With recent developments in echocardiography, most operators need to be familiar with cross-sectional cardiac anatomy. However, the precise spatial relationships of the components of the heart can be difficult to understand. Plastination has been known as an excellent technique that helps to keep anatomical specimens in a dry, firm, shape-preserved condition. In this paper, the method of producing plastinated pig heart specimens for learning echocardiography is described. The final specimens show the detailed structure of the heart, which provides students and beginners who are training for echocardiography a better chance of developing a three-dimensional understanding of the heart.
echocardiography, plastination, porcine heart
It is widely accepted that echocardiography is one of the non-invasive and very useful tools for diagnosing cardiovascular disease. However, its usefulness and the correct diagnosis depends on both the proper skills and the well-understanding of the heart. The precise spatial relationship of the components of the heart can be difficult to understand for students and beginners. Therefore, anatomic drawings, atlases, and models that correspond to standard ultrasound images are necessary to assist echocardiographic interpretation. However, from among the other artificial models of the heart, it is difficult to find instructional materials suitable for students or beginners who have just begun to study echocardiography.
The plastination method has been used to process cadaveric material and make it into dry, odorless, and durable specimens that can be handled without gloves [1-3]. From this standpoint, the potential value of plastination is increasingly appreciated [4-9]. It is possible to produce a plastinated specimen of a porcine heart, cut off the suitable cross-section surfaces for standard echocardiographic images, and make a comparison while performing echocardiography.
Material
The heart of a pig subjected to meat processing with no morphological abnormalities on macroscopic inspection was used.
Dilatation under hydrostatic pressure
The heart was flushed with tap water to remove blood clots.
For the injection into the coronary arteries (left anterior descending artery, left circumflex artery, and right coronary arteries), colored silicone KE24 (Shin-Etsu Chem. Co., Tokyo, Japan), red, blue, and yellow, respectively, was used.
In order to provide a proper shape, short polyethylene tubes of the appropriate dimensions were inserted into the veins of the veins, the vena cavas, the aorta, and the pulmonary artery. After putting the cork stoppers on the veins of lung and the vena cavas, dilatation and fixation were performed with filled 10% formalin solution (10 parts 3538 wt. % formaldehyde solution in water and 90 parts tap water) through the two tubes inserted into the aorta and the pulmonary artery with hydrostatic pressure. The aorta and the pulmonary artery were plugged to keep the fixative inside, and then the heart was immersed in a solution of 10% formalin for 7 days. After fixation, all cork stoppers and tubes were removed, and the heart was then immersed in running tap water for 12-24 hours.
To dehydrate, the precooled heart was freeze-substituted with pure acetone with three changes every week. To degrease, if necessary, the dehydrated heart was left at room temperature without removing it from acetone for an additional week.
Impregnation in a vacuum chamber
The heart was then transferred into the silicone KE108, and vacuum impregnation in the freezer for 1-3 days was started.
Hardening and aftercare
The heart was taken out of the silicone, excess silicone was wiped off, and it was finally mounted on a supporting device to dry out and harden. The plastinated heart was cut with a flensing knife to obtain four cross-sections corresponding to the four standard images of echocardiography from only one heart: the left parasternal cranial long-axis view and the right parasternal short-axis view of papillary muscle level, of mitral valve level, and of aortic valve level. Each piece could be attached or detached using a magnet embedded in each cross-sectional surface.
The final plastinated heart in this study was shaped into the specimen suitable for the aim of this study. On the external surface, the complete anatomical detail of the heart, the right and left ventricles and atria, the interventricular grooves, the coronary venous system, and the main afferent and efferent vessels, were preserved in good condition and easily recognized. Inside, the plastinated heart showed in detail the structure of the interior of both left and right ventricles and atria, the mitral valve, the tricuspid valve, the base of the aorta and the aortic valve(Figure 1). The color-coded coronary arteries were recognized at each cross-sectional surface of the specimen, providing a useful understanding of the perfusion areas of each coronary artery(Figure 1).
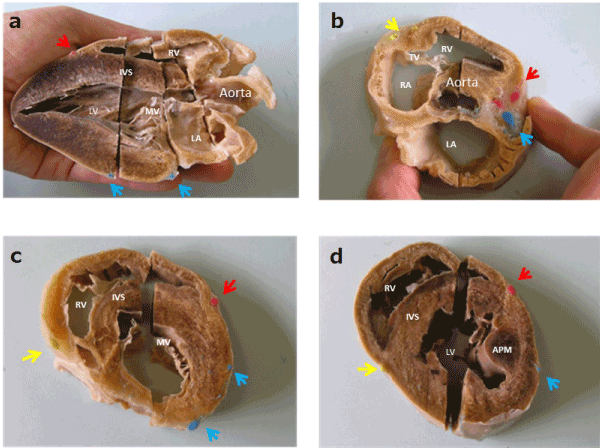
Figure 1. Internal aspects of the plastinated pig heart. a: parasternal long-axis view; b: short-axis view, aortic valve level; c: Short-axis view, mitral valve level; d: Short-axis view, papillary muscle level. Yellow arrow: right coronary artery; Red arrow: left anterior descending artery; Blue arrow: left circumflex artery. (RV: right ventricle; RA: right atrium; LV: left ventricle; LA: left atrium; PA: pulmonary artery; MV: mitral valve; APM: anterior papillary muscle; IVS: inter-ventricular septum.)
Each divided piece of the plastinated heart could be attached or detached using a magnet embedded in each cross-sectional surface, and each could be taken out freely to examine the preferred echocardiographic images, the left parasternal long-axis cranial view and right parasternal short-axis views of the papillary muscle, the mitral valve, and the aortic valve levels, from only one heart(Figure 2). The cutting lines of the complete specimen conformed with the direction of the ultrasound beam when obtaining the standard echocardiographic images, and an image could be made with the ultrasound probe. However, this specimen was fixed as a contracted heart; if a further dilated specimen were obtained, a more realistic specimen could be prepared. In addition, it was hard to get the cutting lines suitable for the echocardiographic images without scrutinizing the plastinated specimen. As a result, completing the plastinated heart takes a long time, about six weeks or more depending on the weight of the heart.
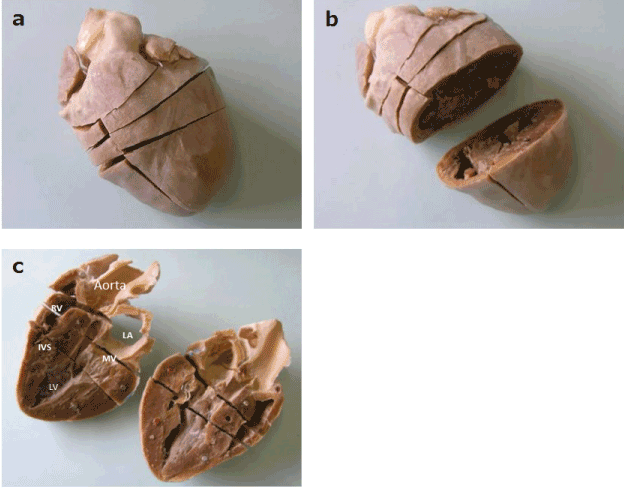
Figure 2. External aspects of the plastinated pig heart showing the anterior (a), transverse incision (b) and longitudinal incision (c), and some embedded magnets. (RV: right ventricle; LV: left ventricle; LA: left atrium; MV: mitral valve; IVS: inter-ventricular septum.)
The goal of this study was to prepare the anatomical material with instructive value to facilitate understanding of cross-sectional images of ultrasonography. For many patients with various diseases, ultrasonography is today the definitive, noninvasive diagnostic tool. Understanding the orientation of the target organ of ultrasonography is important for beginners to understand these images, and to make a correct diagnosis. However, the heart is perhaps one of the most difficult organs to understand for beginners due to its complex anatomy. Therefore, the cross-sectional images of echocardiography have been especially hard for beginners to understand. Undergraduate students, learning anatomy in classes, have been instructed by the use of atlases, prosected cadaver specimens, artificial models, and multimedia packages [10,11]. Although prosected cadaver specimens facilitate understanding of the structural details of organs, they also have a negative side, in that they are expensive, brittle, and emotionally disturbing for some students [12]. However, the senses of touch and sight are missing when using atlases and multimedia packages [13]. Artificial models introduced as substitutes for cadaver specimens are inaccurate and not suitable for perceiving cross-sectional images of ultrasonography. In the present study, the silicone plastination technique was used to produce a heart specimen that showed remarkable structural detail, preservation, durability, and ability to be grasped in the hand. Because of its correspondence with conventional images obtained using echocardiography, each surface of each section of the plastinated heart specimen displayed the cross-sectional anatomy with great detail. In addition, the section lines of the exterior heart specimen reflected the transverse directions of the ultrasound beam, which demonstrate all but the basal images of echocardiography despite using only one heart. The left anterior descending and circumflex coronary arteries and the right coronary arteries, were color-coded by the injection of red, blue, and yellow colored silicone, respectively, to show their perfusion areas. The magnetic-plugged slices made the plastinated heart an adjustable specimen, allowing the observer to view any slice, providing greater usefulness for better anatomic interpretation. Nevertheless, some limitations are involved in the use of plastinated heart specimens for anatomical materials. First, it is difficult to obtain identical heart models. Secondly, although plastinated heart specimens are highly durable, they are not produced on a large-scale. Thirdly, it takes a long time, sometimes as long as two months, to acquire a completed plastinated heart. Fourthly, cutting out the plastinated heart specimen is a difficult process, one that cannot be redone without adequate knowledge of dissection and echocardiographic images. However, by completing a plastinated heart specimen, the students who are able to do it are more likely to be able to successfully identify some of the key constructions on their own before beginning echocardiography [14].
In conclusion, the final specimens show the detailed structure of the heart, which provides students and novices who are training for echocardiography a better chance of developing a three-dimensional understanding of the heart.
This work was supported by KAKENHI(22659104).
None.
This article does not contain any studies with human or animal subjects performed by the any of the authors.
- Hubbell DS, Dwornik JJ, Alway SE, Eliason R, Norenberg RE (2002) Teaching gross anatomy using living tissue. Clin Anat 15: 157–159. [Crossref]
- von Hagens G (1979) Impregnation of soft biological specimens with thermosetting resins and elastomers. Anat Rec 194: 247–256. [Crossref]
- Grondin G, Orly R (2004) The New Plastination Index. International Society for Plastination.
- Fritsch H (1996) Sectional anatomy of connective tissue structures in the hindfoot of the newborn child and the adult. Anat Rec 246: 147–154.
- Qui MG, Zhang SX, Liu ZJ, Tan LW, Wang YS, et al. (2004) Three-dimensional computational reconstruction of lateral skull base with plastinated slices. Anat Rec Part A 278: 437-442. [Crossref]
- Mizer LA, Farnum CE, Schenck PD (2002) The modular resource center: integrated units for the study of the anatomical sciences in a problem-based curriculum. Anat Rec Part B 269: 249–256. [Crossref]
- de Barros N, Rodrigues CJ, Rodrigues Jr AJ, de Negri Germano MA, Cerri GG (2001) The value of teaching sectional anatomy to improve CT scan interpretation. Clin Anat 14: 36-41. [Crossref]
2021 Copyright OAT. All rights reserv
- Reidenberg JS, Laitman JT (2002) The new face of gross anatomy. Anat Rec Part B 269: 81–88. [Crossref]
- Latorre RM1, García-Sanz MP, Moreno M, Hernández F, Gil F (2007) How Useful Is Plastination in Learning Anatomy? JVME 34: 172-176. [Crossref]
- Miles KA (2005) Diagnostic imaging in undergraduate medical education: an expanding role. Clin Radiol 60: 742-745. [Crossref]
- Sugand K, Abrahams P, Khurana A (2010) The Anatomy of Anatomy: A Review for Its Modernization. Anat Sci Educ 3:73-93. [Crossref]
- Arraez-Aybar LA, Castano-Collado G (2008) Dissection as a modulator of emotional attitudes and reactions of future health professionals. Med Edu 42: 563-571. [Crossref]
- Choudhury B, Gouldsborough I (2010) Use of Interactive Sessions and E-learning in Teaching Anatomy to First-year Optometry Students. Anat Sci Educ 3: 39-4. [Crossref]
- Suenaga H, Momose N, Kobayashi Y (2012) Trial and development of effective instructional materials for echocardiography. Jpn J Med Ultrasonic 39: 457-462.


