Key words
mucormycosis, mucor; ketoacidosis, surgical debridement, antifungal therapy, poor survival outcome
Abstract
Mucormycosis can be a devastating and fatal disease if not treated promptly; unfortunately diagnosis can be a delayed process. Depending on the Hospitals´ resources diagnosis can be more accurate or not. The mainstay of treatment is still surgical debridement with antifungal therapy. We present a case of a 75-year-old female, with immunosuppression and uncontrolled diabetes that presents to the emergency room with right hemifacial edema. CT scan revealed occupation of the right maxillary sinus, ethmoidal and sphenoid sinus and cranial base bone erosion with close proximity to the brain. Culture and biopsy specimens were taken with a delayed diagnosis of Mucor. The patient survived three months after surgical and antifungal therapy despite a very poor prognosis.
Introduction
The most common form of mucormycosis in diabetes and immunosuppressive patients is rhinocerebral mucormycosis. Mucorales is referred to as mucormycosis. Mucorales are characterized by nonseptate, thick-walled hyphae. Initial symptoms tend to be silent, and a pattern of different colors may be seen depending on the different clinical phases. As the infection progresses, an erythematous to black necrotic eschar can be seen. Six families of Mucorales can cause mucormycosis, Mucor species being the rarest [1].
Rhinocerebral mucormycosis is a devastating and life-threatening condition of the nasal mucosa that can have a very rapid contiguous. It can invade blood vessels and cause thrombosis and necrosis. The usual site of entry is the nasal mucosa [1].
Culture and histopathology are the initial assessment choices, imaging is important to understand the degree of invasion, but lesions are unspecific for mucormycosis. Histopathology can aid in diagnosing mucormycosis, but can`t describe the species. The best method for this is PCR. Mainstay of therapy is based in surgical debridement and antimycotic therapy, but stabilization of comorbidities is of extreme importance (ketoacidosis, immunosuppression, etc.). Liposomal amphotericin B is the treatment of choice 5 to 10 mg/kg/day. Posaconazole or caspofungin are substitute choices as well. Other antimycotic drugs such as voriconazole, itraconazole or fluconazole do not act on Mucorales. It is important to mention that there can variability in resistance and susceptibility to amphotericin B and other antifungal drugs that can be a downside in successful treatment [2-4].
The proper identification of mycological methods is questioned in many health centers so that statistical data can be biased. Early diagnosis is of extreme importance for survival management. Limitations are nonspecific clinical manifestations, lack of laboratory studies including access to molecular methods, and nonspecific characteristics in radiological studies. Early clinical manifestations in diabetic patients or complicated diabetes like ketoacidosis, may manifest with eye vision complications. Diplopia may be due to involvement of the extraocular nerves. Hyperglycemia is usually manifested as blurry vision, but not diplopia. Diplopia in a diabetic patient, particularly one who may be hyperglycemic or ketoacidotic, usually signifies involvement of the extraocular muscles of the nerves innervating the extraocular muscles, especially in invasive mucormycosis. Hyperglycemia in diabetic patients may also produce blurring of vision but typically does not produce diplopia. Another early clinical manifestation that may be seen is a necrotic black eschar in nasal mucosa, maxillary, sino-orbital, or facial tissues [3-5].
The variability of susceptibility to amphotericin B and resistance to most other conventional antifungal agents leads to major limitations in successful treatment of this devastating infection. Literature refers that voriconazole prophylaxis may have an important increased risk of invasive mucormycosis, especially in immunocompromised patients with bone marrow transplant or hematological malignancy [3-5].
Mucorales and other important filamentous fungi share portal entry mechanisms, but the latter have additional virulence characteristics that make them unique by the way of evasion and progression of the disease. Accuracy and prompt diagnosis for correct therapy initiation (surgical and medically) is of extreme importance, but conventional diagnosis are limited and molecular methods may be more accurate [4,5].
After candidiasis and Aspergillus, Mucormycosis has become the third most common invasive mycosis in patients with hematological malignancies and bone marrow transplantation [6].
Case report
A 75-year-old female is admitted to the hospital for hemifacial edema, dyspnea and fever for the past two days. The past medical history is notable for hypertension and uncontrolled diabetes mellitus type 2. Physical examination revealed a Kussmaul respirations, Glasgow coma scale 12 points, right hemifacial edema, anosmia, nasal congestion, necrotic scar in the right nasal vestibule with foul smell. Laboratory test showed a white blood count: 32,000, neutrophils of 92%, glucose 825 mg/dl, creatinine: 1.32 mg/dl, urea: 120 mg/dl, Cl: 85 mEq/L, K: 2.3 mEq/L, corrected sodium: 130 mEq/L, an effective Osmolarity: 349 mmol/kg. Arterial blood gases; pH: 7.12, HCO3: 6.0, BE: - 15, Anion Gap: 39. Brain and Paranasal CT revealed: Invasion of a dense tissue in ethmoidal cells, sphenoidal, frontal and maxillary sinus. Microbiological and biopsy samples were taken, and patient was admitted into the ICU. The next 10 hours inspite of medical treatment and lowering of glucose levels, worsening of her neurological status was noted. Surgical treatment was decided due to the high suspicion of mucormycosis. Midfacial degloving approach was decided for a good debridement control of the necrotic tissue. Frozen section biopsy was sent to pathology with a conclusion of a fungus suspicion. The Infectology team was consulted because of the results and they decided to administered intravenous voriconazole until the final results. Microbiology and histopathology confirmed Mucormycosis genera Mucor, which led to the decision to administer liposomal amphotericin B. After one week of treatment acute kidney injury developed with a creatinine of 3.2 mg/dl, amphotericin B was discontinued and oral posaconazole was given instead because of a greater in vitro susceptibility. Patient had a slow gradual improvement with no further surgical interventions and after three moths no active manifestation were seen with negative cultures and biopsy samples as well. Sequel of the disease was an opening from the right hard palate and the right floor of the nose. Patient could not eat well, especially liquids, so maxillofacial was consulted and prosthesis at the site was placed with no further complications reported (Figures 1-7).
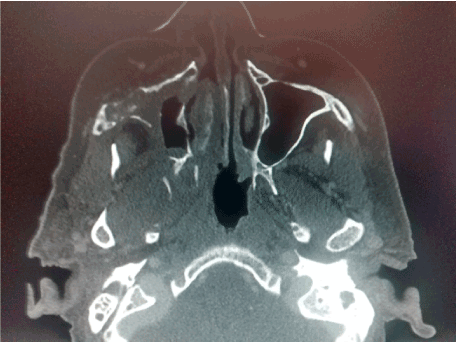
Figures 1. CT scan that involves right maxillary bone destruction with subcutaneous edema.
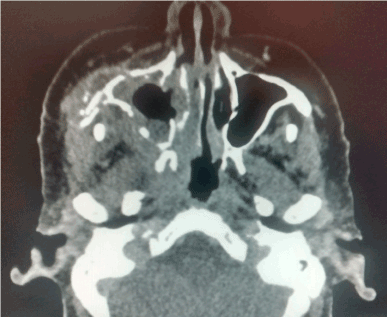
Figure 2. CT scan that involves right maxillary bone destruction with subcutaneous edema.
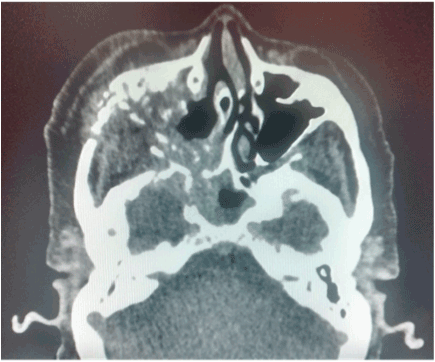
Figure 3. CT scan that involves right maxillary bone destruction with subcutaneous edema.
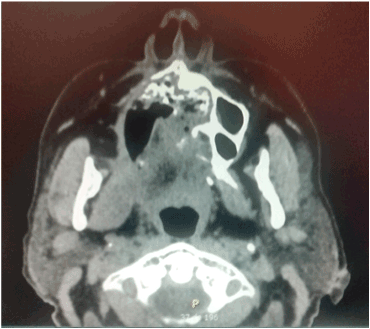
Figures 4. Sequel of right palatal destruction with nasal communication.
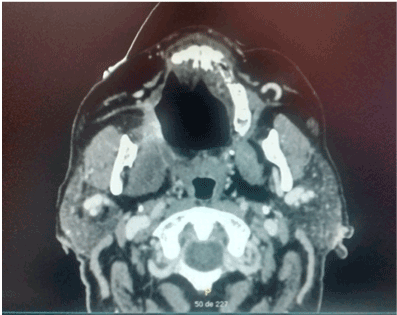
Figures 5. Sequel of right palatal destruction with nasal communication.
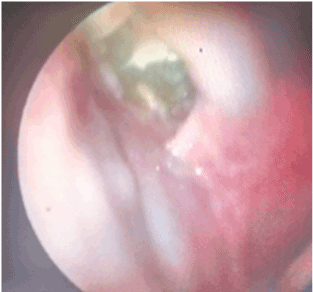
Figures 6. Fibroscopy showing right palatal destruction sequel.
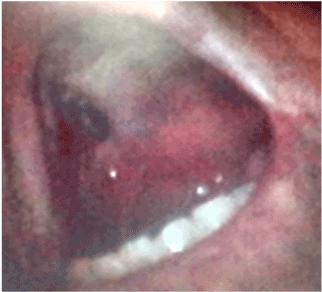
Figure 7. Right palatal destruction sequel.
Discussion
Ketoacidosis, immunosuppression, hematological malignancies remains one of the key factors for mucorales to invade and produce life threatening conditions. In the case presented, early surgical intervention and prompt stabilization of the patient permitted a better outcome than expected. Laboratory detection was delayed, as well as biopsy specimen, but decision based on patients ketoacidosis, clinical symptoms, and signs, and radiological studies made the decision to act quickly as a probable mucormycosis. PCR, in situ hybridization, were not performed due to the lack of such techniques in out center. Results were later confirmed by cultures and aided by the surgical pathology team. This is important to mention, because not all centers have new molecular diagnostic tools due to their costs, even though it is a developed country and a Hospital of reference. There are few Hospitals that are key national centers, but sending specimens would delay the diagnosis and treatment. Antifungal therapy with liposomal amphotericin B is expensive, and like the literature describes, voriconazole administered as prophylaxis in immunocompromised patients can have a high risk of invasiveness of the mucormycosis. In this case, patient stayed in a stable conditioned, although that acute kidney injury was developed because of the use of amphotericin B. Patient was later changed to oral posaconazole and further improvement was seen.
Conclusions
An early diagnosis and treatment is the key element to better survival rate in mucormycosis. Surgery debridement and antifungal drugs remain the mainstay of therapy. It is important to stabilize the patient if there are comorbidities (ketoacidosis, immunosuppression, etc). Better methods of diagnosis have to be developed in regards to cost and time efficiency for a more guided early treatment. The answer is in new molecular methods, but certain but cost are elevated, and worldwide access is still a challenge.
References
- Lunge SB, Sajjan V, Pandit AM, Patil VB (2015) Rhino cerebro cutaneous mucormycosis caused by Mucor species: A rare causation. Indian Dermatol Online J 6: 189-192. [Crossref]
- Bellazreg F, Hattab Z, Meksi S, Mansouri S, Hachfi W, et al. (2014) Outcome of mucormycosis after treatment: report of five cases. New Microbes New Infect 6: 49-52. [Crossref]
- Alvarez E, Sutton DA, Cano J, Fothergill AW, Stchigel A, et al. (2009) Spectrum of zygomycete species identified in clinically significant specimens in the United States. J Clin Microbiol 47: 1650-1656. [Crossref]
- Katragkou A, Walsh 2021 Copyright OAT. All rights reservosis more difficult to cure than more common mycoses? Clin Microbiol Infect 20: 74-81. [Crossref]
- Walsh TJ, Gamaletsou MN, McGinnis MR, Hayden RT, Kontoyiannis DP (2012) Early clinical and laboratory diagnosis of invasive pulmonary, extrapulmonary, and disseminated mucormycosis (zygomycosis). Clin Infect Dis 54: S55-60. [Crossref]
- Petrikkos G, Skiada A, Lortholary O, Roilides E, Walsh TJ, et al. (2012) Epidemiology and clinical manifestations of mucormycosis. Clin Infect Dis 54: S23-34. [Crossref]







