. In one series, 71% of 29 patients had excellent or good pain relief after non-operative interventions [61]. If SRN injections give only temporary relief, cryoneuroablation might be indicated. Davies et al. described 6 patients treated with cryoneuroablation via open visualization of the nerve or neuroma [57]. Patients were followed for a mean of 11 months; all reported good to excellent relief, and all returned to work.
Another example of peripheral nerve entrapment mimicking CRPS is the superficial peroneal (fibular) nerve (SPN). Sports are a relatively common cause of SPN entrapment. Cho et al. [62] evaluated 448 cases of peroneal nerve injury; 84 cases (18%) were sports related included skiing (42 cases), football (23 cases), soccer (8 cases), basketball (6 cases), ice hockey (2 cases), track (2 cases) and volleyball (1 case). The other major cause of SPN entrapment is lateral (inversion) ankle sprains [63]. Symptoms of SPN entrapment include swelling, and burning pain in the distribution of the nerve over the lateral calf and dorsum of the foot (Figure 3), at times associated with sensory abnormality [64,65]. The pain may be aggravated by plantar flexion, which stretches the SPN; this may make pressing the accelerator or brake quite painful [66].
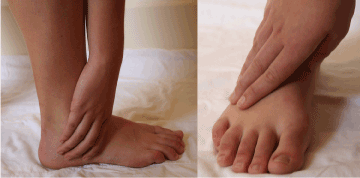
Figure 3. Patient presentation with superficial peroneal nerve entrapment (Image courtesy of Andrea Trescot, MD
SPN entrapment is underdiagnosed [67], and is best diagnosed by physical exam, with tenderness noted over the SPN at the flexor retinaculum (Figure 4). Fifteen percent (18/120) of patients in one study of ankle fractures had symptoms of SPN neuropathy, though only 2 of them were diagnosed before the study; an additional 25 patients were less symptomatic and their SPN injury was only discovered during their study follow-up. In another study, 36% of all ankle fracture patients had SPN injury due to their fracture; less than 2% were diagnosed [68].
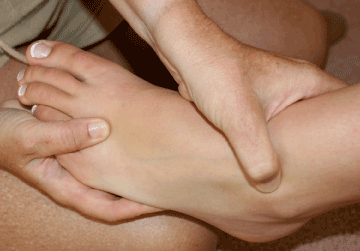
Figure 4. Physical exam to identify superficial peroneal nerve entrapment (Image courtesy of Andrea Trescot, MD
Both of these examples illustrate the similar etiology and symptoms seen with CRPS and peripheral nerve entrapment. In the senior author’s experience, most (if not all) of the patients that present to her clinic with a “CRPS” diagnosis can be diagnosed with a peripheral nerve injury, by taking a careful history and performing a very directed physical exam. A peripheral nerve injection in the office can offer immediate and dramatic relief of the pain, allodynia, and swelling. In her practice, sympathetic blocks are only used if there is no obvious etiology, in an effort to “blow away the smoke to be able to see the fire”; however, it is important to carefully examine the patient immediately after the sympathetic block to identify any persistent areas of pain (and perhaps nerve injury).
Peripheral nerve entrapments can mimic CRPS; more importantly, these unrecognized (and under-recognized) nerve entrapments might actually be the cause of CRPS. The distinction between CRPS type I and II might not be “no nerve injury” versus “nerve injury”, but one of “unrecognized nerve injury” versus “recognized nerve injury”. The focus of treatment should then shift to resolving the nerve entrapment rather than just addressing the sympathetic system.
- Gross CG (1998) Claude Bernard and the consistancy of the internal environment. The Neuroscientist 4: 380-385.
- Mitchell SW, Morehouse GR, Keen WW (1864) Gunshot Wounds and Other Injuries of Nerves. Philadelphia: J. B. Lippincott & Co.
- Mitchell SW (1872) Injuries of the Nerves and Their Consequences. Philadelphia: J. B. Lippincott & Co.
- Coderre TJ, Bennett GJ (2010) A hypothesis for the cause of complex regional pain syndrome-type I (reflex sympathetic dystrophy): pain due to deep-tissue microvascular pathology. Pain Med 11: 1224-1238. [Crossref]
- Veldman PH, Reynen HM, Arntz IE, Goris RJ (1993) Signs and symptoms of reflex sympathetic dystrophy: prospective study of 829 patients. Lancet 342: 1012-1016. [Crossref]
- Albrecht PJ, Hines S, Eisenberg E, Pud D, Finlay DR, et al. Pathologic alterations of cutaneous innervation and vasculature in affected limbs from patients with complex regional pain syndrome. Pain 120: 244-266. [Crossref]
- Oaklander AL, Fields HL (2009) Is reflex sympathetic dystrophy/complex regional pain syndrome type I a small-fiber neuropathy? Ann Neurol 65: 629-638. [Crossref]
- Bruehl S (2010) An update on the pathophysiology of complex regional pain syndrome. Anesthesiology 113: 713-725. [Crossref]
- Oaklander AL (2012) Development of CRPS after shingles: it's all about location. Pain 153: 2309-2310. [Crossref]
- Merskey H, Bogduk N (1994) Classification of Chronic Pain: Descriptions of Chronic Pain Syndromes and Definitions of Pain Terms, (2ndedn), IASP Press.
- Bruehl S, Harden RN, Galer BS, Saltz S, Bertram M, et al. (1999) External validation of IASP diagnostic criteria for Complex Regional Pain Syndrome and proposed research diagnostic criteria. International Association for the Study of Pain. Pain 81: 147-154. [Crossref]
- Harden RN, Bruehl S, Perez RS, Birklein F, Marinus J, et al. (2010) Validation of proposed diagnostic criteria (the "Budapest Criteria") for Complex Regional Pain Syndrome. Pain 150: 268-274. [Crossref]
- Harden RN, Bruehl S, Stanton-Hicks M, Wilson PR (2007) Proposed new diagnostic criteria for complex regional pain syndrome. Pain Med 8: 326-331. [Crossref]
- de Mos M, de Bruijn AG, Huygen FJ, Dieleman JP, Stricker BH, et al. (2007) The incidence of complex regional pain syndrome: a population-based study. Pain 129: 12-20. [Crossref]
- Sandroni P, Benrud-Larson LM, McClelland RL, Low PA (2003) Complex regional pain syndrome type I: incidence and prevalence in Olmsted county, a population-based study. Pain 103: 199-207. [Crossref]
- Jellad A, Salah S, Ben Salah Frih Z (2014) Complex regional pain syndrome type I: incidence and risk factors in patients with fracture of the distal radius. Arch Phys Med Rehabil 95: 487-492. [Crossref]
- Geertzen JH, Bodde MI, van den Dungen JJ, Dijkstra PU, den Dunnen WF (2014) Peripheral nerve pathology in patients with severely affected complex regional pain syndrome type I. Int J Rehabil Res 38: 121-30. [Crossref]
- Dilek B, Yemez B, Kizil R, Kartal E, Gulbahar S, et al. (2012) Anxious personality is a risk factor for developing complex regional pain syndrome type I. Rheumatol Int 32: 915-920. [Crossref]
- Harden RN, Oaklander AL, Burton AW, Perez RS, Richardson K, et al. (2013) Complex regional pain syndrome: practical diagnostic and treatment guidelines, 4th edition. Pain Med 14: 180-229. [Crossref]
- Puchalski P, Zyluk A (2005) Complex regional pain syndrome type 1 after fractures of the distal radius: a prospective study of the role of psychological factors. J Hand Surg 30: 574-580. [Crossref]
- Beerthuizen A, Stronks DL, Van't Spijker A, Yaksh A, Hanraets BM, et al. (2012) Demographic and medical parameters in the development of complex regional pain syndrome type 1 (CRPS1): prospective study on 596 patients with a fracture. Pain 153: 1187-1192. [Crossref]
- Moseley GL, Herbert RD, Parsons T, Lucas S, Van Hilten JJ, et al. (2014) Intense pain soon after wrist fracture strongly predicts who will develop complex regional pain syndrome: prospective cohort study. J Pain 15: 16-23. [Crossref]
- Dijkstra PU, Groothoff JW, ten Duis HJ, Geertzen JH (2003) Incidence of complex regional pain syndrome type I after fractures of the distal radius. Eur J Pain 7: 457-462. [Crossref]
- Dijkstra PU, van der Schans CP, Geertzen JH (2003) Risk perception of developing complex regional pain syndrome I. Clin Rehabil 17: 454-456. [Crossref]
- Allen G, Galer BS, Schwartz L (1999) Epidemiology of complex regional pain syndrome: a retrospective chart review of 134 patients. Pain 80: 539-544. [Crossref]
- van Rijn MA, Marinus J, Putter H, Bosselaar SR, Moseley GL, et al. (2011) Spreading of complex regional pain syndrome: not a random process. J Neural Transm (Vienna) 118: 1301-1309. [Crossref]
- Menorca RM, Fussell TS, Elfar JC (2013) Nerve physiology: mechanisms of injury and recovery. Hand Clin 29: 317-330. [Crossref]
- Kopell HP, Thompson WA (1976) Peripheral Entrapment Neuropathies. Baltimore: Williams and Wilkin.
2021 Copyright OAT. All rights reserv
- Hirose CB, McGarvey WC (2004) Peripheral nerve entrapments. Foot Ankle Clin 9: 255-269. [Crossref]
- Vinik A, Mehrabyan A, Colen L, Boulton A (2004) Focal entrapment neuropathies in diabetes. Diabetes Care 27: 1783-1788. [Crossref]
- Dellon AL (2014) Susceptibility of nerve in diabetes to compression: implications for pain treatment. Plast Reconstr Surg 134: 142S-50S. [Crossref]
- Rydevik B, Lundborg G, Bagge U (1981) Effects of graded compression on intraneural blood blow. An in vivo study on rabbit tibial nerve. J Hand Surg Am 6: 3-12. [Crossref]
- Dellon AL (2008) The four medial ankle tunnels: a critical review of perceptions of tarsal tunnel syndrome and neuropathy. Neurosurg Clin N Am 19: 629-648. [Crossref]
- Smith EM, Sonstegard DA, Anderson WH Jr (1977) Carpal tunnel syndrome: contribution of flexor tendons. Arch Phys Med Rehabil 58: 379-385. [Crossref]
- Pechan J, Julis I (1975) The pressure measurement in the ulnar nerve. A contribution to the pathophysiology of the cubital tunnel syndrome. J Biomech 8: 75-79. [Crossref]
- Rydevik B, McLean WG, Sjöstrand J, Lundborg G (1980) Blockage of axonal transport induced by acute, graded compression of the rabbit vagus nerve. J Neurol Neurosurg Psychiatry 43: 690-698. [Crossref]
- Fern R, Harrison PJ (1991) The effects of compression upon conduction in myelinated axons of the isolated frog sciatic nerve. J Physiol 432: 111-122. [Crossref]
- Mackinnon SE (1992) Double and multiple "crush" syndromes. Double and multiple entrapment neuropathies. Hand Clin 8: 369-390. [Crossref]
- Upton AR, McComas AJ (1973) The double crush in nerve entrapment syndromes. Lancet 2: 359-362. [Crossref]
- Dellon AL, Mackinnon SE (1991) Chronic nerve compression model for the double crush hypothesis. Ann Plast Surg 26: 259-264. [Crossref]
- Kernohan J, Levack B, Wilson JN (1985) Entrapment of the superficial peroneal nerve. Three case reports. J Bone Joint Surg Br 67: 60-61. [Crossref]
- Ikeuchi M, Izumi M1, Aso K1, Sugimura N1, Tani T1 (2013) Clinical characteristics of pain originating from intra-articular structures of the knee joint in patients with medial knee osteoarthritis. Springerplus 2: 628. [Crossref]
- Kachar SM, Williams KM, Finn HA (2008) Neuroma of the infrapatellar branch of the saphenous nerve a cause of reversible knee stiffness after total knee arthroplasty. J Arthroplasty 23: 927-930. [Crossref]
- Trescot AM, Brown MN, Karl HW (2013) Infrapatellar saphenous neuralgia - diagnosis and treatment. Pain Physician 16: E315-324. [Crossref]
- Unwin A, Scott J (1999) Nerve palsy after hip replacement: medico-legal implications. Int Orthop 23: 133-137. [Crossref]
- Sapienza P, Edwards JD, Mingoli A, McGregor PE, Cavallari N, et al. (1996) Ischemia-induced peripheral arterial vasospasm role of alpha 1- and alpha 2-adrenoceptors. J Surg Res 62: 192-196. [Crossref]
- Treede RD, Davis KD, Campbell JN, Raja SN (1992) The plasticity of cutaneous hyperalgesia during sympathetic ganglion blockade in patients with neuropathic pain. Brain 115: 607-621. [Crossref]
- Galer BS, Bruehl S, Harden RN (1998) IASP diagnostic criteria for complex regional pain syndrome: a preliminary empirical validation study. International Association for the Study of Pain. Clin J Pain 14: 48-54. [Crossref]
- Del Pinal F (2014) Reply: F del Piñal. Editorial. I have a dream ... reflex sympathetic dystrophy (RSD or Complex Regional Pain Syndrome - CRPS I) does not exist. J Hand Surg Eur Vol 39: 215-6. [Crossref]
- Chung K, Chung JM (2001) Sympathetic sprouting in the dorsal root ganglion after spinal nerve ligation: evidence of regenerative collateral sprouting. Brain Res 895: 204-212. [Crossref]
- Trescot AM, Murinova N (2016) Peripheral nerve entrapment - injection technique. In: Trescot AM (Ed.), Peripheral Nerve Entrapment, Clinical Diagnosis and Management. New York, NY: Springer.
- Trescot AM (2003) Cryoanalgesia in interventional pain management. Pain Physician 6: 345-360. [Crossref]
- Gulati A, Karl HW, Zhang T, Trescot AM (2016) Neuromodulation for the treatment of nerve entrapment syndromes. In: Trescot AM, ed. Peripheral Nerve Entrapment, Clinical Diagnosis and Management. New York, NY: Springer.
- Gulati A, Krashin D, Karl HW (2016) Surgical treatment of nerve entrapment syndromes. In: Trescot AM (Ed.), Peripheral Nerve Entrapment, Clinical Diagnosis and Management. New York, NY: Springer.
- Keefe DT, Lintner DM (2004) Nerve injuries in the throwing elbow. Clin Sports Med 23: 723-742, xi. [Crossref]
- Braidwood AS (1975) Superficial radial neuropathy. J Bone Joint Surg Br 57: 380-383. [Crossref]
- Davies E, Pounder D, Mansour S, Jeffery IT (2000) Cryosurgery for chronic injuries of the cutaneous nerve in the upper limb. Analysis of a new open technique. J Bone Joint Surg Br 82: 413-415. [Crossref]
- Massey EW, Pleet AB (1978) Handcuffs and cheiralgia paresthetica. Neurology 28: 1312-1313. [Crossref]
- Chodoroff G, Honet JC (1985) Cheiralgia paresthetica and linear atrophy as a complication of local steroid injection. Arch Phys Med Rehabil 66: 637-639. [Crossref]
- Trescot AM, Karl HW (2016) Superficial radial nerve. In: Trescot AM (Ed.), Peripheral Nerve Entrapment, Clinical Diagnosis and Management. New York, NY: Springer.
- Lanzetta M, Foucher G (1993) Entrapment of the superficial branch of the radial nerve (Wartenberg's syndrome). A report of 52 cases. Int Orthop 17: 342-345. [Crossref]
- Cho D, Saetia K, Lee S, Kline DG, Kim DH (2011) Peroneal nerve injury associated with sports-related knee injury. Neurosurg Focus 31: E11. [Crossref]
- O'Neill PJ, Parks BG, Walsh R, Simmons LM, Miller SD (2007) Excursion and strain of the superficial peroneal nerve during inversion ankle sprain. J Bone Joint Surg Am 89: 979-986. [Crossref]
- Murinova N, Chuiu SC, Krashin D, Karl HW (2016) Superficial peroneal nerve. In: Trescot AM (Ed.), Peripheral Nerve Entrapment, Clinical Diagnosis and Management. New York, NY: Springer; 2016.
- Lowdon IM (1985) Superficial peroneal nerve entrapment. A case report. J Bone Joint Surg Br 67: 58-59. [Crossref]
- Anandkumar S (2012) Physical therapy management of entrapment of the superficial peroneal nerve in the lower leg: a case report. Physiother Theory Pract 28: 552-561. [Crossref]
- Styf J (1989) Entrapment of the superficial peroneal nerve. Diagnosis and results of decompression. J Bone Joint Surg Br 71: 131-135. [Crossref]
- Redfern DJ, Sauvé PS, Sakellariou A (2003) Investigation of incidence of superficial peroneal nerve injury following ankle fracture. Foot Ankle Int 24: 771-774. [Crossref]




