Abstract
Magnetoelastic (ME) biosensors were developed by immobilizing polyclonal antibody using Langmuir-Blodgett (LB) technique onto a magnetostrictive platform, which can offer a unique advantage of wireless and remote detecting over conventional sensor platforms. Due to the magnetoelastic nature of the amorphous magnetostrictive alloy, the sensor exhibits a physical resonance when it undergoes a time-varying magnetic field, a shift in resonance frequency occurs when its mass changes due to bacteria binding. In this study, the surface of the biosensors after binding different kinds of bacterial cells was observed by scanning electron microscopy (SEM), and the specificity of the ME biosensor was determined by evaluating the density of bacteria bound on the biosensor. The results show that the prepared polyclonal antibody-based biosensor presents excellent specificity to detect bacteria of Salmonella species, and especially prefers to bind Salmonella typhimurium, almost zero affinity for non-Salmonella species of Listeria monocytogenes by detecting solution with different kinds of bacteria, including Salmonella species and non-Salmonella species, and the bound bacterial cells were visibly confirmed.
Key words
magnetostrictive, biosensor, antibody, salmonella, specificity
Introduction
Magnetostrictive materials are made of amorphous ferromagnetic alloys composed of Iron, Nickel, Molybdenum and Boron. Due to the magnetoelastic nature of the amorphous alloy, the sensor exhibits a physical resonance when exposed to a longitudinal time-varying magnetic field and a DC biasing field. By employing a suitable bioprobe for detecting pathogens, a biosensor can be developed. This characteristic allows the biosensors to be monitored remotely without the use of direct physical connections, such as wires, enabling detection of environmental parameters from within sealed, opaque containers [1-3].
Antibodies are immune system-related proteins called immunoglobulins, which can selectively bind target antigen due to their special structure and different amino acid sequence [2-4]; hence, it was selected as the bioprobe in this research.
It is necessary for biosensors to have good specificity in practical applications. Therefore, an essential test for the characterization of any biosensor is to investigate the specificity of the bioprobe (the bio-molecular recognition element). Hence it is of important research value to investigate the specificity of the developed biosensors.
In this work, Polyclonal Antibody immobilized magnetostrictive biosensors with the size of 5mm x 1mm were prepared and the specificity of the magnetostrictive sensors for detecting different kinds of bacteria, including 8 kinds of Salmonella species and 1 kind of non-Salmonella species was studied.
Materials and methods
Materials
The magnetostrictive material used in this work was purchased commercially from Honeywell International Corporation (2826 MB); its composition is Fe40Ni38Mo4B18 with a thickness of about 30µm. The flat sensor samples were diced from this magnetostrictive material after dry polishing. Polyclonal Antibody used in this work was purchased from AbCam Company. All kinds of bacterial cells were prepared in the department of Nutrition and Food Science at Auburn University.
Sensor platform
The biosensor is comprised of two main parts: the first part is magnetostrictive substrate used as transducer; the second part is Polyclonal Antibody layer used as the bioprobe for binding bacterial cells, which was immobilized onto the sensor surface using the Langmuir—Blodgett (LB) method. The biosensor exhibits a physical resonance when it undergoes a time-varying magnetic field, thus emitting magnetic flux, this can be monitored remotely without the use of direct physical connections, due to the magnetoelastic nature of the amorphous magnetostrictive alloy [5]. Schematic illustration of the wireless nature of the magnetostrictive biosensors was shown in Figure 1.
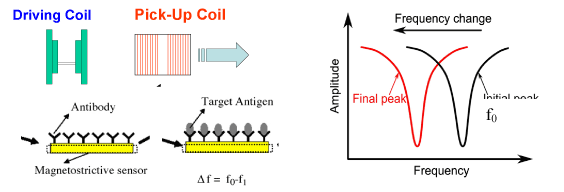
Figure 1. Schematic illustration of the wireless nature of the magnetostrictive biosensors and principle for bacteria detection
Experimental
After biosensors with the size of 5mm x 1mm were prepared, they were immersed in 1 ml solution of the following eight kinds of Salmonella species, S. typhimurium, S. heldelberg, S. typhi, S. eteritidis, S. mission, S. thompson and S. montevideo, S. panama, and one kind of non-Salmonella species of Listeria monocytogenes for 30 minutes. All the bacteria solutions were adjusted to the same concentration of 109 CFU/ml. Then the sensors were removed and exposed to Osmium tetra Oxide (OsO4) vapor for one hour to fix the bacterial cell’s wall. Finally, SEM images were taken using JEOL 7000F, operating at 5kV, which was used for investigating the physical distribution and density of the bacterial cells attached to the sensor surface.
Results and discussions
SEM images were taken for the biosensors to clearly show the binding of bacterial cells after exposing to different kinds of species with the same concentration of 109 CFU/ml. Figure 2 shows the typical SEM images for each kind of species, which clearly illustrates different density on the sensor surfaces, with highest binding density for S. typhimurium, and almost no binding for Listeria monocytogenes.
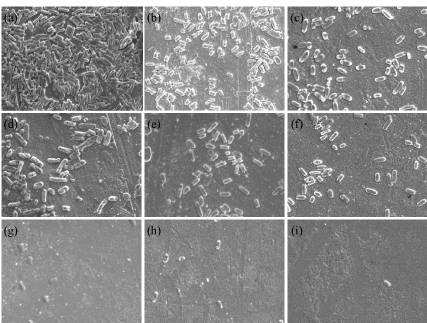
Figure 2. Typical SEM images of the biosensors after exposed to different kinds of bacteria with the same concentration of 109 CFU/ml (5mm x 1mm)
- S. typhimurium, (b) S. heldelberg, (c) S. typhi, (d) S. eteritidis, (e) S. mission, (f) S. thompson and (g) S. montevideo, (h) S. panama, and (i) Listeria monocytogenes, (a) ~ (h) represents bacteria of Salmonella species, (i) represents non-Salmonella species
Statistic density of the bacterial cells
In order to investigate the statistical density of the different kinds of bacterial cells, the number of cells bound on each sensor surface was counted from SEM images, and the actual density of bacterial cells were obtained by dividing the total number of cells by the area of the surface. Figure 3 shows the statistic density of the different kinds of bacterial cells attached to the polyclonal antibody immobilized sensor surface.
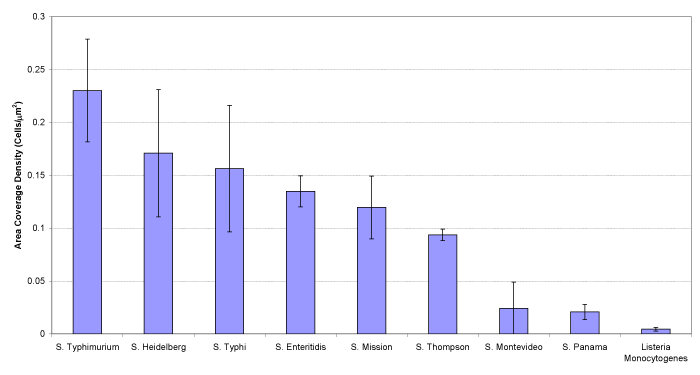
Figure 3. The statistic density of the bacterial cells attached to the polyclonal antibody immobilized sensor surface after exposed to different kinds of bacteria with the same concentration of 109 CFU/ml.
Figure 3 shows that the polyclonal antibody preferred to bind Salmonella typhimurium, though existed some affinity to other Salmonella species, but almost zero affinity was seen with Listeria monocytogenes. Thus, the conclusion could be drawn that the polyclonal antibody exhibited excellent specificity to Salmonella species bacteria.
Conclusions
Polyclonal antibody immobilized magnetostrictive biosensors were prepared with a unique advantage over conventionally used platforms, which needs no physical contact in order to obtain a response. The specificity to detect different kinds of bacteria, including Salmonella species and non-Salmonella species was studied. It was found that polyclonal antibody showed excellent specificity to detect bacteria of Salmonella species, and especially preferred to bind Salmonella typhimurium, almost zero affinity for non-Salmonella species of Listeria monocytogenes.
Acknowledgements
This project was supported by National Natural Science Foundation of China (51271039) and USDA Grant 2011-51181-30642A.
References
- Guntupalli R, Hu J, Lakshmanan RS, Huang TS, Barbaree JM, et al. (2007) A magnetoelastic resonance biosensor immobilized with polyclonal antibody for the detection of Salmonella typhimurium. Biosens Bioelectron 22: 1474–1479. [Crossref]
- Guntupalli R, Lakshmanan RS, Wan J (2008) Analytical performance and characterization of antibody immobilized magnetoelastic biosensors. Sens Instrum Food Qual 2: 27-33.
- Dadarwal R, Namvar A, Thomas DF, Hallc JC, Warriner K , et al. (2009) Organic conducting polymer electrode based sensors for detection of Salmonella infecting bacteriophages. Mater Sci Eng C 29: 761-765.
- Ye XM, Guntupalli R, Lakshmanan RS, Chin BA, Hu J (2014) Comparative study of thermal stability of magnetostrictive biosensors between two kinds of biorecognition elements. Mater Sci Eng C 41: 78–82. [Crossref]
- Liang C, Morshed S, Prorok BC (2007) Correction for longitudinal mode vibration in thin slender beams. Appl Phys Lett 90: 221912-1-221912-3.



