Abstract
Titanium and zirconia are the biomaterials used in medicine and dentistry for restorations and implants. However, the adhesion of zirconia to other materials is weak. The implant fixation of titanium and zirconia is slow. This is due to the chemical and bio-inertness. The performance can be improved by coating with other materials. This mini review will discuss the coating methods with zirconia to improve adhesion in dental restorations and bioactive coating in titanium and zirconia implants to enhance the rate of bone healing at early implanation times.
Key words
titanium, zirconia, implants, coatings, bioactive materials
Introduction
Nowadays, the world population is increasing dramatically for the past few decades. This presents many public health challenges. One of the challenges is to restore the damaged and lost bone tissues due to accidents and aging. Zirconia and titanium and its alloys are the two most important biomaterials used in medicine and dentistry to restore, repair and regenerate the bone tissues [1,2].
Titanium and its alloys are widely used for medical and dental implants because of the excellent biocompatibility, good mechanical properties and high corrosion resistance. They are also used in dentistry as bridges, crowns and overdentures [3]. The rate of osseointegration of titanium implant fixation to bone tissue is slow. It takes a longer period for the strong bond formation between titanium and bone tissue cells [4]. This lengthens the clinical treatment duration.
Zirconia is used in dentistry as orthodontic brackets, ceramic restorations and also a substitute for titanium as implant material because of the excellent mechanical, biocompatibility, tooth-like colour and minimal ion release [5]. However, zirconia is chemically inert and bonding to other materials such as resin composites is difficult. As an implant material, bone healing process is slow owing to the biological inertness [6].
Surface coatings of zirconia and titanium are one of the best approaches to improve the adhesion to other materials in restorations and rate of implant fixation to bone tissues. After surface coatings, there is a change of both (or one of) physical and chemical properties of the surface. On the one hand, it alters the surface topography such as increasing the surface roughness to enhance micro-mechanical interlocking. On the other hand, it alters the surface chemistry. This result is an activation of the surface towards the adhesion to other materials and enhancement of the biological activity to implant fixation.
Surface coatings
Coatings for adhesion promotion in restorations
Grit blasting: In dental laboratories, the standard procedures for zirconia restorations are grit-blasting with alumina or silica-coated alumina sand particles under an air pressure (Figure 1). After grit-blasting, the surface roughness is increased which enhances the adhesion by micro-mechanical interlocking. Moreover, a silica-coated alumina or alumina thin layer is formed. This is followed by an application of silane coupling agent before cementation by light curing [7]. The silane coupling agent which contains two functional groups connects the resin composite and the zirconia. One functional group, -SiOR, which forms silanol, -SiOH, after activation reacts with the surface silica or alumina layer to form strong siloxane, -Si-O-Si-, or -Si-O-Al- linkages. The other functional group, -C=C-, reacts with the resin monomers containing -C=C- to form -C-C- bond by a free radical reaction [8].

Figure 1.SEM micrograph of grit-blasted zirconia surface using silica-coated alumina sand particles.
Sol-gel coatings: The grit-blasting method used has a main drawback: it would cause surface defects or crack formation because of high energy sand particles impacting the surface. The surface damage also depends on the sand particle size, e.g. 30 vs 110 µm, of the sand powder used and the blasting pressure applied. That results in affecting the long term clinical performance [9]. Therefore, other methods are developed to promote resin zirconia bonding without any surface cracks formed.
Sol-gel method is an alternative technique. It is a mild method with relative low processing temperature, more economic and energy saving. The basic principle is the formation of a silica network by hydrolysis of silica precursors under acidic and basic medium. The silica particles react and deposit on the surface to form a silica layer. A silane coupling agent is applied and followed by a cementation. Some methods reported include tetraethoxysilane hydrolysis and silicon nitride hydrolysis under basic medium (Figure 2). These coating methods would not induce any surface flaws. However, the resin zirconia bond strengths reported are lower than the grit-blasting method after thermocycling aging [10,11].

Figure 2.Sol-gel coatings on zirconia surface: a. tetraethoxysilane hydrolysis and b. silicon nitride hydrolysis. A silica coating is formed.
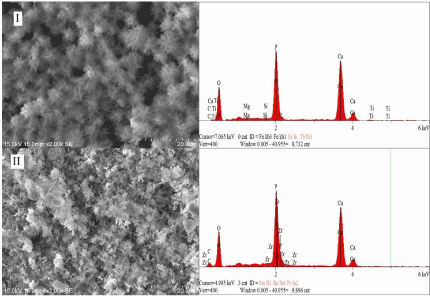
Figure 3.SEM micrographs (2000x) and EDX analysis of hydroxyapatite coating: I. titanium surface and II. zirconia surface modified with a novel silane coupling agent.
Other coating methods: Different coating methods have been investigated and reported. These include: 1, selective infiltration etching, 2. nano-structured alumina coating, and 3. chemical vapor deposition have been studied to assess the resin zirconia bonding [8]. They have different mechanisms to modify the zirconia surface by coating different materials.
Coatings to enhance implant ossteointegration
Bioactive materials such as hydroxyapatite and bioactive glass are used for coating zirconia and titanium implants to improve the biological interactions with tissue cells. Many studies have been reported these coating materials are able to enhance osseointegration and promote rapid and early implant stabilization.
Bioactive glass coatings: The bioactive glass, silicate-based (SiO2-CaO-P2O5), has been reported to be able to bond to tissue cells because of the formation of a hydroxyapatite surface layer. It can elicit a specific biological response between the implant material and the tissue cells [12]. Coating with the bioactive glass on metallic and ceramic implants has been reported to improve the rate of osseointegration [13-15]. Several techniques are used for bioactive glass coating: enameling, sol-gel, plasma spraying, and electrophoretic deposition [16].
Hydroxyapatite coatings: Hydroxyapatite is the most widely used biomaterials in implant coating due to the chemical similarity to human bone and teeth. It has excellent biocompatibility, good osteoconduction and osseointegration. There are numerous studies reported the improved osseointegration at the early stage after implantation of hydroxyapatite coatings on titanium implants [17-19]. Several methods have been studied for hydroxyapatite coating on a zirconia and titanium implant surface to promote implant fixation. Some examples are plasma spraying, sol-gel, electrochemical deposition and biomimetic coatings [20]. The biomimetic coating method is based on the nucleation and growth of bone-like hydroxyapatite on a metal, ceramic or polymer surface by immersing in a simulated body fluid (SBF). It produces a uniform and homogenous coating on various shapes of implant objects. Biomimetic coating on zirconia and titanium by applying a novel silane coupling agent is being developed (Figure 3) [21].
Future developments
The bioactive glass and hydroxyapatite used in implant coating have drawbacks such as brittleness, low mechanical strengths and bacterial infection of implants. Therefore, it is in need to modify these materials or search for other new biomaterials. Several strategies are explored: i. development of bioactive composite and hybrid coatings. Basically, the coating contains both inorganic (ceramic or glass) and organic (polymer) components which combines the advantages of both components; ii. incorporation of antibacterial materials such as silver and chitosan into the coatings; and iii. multifunctional coatings that combine multiple functional components. The future trend of implant coating design will not be only to improve osseointegration and prevent bacterial infection but also to extend the long-term implant performance.
References
- Brunette DM, Tengvall P, Textor M, Thomsen P, editors (2001) Titanium in medicine: material science, surface science, engineering, biological responses and medical applications. Berlin: Springer-Verlag; pp: 771-873.
- Piconi C, Maccauro G (1999) Zirconia as a ceramic biomaterial. Biomaterials 20: 1-25. [Crossref]
- Elias CN, Lima JHC, Valiev R, Meyers MA. Biomedical applications of titanium and its alloys. JOM 2008; 60: 46-49.
- Jemat A, Ghazali MJ, Razali M, Otsuka Y (2015) Surface Modifications and Their Effects on Titanium Dental Implants. Biomed Res Int 2015: 791725. [Crossref]
- Malkondu Ö, Tinastepe N, Akan E, Kazazoglu E (2016) An overview of monolith zirconia in dentistry. Biotechnol Biotechnol Equip 30: 1-9.
- Özkurt Z, Kazazoğlu E (2011) Zirconia dental implants: a literature review. J Oral Implantol 37: 367-376. [Crossref]
- Lung CY, Kukk E, Matinlinna JP (2012) Shear bond strength between resin and zirconia with two different silane blends. Acta Odontol Scand 70: 405-413. [Crossref]
- Lung CY, Matinlinna JP (2012) Aspects of silane coupling agents and surface conditioning in dentistry: an overview. Dent Mater 28: 467-477. [Crossref]
- Cattani Lorente M, Scherrer SS, Richard J, Demellayer R, Amez-Droz M, et al. (2010) Surface roughness and EDS characterization of a Y-TZP dental ceramic treated with the CoJetâ„¢ Sand. Dent Mater 26: 1035-1042. [Crossref]
- Lung CY, Kukk E, Matinlinna JP (2013) The effect of silica-coating by sol-gel process on resin-zirconia bonding. Dent Mater J 32: 165-172. [Crossref]
- Lung CY, Liu D, Matinlinna JP (2015) Silica coating of zirconia by silicon nitride hydrolysis on adhesion promotion of resin to zirconia. Mater Sci Eng C Mater Biol Appl 46: 103-110. [Crossref]
- Hench LL, Polak JM (2002) Third-generation biomedical materials. Science 295: 1014-1017. [Crossref]
- Omar S, Repp F, Desimone PM, Weinkamer R, Wagermaier W, et al. (2015) Sol-gel hybrid coatings with strontium-doped 45S5 glass particles for enhancing the performance of stainless steel implants: electrochemical, bioactive and in vivo response. J Non Cryst Solids 425: 1-10.
- Stanic V, Aldini NN, Fini M, Giavaresi G, Giardino R, et al. (2002) Osteointegration of bioactive glass-coated zirconia in healthy bone: an in vivo evaluation. Biomater 23: 3833-3841.
- Merolli A, Gabbi C, Santin M, Locardi B, Leali PT (2004) Bioactive glass coatings on Ti6Al4V promote the tight apposition of newly-formed bone in-vivo. Key Eng Mater 254-256: 789-792.
- Sola A, Bellucci D, Cannillo V, Cattini A (2011) Bioactive glass coatings: a review. Sur Eng 27: 560-572.
- Darimont GL, Cloots R, Heinen E, Seidel L, Legrand R (2002) In vivo behaviour of hydroxyapatite coatings on titanium implants: a quantitative study in the rabbit. Biomaterials 23: 2569-2575. [Crossref]
- Sörensen JH, Dürselen L, Welch K, Sörensen TC, Procter P, et al. (2015) Biomimetic hydroxyapatite coated titanium screws demonstrate rapid implant stabilization and safe removel in vivo. J Biomater Nanobiotechnol 6: 20-35.
- Wang H, Eliaz N, Xiang Z, Hsu HP, Spector M, et al. (2006) Early bone apposition in vivo on plasma-sprayed and electrochemically deposited hydroxyapatite coatings on titanium alloy. Biomater 27: 4192-4203.
- Surmenev RA, Surmeneva MA, Ivanova AA (2014) Significance of calcium phosphate coatings for the enhancement of new bone osteogenesis--a review. Acta Biomater 10: 557-579. [Crossref]
- Lung CYK. Biomimetic coating on zirconia and titanium implant modified with a novel silane coupling agent. Unpublished results.



