Abstract
Combining two sets of classic, physiologic studies published between 1945 and 1953, with some recent observations, offers a new view of pre-eclampsia (the “uterine denervation” view). In 1945, Dr AT Hertig, (Boston, MA) described narrowing of uterine spiral arterioles, associated with irregular hyperplasia of their vessel walls in the placental bed of women with pre-eclampsia. His illustrations show a “halo” of hyalinised cells around the injured arterioles that has been largely undiscussed in the literature. In many gynecologic syndromes, concentric layers of abnormal, injured nerves surround similarly-narrowed arterioles. There is no “halo” of injured nerves in the “obstetric lesion” because the injured nerves, unlike the narrowed spiral arterioles, cannot extend to the placental bed in pregnancy.
In 1953, KJ Franklin and GJ Sophian (London, UK) published results of experiments distending balloons in rabbit uteri where they observed immediate blanching of the kidneys followed by oliguria. Denervating the kidneys abolished the “uterorenal reflex”; persistent distension of the uterus led to acute renal failure. These experiments demonstrated direct neural connections between the uterus and kidneys that caused redistribution of blood flow from the renal cortex to the medulla that have been confirmed in a series of contemporary renal, Doppler studies.
The “uterine denervation” view of preeclampsia proposes that stretching injured myometrium and arterioles expressing “new” purinergic (P2X3) “stretch” receptors, activates the uterorenal reflex, redistributing renal blood flow from cortex to medulla, activating the renin-angiotensin system, resulting in hypertension, and, proteinuria. Delivery of the baby relieves the “stretch” leading to immediate resolution of the pre-eclamptic syndromes. Exposing pregnant women to sustained levels of hypertension may injure renal vasomotor nerves contributing to early-onset (aged 40-50) hypertension and cardiovascular events following pregnancies complicated by pre-eclampsia. Exposing babies to sustained hypertension in utero may cause injuries to their kidneys resulting in childhood hypertension and its sequelae (Fig.6).
Key Words
pre-eclampsia, hypertension, arteriolitis, perivascular nerve fibre proliferation, uterorenal nerves, corticomedullary shunt
Reinterpreting two strands of historical research provides a new framework for understanding pre-eclampsia that explains important clinical aspects of the condition that have not been settled by prior studies [1]. The first, is the histologic observation that there is a partial, or complete, “halo of hyalinised cells” (“empty” cells) around narrowed uterine arterioles in almost all accounts of the histology of the placental bed (Figure 1a) [2,3]. Not only is this histologic feature rarely described; it is not often discussed in contemporary research. However, Professor Brosens (Leuven, Belgium) has collected examples of this lesion in all of the “great” obstetric syndromes including preeclampsia, intrauterine growth retardation, preterm labor, midtrimester loss, placental abruption, etc. confirming that it is not specific to any particular condition [4]. It is an historic, non-specific finding that, in this account, reflects some degree of underlying neural injury to the lower genital tract that is common to the obstetric syndromes BUT also to many gynecologic syndrome (Figures 1a-d; 2a-f) [3].
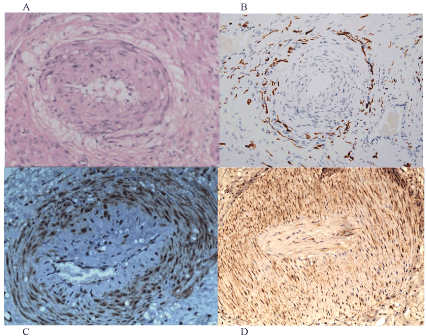
Figure 1. Injured uterine arterioles in different clinical syndromes
Narrowed uterine arteriole in a pregnant uterus (A, the “obstetric lesion”) demonstrates a “halo of hyalinised cells” around the circumference of an injured arteriole with irregular hyperplasia of the vessel wall, whereas narrowed uterine arterioles in a non-pregnant uterus (B, the “gynecologic lesion”) demonstrates a corresponding “halo of injured nerves” around the circumference of a narrowed arteriole with irregular hyperplasia of the tunica media and intima.
Injured nerves cannot extend from the isthmus of the uterus (B) to the placental bed (A) during pregnancy accounting for these similar appearances. Figure 1C demonstrates purinergic, “P2X3”, stretch receptors (brown) that may be found in arteriolar walls and injured myometrium, as may VEGF receptors (brown) in Figure 1D.
The second strand of historical research, is a series of animal experiments conducted in Oxford and London, from 1945-1953 by an English physiologist, Greek gynaecologist, and, Spanish orthopaedic surgeon [5,6]. They demonstrated autonomic neural connections between the uterus and the kidneys (the “uterorenal reflex”, Figure 3) [5], that caused redistribution of renal blood flow from the cortex to medulla (“the Oxford shunt”, Figure 4) [6]. Recent, insights in autonomic pathophysiology suggest that injured uterine, autonomic nerves induce primitive, “stretch” receptors that trigger the uterorenal reflex, and, redistribution of renal blood flow in the “uterine denervation” view of pre-eclampsia [7]. This view provides an explanation for the gradual development of hypertension and proteinuria in the second half of pregnancy as the uterus increases in size, and, its prompt resolution with removal of “stretch” associated with delivery of the fetus and placenta. These are daily clinical experiences of preeclampsia that are not fully explained by earlier hypotheses.
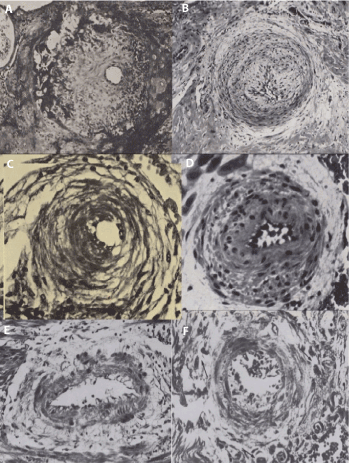
Figure 3. . Peri-arteriolar “halos of hyalinisation” in different accounts of pre-eclampsia (1945-1972
The original description of thickening of arteriolar walls in different obstetric syndromes (A, Hertig, 1945), demonstrates a partial halo of “empty” hyalinised, cells (9-12 o’clock). Complete “halos of hyalinisation” around thickened arterioles in subsequent studies (B, Zeek & Assali, 1951), (C, Dixon & Robertson, 1958), (D, Brosens, 1962), (E,F Pijnenborg, 1972). Compare with the “halos of injured nerves” in thickened arterioles in gynecologic syndromes (Fig 1b).
Finally, contemporary studies demonstrate new, purinergic, P2X3, “stretch” receptors in injured arterioles and myometrium in the lower half of uteri in pregnancy hysterectomy specimens (Figure 1c and Figures 5a-d) [1]. These “purinergic” receptors become active after various pre-pregnancy injuries to intrauterine nerves e.g. physical efforts during defecation, treatment of miscarriages and terminations, etc. They are found in myometrium (“late-onset”, preeclampsia with AGA fetus) (Figure 5b) and in the walls of narrowed arterioles (“early-onset preeclampsia with SGA fetus) (Figure 1c). Stretching injured tissues e.g. nulliparous pregnancy, hydramnios, multiple pregnancy, molar pregnancy, etc. releases adenosine triphosphate (ATP) that is the neurotransmitter at these “stretch” receptors leading to activation of uterorenal nerves, redistribution of renal blood flow, activation of the renin-angiotensin system, hypertension and proteinuria [7]. Removal of the “stretch” by amniotomy, or delivery, reverses these physiologic changes [8,9]. We term this the “uterine denervation” view of preeclampsia that was first articulated by Professor Otto Speigelberg, Breslau, 1882 [8], and, developed by Mr John Sophian, London, 1953 [5]. Our group have added the intrauterine sensory mechanism i.e. the purinergic, P2X3 receptors to assemble the completed pathway [1,7].
Uterine nerves
Robert Lee, FRS, published an account of the anatomy of the uterine nerves, preserved and prosected, in alcohol at St Georges Hospital, London in 1841 [10] (Figure 3). Since 1945, many medical schools preserved cadaveric material in formalin to extend its educational life; however, formalin destroys fine branches of autonomic nerves. As a result many colleagues have not seen the full morphology of the branches of the pelvic plexi in fresh cadaveric material (Figure 3). Typically, uterine, sympathetic nerves (T10-L2) run through the superior and inferior hypogastric plexi to reach the uterovaginal plexus (Lee-Frankenhauser plexus). KE Krantz (Kansas City, KS) described nerve plexi in the subserosal and endometrial-myometrial layers that receive nerves running with the uterine artery, and, through the uterosacral ligaments, respectively [11]. Parasympathetic fibres from sacral segments (S2-4) follow similar courses. An ovarian contribution to the distal third of the Fallopian tube runs through the free edge of the mesosalpinx, and, mesosalpingeal branches adjacent to the uterosacral bundle to supply the outer longitudinal layer of smooth muscle of the isthmus of the Fallopian tube [12].
Causes of injuries to uterine nerves
There are a number of distinctive sources of injury to uterine nerves: (1) difficult first labours, (2) persistent physical efforts during defaecation, (3) gynaecological surgery, particularly medical and surgical procedures to evacuate the uterus, (4) hypertension that disrupts vasomotor nerves, (5) miscellaneous conditions e.g. traumatic injuries to the pelvis [13,14]. In China, recurrent and late, abortions cause widespread injuries that result in infertility and ectopic pregnancy in women in their 20’s , and, advanced painless adenomyosis in their 40’s [11]. Persistent physical efforts during defecation are endemic in Western populations (20-30%, 23) [13]. One per cent (1%) of women only achieve defecation once per week; 0.1% once per month. The typical histologic lesion is perivascular nerve fiber proliferation (Figure 1b) where there are large numbers of injured nerves around thickened arterioles that are the basis for many premenstrual syndromes (Figure 1b) [13,14]. In the context of pre-eclampsia, physical efforts during defaecation in Western countries, and, recurrent or “late” (>16 weeks) terminations of pregnancy in developing countries, may be important in causing injuries to nulliparous, uterine nerves that leads to the induction of purinergic, P2X3, “stretch” receptors to provide an alternative form of intercellular communication [7,13,14].
Consequences of injuries to uterine nerves
Traumatic injuries to nerve bundles lead to a predictable sequence of molecular and cellular responses in different clinical syndromes including chronic pelvic pain, menstrual disorders, vaginal and vulval pain, irritative bladder and result in new nerves regenerating from the proximal stump to attempt to restore function [15]. Cellular reorganisations include the development of growth cones at the tip of injured axons, swelling of neuronal cells with marked increases in protein synthesis, and, the dispersion of rough endoplasmic reticulum throughout the neuronal cytoplasm. The ensuing, reactive “soup” of cytokines and growth factors may have an impact on both nerve bundles and adjacent tissues. In the context of pre-eclampsia, injuries to uterine nerves lead to hyperplasia of the regenerating nerves from the proximal stump of the nerve, and, hyperplasia of the now-denervated arteriolar walls appearing as irregular hyperplasia of the tunica media and intima (Figure 1b, the “gynecologic” lesion) [3]. This lesion appears in all pelvic organs and fascial supports in many gynecologic syndromes [13,14]. Injured arterioles can extend to the placental bed during pregnancy, though injured nerves cannot, leading to the “halo of hyalinisation”, or, “empty” cells, where the injured nerves had been originally (Figure 1a; 2a-f). Importantly, these injured uterine nerves differ in both structure and function from the original innervation. Injuring nerves leads to induction of purinergic (P2X3) receptors that respond to “stretch”, and, TRPV-1 receptors that respond to pain e.g in endometriosis and adenomyosis (Figure 1c-d) [16].
Narrowing of uterine arterioles
In 1945, Dr Arthur T.Hertig (Boston, MA) published illustrations of narrowing of the uterine spiral arterioles in pre-eclampsia within a review of “The Toxemias of Pregnancy” by Dr Foster S. Kellogg (Boston, MA) [17,18].Low and high-power photographs show incomplete “halos” of empty cells around the circumference of the vessel (Figure 2a). In 1951, Drs Pearl M. Zeek and Norman S. Assali, (Cincinnati, OH) developing the view that uterine ischemia is the important feature in eclamptogenic toxaemia, published similar illustrations demonstrating narrowing of uterine arterioles with similar “halos of hyalinisation” around the vessel (Figure 2b) [19]. In 1958, Drs HG Dixon and WB Robertson, using the innovative technique of placental bed biopsy in 36 women in the West Indies, coined the term “acute arteriolitis” though did not substantiate the findings of “acute atherosis” described by Hertig, Zeek and Assali [20].Instead, they ascribed the vascular changes to pre-existing hypertension. They, too, included illustrations of the “halo of hyalinisation” (Figures 2c-d). Almost all, subsequent authors, including Professor Brosens and Pijnenborg (Leuven, Belgium) have produced similar illustrations commenting on the different, degenerative patterns in the vessel walls but not on the partial, or complete, “halo” of hyalinised cells (Figures 2e-f) [21,22]. Clearly, these illustrative images may be subject to cutting, staining and selection bias nevertheless they are a consistent feature of most published reports of placental bed biopsy in pre-eclampsia. However, these authors were unaware of the characteristic features of perivascular nerve fibre proliferation in almost all gynecologic syndromes that are also consistently associated with narrowing of arterioles [13,14].Apposing the two lesions is persuasive (Figures 1a-b) though further studies of these injuries will be helpful [2,3].
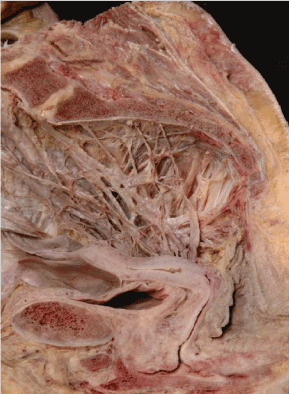
Figure 2. The pelvic autonomic nerves prepared in alcohol (not formalin)
The pelvic autonomic plexi and nerves in the right hemipelvis of a nulliparous cadaver embalmed in alcohol (not formalin) demonstrating the superior and inferior hypogastric plexi converging on the uterovaginal plexus of Frankenhauser adjacent to the vaginal vault. (Dissected by Ross Spackman, and Alice Roberts after preparation in an alcohol-based, embalming solution by Bill Wrigley, University of Bristol, 2002) [40].
The “uterorenal reflex” and the “Oxford shunt”
Between 1945 and 1953, Professor KJ Franklins’ laboratory in Oxford, and, then in London, performed a number of animal experiments (i) looking at the effects of “crush” syndromes with Josep Trueta, an orthopaedic surgeon (1945-1947), and, (ii) distending balloons in rabbit uteri with Dr John Sophian, a Greek gynaecologist (1949-1953) [5,6]. Trueta, and his multidisciplinary team, applied tourniquets to animal limbs resulting in ischaemia of the limb; release of the tourniquet led to temporary improvement followed by progressive renal impairment with death one week later. Cine-radiography of the renal circulation demonstrated a vascular shunt from the renal cortex to the medulla (Figures 4a-b). Other visceral reflexes activated this shunt indicating that the glomerular circulation is subject to direct regulation by the central nervous system through the sympathetic nervous system. The “Oxford shunt” provides a common pathway to acute renal failure through several different mechanisms.
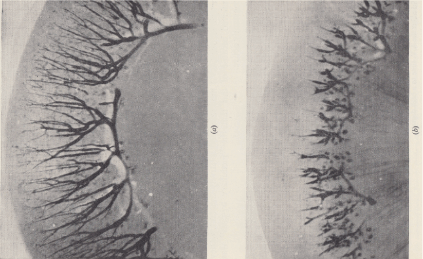
Figure 4. The renal, corticomedullary “shunt” redistributes blood flow from the cortex to the medulla in both rabbits [5,6] and humans [23].
The renal cortico-medullary shunt was discovered in the laboratory of Prof KJ Franklin by Joseph Trueta and John Sophian through cine-radiography of rabbit kidneys. Normal blood flow in the renal cortex is shown in the left figure, A, though this was markedly reduced (right figure, B) following inflation of a balloon in the uterus. Trueta was interested in the remote consequences of blast injuries and limb fractures, whereas Sophian made the analogy with renal function in preeclampsia. Contemporary Doppler studies have confirmed the changes in blood flow [23].
In a subset of these experiments, GJ Sophian distended balloons in rabbit uteri resulting in immediate blanching of the kidneys with oliguria [5]. Denervating the kidneys abolished the uterorenal reflex; persistent distension caused acute renal failure. They had demonstrated specific neural connections between the uterus and kidneys as one of a number of viscero-visceral reflexes in different abdominal organs (cf cardiorenal, hepatorenal, lienorenal connections). Contemporary Doppler studies confirm accompanying changes in waveforms in the renal circulation in pre-eclampsia [23]. Sophian, was quite clear that the uterorenal reflex, causing redistribution of renal blood flow, was the “cause” of pre-eclampsia, though KJ Franklin, recognised the important absence of an afferent, intrauterine pathway [24]. Sophians’ views did not gain traction in the 1950’s because of the opposing opinions of GW Theobald, FRCOG [25], a transatlantic dispute about the mechanism of the “Oxford shunt” with Dr HW. Smith (Boston, NY) [26], and, recent sets of histologic observations re-emphasising the placental ischaemia view [27]. Dr EW Page (UCSF, CA) referred briefly, to Sophian’s work in the conclusion of his RCOG, Green-Armytage Lecture in 1972 [27] though Dr LC Chesley (SUNY, NY) “resuscitated” both the “uterorenal reflex” and the “Oxford shunt” in the first edition of his classic textbook in 1978 [28]. By the 4th edition they had “re-disappeared” [29].
The “uterine denervation” view of pre-eclampsia
Stretching injured intrauterine tissues activates novel purinergic, (P2X3), “stretch” receptors in (i) narrowed uterine arterioles (“early-onset” preeclampsia), (Figure 1c), and, (ii) in the myometrium (“late-onset” pre-eclampsia), (Figure 5b) that, in turn, triggers the uterorenal reflex leading to diversion of renal blood flow from the cortex to medulla resulting in the classical signs of hypertension and proteinuria [7]. In early-onset, pre-eclampsia syndrome, narrowed uterine arterioles in the placental bed cause reduced uteroplacental blood flow that may result in a small-for-gestational-age fetus. Relief of “stretch” by delivery or amniotomy, as originally recorded by Professor Otto Spiegelberg [8], (Breslau, Germany) and some decades later by Professor HJ Drew-Smythe [9] (Bristol, UK) results in rapid improvement of clinical symptoms and signs. There are no detailed, quantitative studies of the effects of amniotomy on maternal blood pressure in the pre-eclamptic syndromes though we know it is an important feature of the initial management of placental abruption to prevent the adverse renal effects of hypovolaemia and the uterorenal reflex. By necessity, puerperal eclamptic convulsions on days 5-6 post-delivery, are not the result of intrauterine stretch, but occur for other reasons that are yet to be established. Spiegelberg (1882) had set out a prior version of the “uterine reinnervation view” whilst continuing to contend that toxins from placental infarcts were the primary source of the condition:
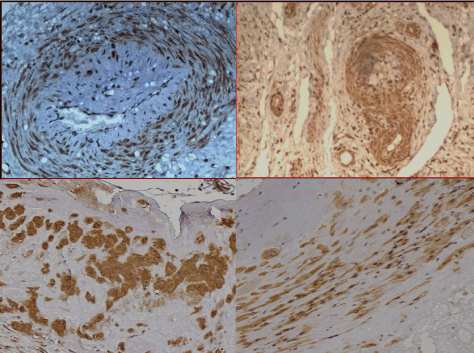
Figure 5. Purinergic, P2X3, “stretch” mechanoreceptors in the lower half of the uterus in pregnancy hysterectomy specimens
Purinergic, P2X3, “stretch” receptors (brown) in the walls of different sizes of arterioles (Figs. 5a-b) with similar receptors in different structural elements (Fig 5c-d) in the lower half of the uterus of pregnancy hysterectomy specimens (stained with anti-P2X3, x100-400).
In the “uterine denervation” view of preeclampsia, stretching injured arterioles and myometrium leads to release of adenosine triphosphate (ATP) with activation of uterorenal reflexes that create a renal corticomedullary shunt, that activates the renin-angiotensin system, resulting in “early-onset” (injured arterioles) and “late-onset” (injured myometrium or structural elements) preeclampsia.
”It is not improbable that the vasomotor irritation proceeds from the uterus; indeed the association of some special irritation of the sensory nerves with the convulsions, has been frequently and properly pointed out; and if we suppose that the vasomotor nerves of the kidneys, which run in the splanchnic nerve, can be irritated in a reflex manner by the uterus.”
Otto Speigelberg, Textbook of Midwifery, 2nd edition, 1882
Since the second edition of Speigelberg’s textbook in 1882 there have been few forays into neurologic explanations of the pre-eclamptic syndromes until Franklin, Sophian & Trueta’s experiments between 1945 and 1953. Fortunately, Dr LC Chesley’s scholarship in compiling the first edition of his textbook on “Hypertensive Diseases in Pregnancy”, gathered all these studies together in one place [28]. We add direct evidence for purinergic, P2X3 “stretch” receptors in uterine arterioles and myometrium [1,7] as well as radiologic evidence for changes in renal blood flow [23]. Contemporary research in preeclampsia focuses on a range of soluble proteins in the maternal blood though we know that stretching injured tissues does result in the release of such proteins. Stretching also releases adenosine triphosphate, which is integral to the “uterine denervation” hypothesis because it is the neurotransmitter in purinergic mechanosensory transduction i.e stretching injured tissues results in transmission through the purinergic signalling system instead of the injured nervous system [7,30].
Postpartum hypertension
In 1995, DJP Barker published studies connecting low birth weights with later hypertension in well-documented clinical records from small towns in the UK in the 1920-30’s [31]. Low birthweight babies tended to be hypertensive in early adulthood compared to mean birthweight babies with an increased risk of cardiovascular events such as myocardial infarction and stroke in their 40-50’s. Dr Barker advanced the view that adverse, in utero, environments contributed to these findings. Our group now describes histologic evidence of in utero hypertension in the proximal ten centimetres of the umbilical cord where the umbilical arteries demonstrates varying degrees of (a) hyperplasia of the tunica media, (b) disruption of the tunica adventitia, (c) perivascular reinnervation in some cases. These may include asymptomatic, anxious, diabetic, hypertensive, IUGR, oligohymnios and preeclamptic women that matches some of Barker’s data. The precise mechanisms are not yet clear but in some cases of reducing amniotic volumes it is possible that these may result from reductions in fetal urine output owing to renal dysfunction. Outcomes may include childhood hypertension, “essential” hypertension that is discovered in pregnancy, preeclampsia and early-onset” hypertension (aged 40-50) [31].
In adult hypertension we observe similar lesions in most visceral circulations including kidney, spleen, adrenal, pancreas and liver to those in the uterus (Figure 1b) though it is only the uterine neurovascular lesion that appears to stain positive for P2X3, purinergic receptors (Figure 1c) [32,33]. Visceral arteriolar stenosis was originally described by Drs Alan Moritz and Mary Oldt in Cleveland, OH, in 1937 [34] though its origins in injuries to vasomotor nerves have only recently been described [32,33]. Injuries to visceral, vasomotor nerves release cytokines and growth factors that act on denervated arteriolar walls to produce mural hyperplasia and arteriolar stenosis in similar fashion to that in the uterus and umbilical cord. At this stage we believe that there are multiple, layered sources of hypertension that commence in utero, that may be exacerbated by pregnancy, and, result in early-onset, cardiovascular events (strokes and heart attacks) that are preventable. The inter-generational spiral of possible injuries to maternal and fetal renal function clearly needs to be interrupted to prevent “transmission” of a mother’s hypertension to her child before it has taken its first breath?
Conclusions
Forgotten, physiologic experiments conducted between 1945 and 1953 are pivotal to an understanding of the “uterine denervation” view of pre-eclampsia. Demonstrating uterorenal neural connections and redistribution of renal blood flow associated with distension of the uterus is a powerful, experimental model that may account for many of the clinical phenomena in pre-eclampsia. Testing individual elements of this hypothesis, may add persuasive evidence. Does amniotomy reduce maternal blood pressure in the different types of pre-eclamptic syndromes, and, to what extent? What are the structure and properties of intrauterine “stretch” receptors? What other viscero-visceral reflexes operate in the complications of pre-eclampsia e.g. HELLP syndrome? What are the relationships between renal nerves and arterioles in hypertensive patients? Adopting the “uterine denervation” view as a tentative, etiological framework for pre-eclampsia, may assist in answering some of these questions.
Combining these physiologic observations with new explanations of narrowing of uterine arterioles, and, purinergic, P2X3, ‘stretch” receptors provides a revised view of the preeclamptic syndromes that may require changes in clinical management to protect both fetal and maternal renal function. Clearly, further studies are necessary to “fill in the details” of this hypothesis but it is a theme that runs through 150 years of research into preeclampsia since Otto Speigelberg first presented it in 1882, and, John Sophian further developed it in the 1950’s. This story is not yet over.
References
- Wu XQ, Xu XX, Zhou ZY, Quinn MJ (2018) The arteriolar injury in pre-eclampsia. BJOG 125: 511-512.
- Alnaes-Katjavivi P, Lyall F, Roald B, Redman CW, Staff AC (2016) Acute atherosis in vacuum suction biopsies of decidua basalis. Placenta 37: 26-33. [Crossref]
- Quinn MJ (2016) The aetiology of narrowed uterine arterioles in obstetric and gynaecological syndromes. Placenta 44: 114. [Crossref]
- Brosens I, Pijnenborg R, Vercruysse L, Romero R (2011) The "Great Obstetrical Syndromes" are associated with disorders of deep placentation. Am J Obstet Gynecol 204:193-201. [Crossref]
- Sophian GJ. Toxaemias of Pregnancy. Butterworth & Co. Ltd., 1953.
- Trueta J, Barclay AE (1948) Studies of the Renal Circulation. Published by Charles C. Thomas Springfield.
- Wu XQ, Cai YY, Xia WT, Quinn MJ (2016) The aetiology of pre-eclampsia, 1945-1953. BJOG 123: 2130. [Crossref]
- Speigelberg O. Lehrbuch für Geburtshilfe “A Textbook of Midwifery”, Second Edition, 1882. Translated by Dr Jamieson B.Hurry (Reading, Berkshire) pp 215.
- Drew Smythe HJ (1949) Surgical induction of labour. J Obstet Gynaecol Br Common 56: 431-438.
- Lee (1841) On the nervous ganglia of the uterus. Phil Trans 131: 269-275.
- Krantz KE (1959) Innervation of the human uterus. Ann N Y Acad Sci 75: 770-784. [Crossref]
- Zhang XM, Huang X, Xu H, Quinn MJ (2014) Altered innervation of the fallopian tube in ectopic pregnancy. J Obstet Gynaecol 34: 531-532. [Crossref]
- Quinn MJ (2011) Endometriosis: the consequence of uterine denervation-reinnervation. Arch Gynecol Obstet 284: 1423-1429. [Crossref]
- Quinn MJ (2014) Autonomic denervation and Western diseases. Am J Med 127: 3-4. [Crossref]
- Makwana M, Raivich G (2005) Molecular mechanisms in successful peripheral regeneration. FEBS J 272: 2628-2638. [Crossref]
- Cho T, Chaban VV (2012) Expression of P2X3 and TRPV1 receptors in primary sensory neurons from estrogen receptors-α and estrogen receptor-β knockout mice. Neuroreport 23: 530-534. [Crossref]
- Hertig AT (1945) Vascular pathology in the hypertensive, albuminuric toxemias of pregnancy. Clinics 4: 602-613.
- Kellogg FS (1945) Toxemias of pregnancy. Clinics 4: 585-648.
- Zeek PM, Assali NS (1950) Vascular changes in the decidua associated with eclamptogenic toxemia of pregnancy. Am J Clin Pathol 20: 1099-1109. [Crossref]
- Dixon HG, Robertson WB (1958) A study of the vessels of the placental bed in normotensive and hypertensive women. J Obstet Gynaecol Br Emp 65: 803-809. [Crossref]
- Brosens I, Robertson WB, Dixon HG (1967) The physiological response of the vessels of the placental bed to normal pregnancy. J Pathol Bacteriol 93: 569-579. [Crossref]
- Pijnenborg R, Bland JM, Robertson WB, Brosens I (1983) Uteroplacental arterial changes related to interstitial trophoblast migration in early human pregnancy. Placenta 4: 397-414. [Crossref]
- Miyake H, Nakai A, Koshino T, Araki T (2001) Doppler velocimetry of maternal renal circulation in pregnancy-induced hypertension. J Clin Ultrasound 29: 449-455. [Crossref]
- Franklin KJ, Winstone NE (1955) A review of some experimental findings of obstetrical interest. J Obstet Gynaecol Br Emp 62: 29-36. [Crossref]
- GW Theobald (1955) The Pregnancy Toxaemias. London: Henry Kimpton.
- Smith HW, Finkelstein N, Aliminosa L, Crawford B, Graber M (1945) The renal clearances of substituted hippuric acid derivatives and other aromatic acids in dog and man. J Clin Invest 24: 388-404. [Crossref]
- EW Page (1972) On the pathogenesis of pre-eclampsia and eclampsia. J Obstet Gynaecol Br Commonw 79: 883-894.
- Chesley LC. Hypertensive Disorders in Pregnancy. Appleton-Century-Crofts, New York, 1978 (First Edition).
- Taylor RN, Roberts JM, Cunningham FG, Lindheimer MD. Chesley's Hypertensive Disorders in Pregnancy, Academic Press, 2014 (Fourth Edition).
- Burnstock G (2009) Purinergic mechanosensory transduction and visceral pain. Mol Pain 5: 69. [Crossref]
- Barker DJP (1992) Fetal and infant origins of adult diseases. British Medic Journal Publishing.
- Li YY, Gu QR, Chen GR, Quinn MJ (2018) Arteriolar injury in hypertension. Am J Med 131: e133-e134. [Crossref]
- Gu QR, Li YY, Chen GR, Quinn MJ (2018) The arteriolar injury in hypertension. Med Hypotheses 111: 66-69.
- Moritz A, Oldt M (1937) Arteriolar sclerosis in hypertensive and non-hypertensive individuals. Am J Pathol 13: 679-728.7. [Crossref]





