Abstract
Introduction: In Mexico over 35% of total medical consultations are register on younger than 3 years old, and are associated with Upper Respiratory Tract Viral Infections (URTVI), in average up to 14 episodes per child per year on general population and airway obstruction is frequent.
Methods and objective: The pilot study was an open and prospective clinical design to evaluate safety and efficacy of water particles (saline solution 0.9%) administration with a portable nebulizer NE-C801KD (COMPA-I-R®) to enhance secretions movement in paediatric out patients with non-complicated upper respiratory tract viral infections, patients with chronic pulmonary illness were excluded. For safety, evaluation at initial visit all caregivers were trained to operate the device and the patient diaries use to register adverse and/or incident event related with device use and symptoms. For efficacy, outcomes clinical evaluation and symptoms inventory were done and symptomatic treatment prescribed. Caregiver and investigator completed initial and end-of-study assessments a 7-day treatment period. Written informed consent was provided before starting the study. Descriptive statistics and differences between initial and final measurements were examined, p values of <0.05 were considered statistically significant.
Results: Thirty patients were included. The mean of age was 2.6 years old, there are ten (34%) males, and main diagnosis was rhinopharyngitis followed by rhinitis. Safety evaluation did not identify incident neither adverse event with device. Symptom inventory improve in 93% and the patient’s perception of wellbeing was towards an improvement at least 2 points at third day with a statistical significance of p 0.000 consistent amongst the physician and the caregiver. Only two patients needed a change or adding medication.
Conclusions: Portable nebulizer was safe there were not adverse/incident events neither device failure reported and patients showed symptoms improvement, decreasing respiratory malaise observed both physicians and caregivers with statistical significance.
Key words
saline solution 0.9%, acute upper respiratory tract viral infections, paediatric patients, safety, technovigilance, nebulizer, pilot study
Introduction
Upper respiratory tract viral infections (URTVI) are an important public health problem in paediatric patients, it is defined as an infectious disease that affects upper respiratory tract above the epiglottis, during a period of less than 15 days (1). It constitutes over 2 million medical consultations within the Health Secretary, and 35% of them occur in younger than a year of age, with a frequency of 3 to 7 episodes/year and up to 14 episodes per year per child in day care children age. Their incidence declines with age, as does their intensity and general compromise; but can be mild, moderate or severe; these are responsible for an important morbidity with probable long-term pulmonary damage in toddlers and younger than 5 years of age [1]. This is due to the calibre of the respiratory tracts at these ages, with makes it harder for secretions to be moved. The identification of these episodes in early stage, an adequate treatment and other general measures are important to resolve these events [2]. It has been reported that secretions management with saline solution is beneficial for healthy children as for those with pre-existent diseases [3]. Administration of water vapour is recommend to enhance the movement and expulsion of secretions and to provide a general wellbeing to patients [1].
Nebulizers are devices that transform liquids in aerosol to be able to inhale through a face mask or a mouthpiece. The European Society of Respiratory Diseases, ERS, recommend their use in diseases that require improvement in secretion movement [4]. Comfort and/or wellbeing of patients require ambulatory secretion movement within their households demands for use to enhance nebulizer´s use, as well as the identification and evaluation of incidents (technovigilance), to ensure that these devices fulfill their goal and to warrant quality, safety and efficiency [5]. The main objective is to explore safety (technovigilance) and efficacy of water particles (saline solution 0.9%) administration with a portable nebulizer NE-C801KD (COMPA-I-R®) to enhance secretions movement in paediatric out patients with non-complicated upper respiratory tract viral infections
Materials and methods
A prospective, observational clinical study was conducted. Consecutive paediatric patients with non-complicated upper respiratory tract viral infection diagnosis, any gender without chronic pulmonary or respiratory illness were included. Exclusion criteria included systemic chronic disease or history of asthma. Scientific and Ethical local Committee (16/04/2016) approved the protocol. All parents or legally authorized representatives were required to provide written informed consent before starting the study.
All patients received pharmacological treatment considered by physician’s election and non-pharmacological measures (nebulizing sessions with saline solution 0.9% using the nebulizer as part of the indications for a humid environment and secretion movement).
An inventory of symptoms was develop base on expert opinion to report patient´s diary evaluation by caregiver of everything observed during the initial visit with focus on clinical data related with secretions and general state of the patient. This diary patient’s is a questionnaire to document both medical and caregiver observations on general state and symptoms non-complicated upper respiratory tract viral infections related, as well as data on the usage, functioning and incidents with the device. The diary had two parts; the first for physician to recorder initial clinical data, inclusion criteria, treatment and date for next visit; the second for the care giver with open and closed questions about breathing, nasal obstruction, nasal discharge, cough, pain or irritation, sneezes, phlegm, hoarseness the measure was Likert scale (nothing, very little, Little, a lot, much, too much). Questions on treatment adherence or modification were included, as well as malaise perception evaluated on a numeric scale 0-10, where 0 is no malaise and 10 is a high malaise. It also had questions about any problems or incidents with the use of the nebulizer, the administration of a humid environment, a technical need, as well as perception on the facility of use. The final evaluation performed on day 7 (medical release) by the physician indicated an improvement rate on general patient´s state and symptoms diary based on the registry from the patient´s initial symptoms, and an interview was directed for adverse incidents or problems with the nebulizer. All received information, training for assembly, manipulation and use of the device [6] and diary report.
Statistical analysis: a descriptive analysis was performed (signs and symptoms of upper respiratory tract viral infections, patient status, incidents and satisfaction) Continuous variables were summarised by descriptive statistics (sample size, mean, SD or median). Categorical variables were summarized by frequency (N) and percentages (%). Due the study is exploratory nature, a before-after analysis was performed of the variables related to symptoms and perception. Changes in discomfort, symptoms and perception were analysed though time from day 1-7 of the caregiver. A statistical significance was set at p< 0.005 with a power of 80. We used Stata/IC 14 software.
Results
Thirty patients were included, twenty-nine completed the diary and attended the final evaluation; one patient lost follow-up. Safety evaluation not identified incidents/adverse events or device failure. They were 10 (34%) male and 19 (56%) female with an average age of 2.6 years (range 3 months- 13 years old); the most frequent initial diagnosis was rhinopharyngitis followed by rhinitis, bronchitis, etc. Table 1 shows the main demographic characteristics at the inclusion. Only two (7.41%) caregiver had previous nebulizer use experience.
Table 1. Demographic and clinical characteristics at inclusion (n=29)
Variables |
n(%) |
Female gender |
19(65) |
Mean age (median) months |
31.4 (14) |
Initial diagnosis: |
|
Viral rhinopharyngitis |
19(65) |
Rhinitis |
4(13.8) |
Sinusitis |
2(6.8) |
Other (pharyngitis, bronchitis, other) |
4(13.8) |
Basal clinical data about patient |
|
- Breaths by mouth |
22(81.4) |
-Has Nasal obstruction- A lot |
24(82.7) |
-Has Nasal discharge - A lot |
21(72.4) |
-Has Cough- Little |
23(79.3) |
-Sneezes- A lot |
10(34.4) |
The progressive decrease in malaise perception from caregiver evaluation is show in figure1. These values showed a statistical significance when comparing evaluation on day 1 vs day 3 (5.8 ± 2.7 vs 4.7 ± 2.2, p 0.001) and patient improvement is maintaining and increasing this significance until last day evaluated (Table 2).
Table 2. Patient symptom affection evaluation by physicians initial vs final
|
Initial n 29 |
Final n 29 |
Day 1 |
Day 7 |
|
Yes
(A lot) |
NO
(little or less) |
Yes
(A lot) |
NO
(little or less) |
The patient breath through the mouth* |
24 |
5 |
1 |
28 |
The patient has nasal obstruction * |
25 |
4 |
2 |
27 |
The patient has nasal discharge * |
21 |
7 |
1 |
28 |
The patient has cough |
6 |
23 |
1 |
28 |
The patients manifests pain or irritation (nasal, throat etc.) |
3 |
26 |
1 |
28 |
The patient sneezes * |
10 |
19 |
1 |
28 |
2021 Copyright OAT. All rights reserv
The patients has phlegm |
12 |
17 |
1 |
28 |
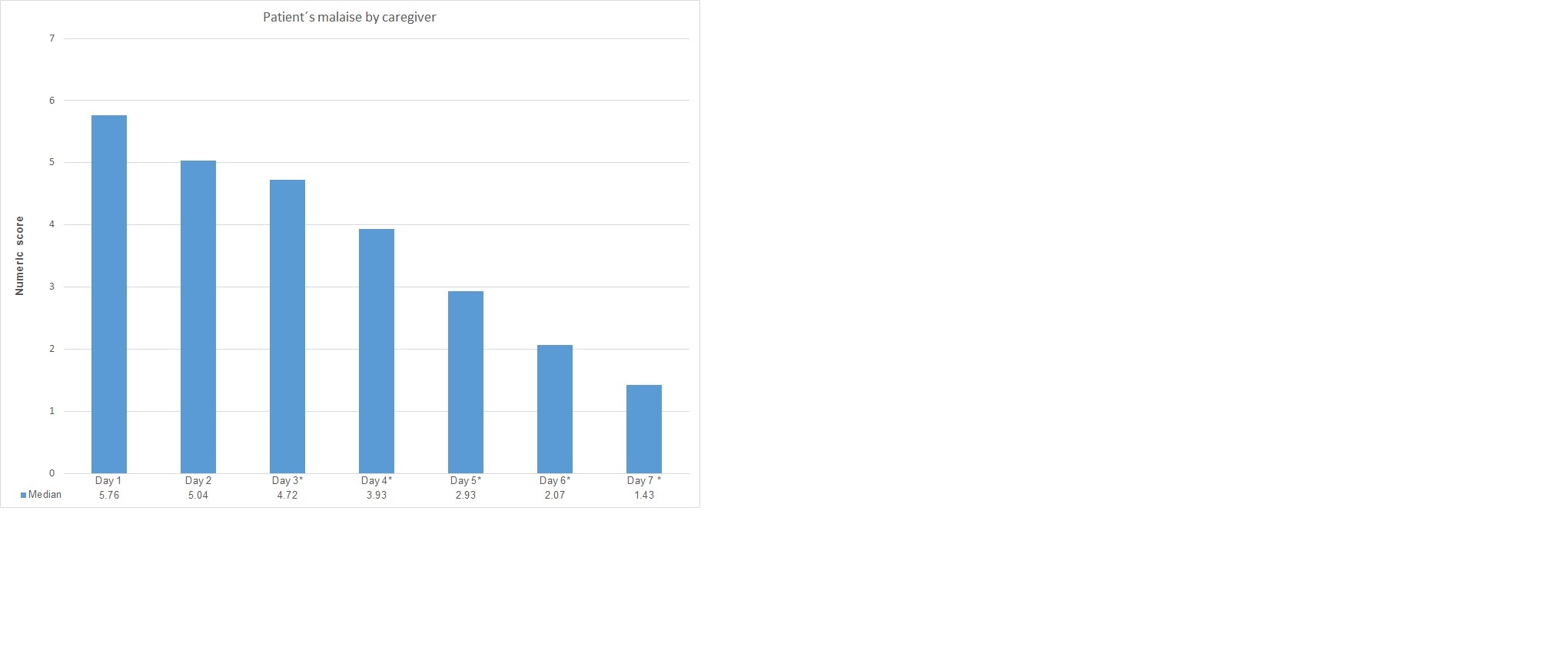
Figure 1. Diary score for malaise perception from a caregiver evaluation
*Chi square test and fisher exact test if is indicated p < 0.05
This evaluation had consistence with paediatric perception on the malaise scale evaluated by them on day 1 compared to final evaluation the mean differences was statistically significant (5.76 vs 1.18) p = 0.0000. (Figure 2), this improve in perceptions is related with decrease in symptoms 27(93.2%) patients improve in all symptoms at final evaluation; the symptoms associated with secretions as nasal obstruction or discharge and breath through the mouth had higher improvement from a lot to little.

Figure 2. Mean value + standard deviation of a numeric scale evaluation malaise from physician evaluation
Patients received symptomatic treatment with antihistaminic and acetaminophen, and only two patients required change or additional treatment (one needed nasal steroid for allergic rhinitis and the other budesonide).
Satisfaction perception is show on Figure 3. Likert scale score by the caregiver did evaluation during the final evaluation phase. The “low satisfaction” was related with the illness’ evolution itself.

Figure 3. Satisfaction perception on the nebulizer use
Figure 3 represents the caregiver criteria where they show a 96.2% high satisfaction rate with the nebulizer as part of the treatment, without indicating any desertion due to mechanical or technical functionality or deterioration of the device. With this, we can say that the nebulizer is safe and effective to support management of secretion accumulation.
Discussion and conclusions
The upper respiratory tract viral infection had wide and unspecific natural history, in general most symptoms lasted for 5-11 days, but this time could be longer; management guidelines recommend secretion management with a knob and other mechanical alternatives such as the headboard use or acquiring positions that favour drainage. Warm water vapour is discouraged because of the risk of burns, and the use of humidifiers has been associated with secondary infections through colonization of the devices. Nevertheless, this pilot study showed that a correct training, allows the correct functioning, use and hygiene to the caregivers. The device portability is on favour to comfort for the procedure to contribute to maintain the natural history of the disease without complications, avoiding unnecessary risks for the patients. Secretion movement was adequate, and this has special importance when preventing complications and bacterial over infection for this viral cases.
Literature review on phase 4 technovigilance is few, and authors did not find any published work for this scenario. Documented results on this pilot study show that the nebulizer NE-C801KD COMPA-I-R ® is efficient and safe on patients included in this study.
Documented safety of the device for ambulatory use is important because it allows for a safe alternative, easy to handle and keep clean, and favours recovery of these events without further complications. In this exploration of the safety of domestic use, no adverse incidents were reported.
The secretion management is done with the knowledge that the main reason why upper respiratory tract viral infections can generate more problems. This is because inflammation favours mucus production, making it difficult to breath via obstructing nasal fossa, which obliges children to initiate oral breathing; so the physiological effect of nasal breathing, which is to warm and humidify air, is avoid.
Treatment with a portable nebulizer is safe and effective for secretion movement in non-complicated upper tract respiratory viral infections with a high rate of improvement within the first 3 days on malaise reported by caregiver and physicians with a statistical significance. No treatment change was necessary and no anomalies with device function, nor difficulties with handling it or any adverse events/incidents reported.
Acknowledgements
Authors appreciate the logistic support to Carolina Goycochea. HS estudios farmacoeconomicos staff.
Funding
Authors appreciate the support of Omron Healthcare Mexico S.A. de C.V for a non-restricted grant for publication.
References
- CENETEC SN (2009) Diagnóstico y Manejo de la Infeccion Aguda de Vías Aéreas Superiores en Pacientes Mayores de 3 Meses hasta 18 años de Edad. Guía de Práctica clínica: 19.
- Guiscafré-Gallardo H (2008) La atención integral del niño con infección respiratoria aguda:lo apropiado y lo actual. Bol Med Hosp Infant Mex: 249-260.
- Daviskas E1, Anderson SD, Gonda I, Eberl S, Meikle S, et al. (1996) Inhalation of hypertonic saline aerosol enhances mucociliary clearance in asthmatic and healthy subjects. Eur Respir J 9: 725-732. [Crossref]
- Boe J, Dennis JH, O'Driscoll BR, Bauer TT, Carone M, et al. (2001) European Respiratory Society Guidelines on the use of nebulizers. Eur Respir J 18: 228-242. [Crossref]
- Comision Federal para la Protección contra Riesgos Sanitarios (2012) Norma Oficial Mexicana NOM-240-SSA1-2012, Instalación y operación de la tecnovigilancia. Diario Oficial Federal.
- HEALTHCARE, Omron (2011) Manual de instrucciones Nebulizador NE-C801KD.



