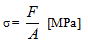The intensive production of various biomedical composites made from different biomaterials showed a huge volume of information on properties of composite materials for appropriate research field. The matrix of composite materials is very important but the reinforcement materials known as fillers play an important role for the final properties of biomedical composites like mechanical properties or biodegradability. In the last years, many research groups proved that the modification of ceramics or polymeric matrix by adding magnesium powder will conduct to the biomedical composites that will enhance bioactivity and mechanical properties. The aim of this paper was to study the effect of adding magnesium powder as filler in collagen-based composites for biomedical applications. By freeze-drying technology we obtained some collagen-based composite materials reinforced with Mg powder that have shown good homogeneity, good integration of the reinforcement material and superior mechanical properties. The intensive production of various biomedical composites made from different biomaterials showed a huge volume of information on properties of composite materials for appropriate research field. The matrix of composite materials is very important but the reinforcement materials known as fillers play an important role for the final properties of biomedical composites like mechanical properties or biodegradability. In the last years, many research groups proved that the modification of ceramics or polymeric matrix by adding magnesium powder will conduct to the biomedical composites that will enhance bioactivity and mechanical properties. The aim of this paper was to study the effect of adding magnesium powder as filler in collagen-based composites for biomedical applications. By freeze-drying technology we obtained some collagen-based composite materials reinforced with Mg powder that have shown good homogeneity, good integration of the reinforcement material and superior mechanical properties.
biomedical composites, filler, magnesium powder
In the last period, the healing and reconstruction of human tissue has become a real challenge for scientists from the medical and engineering fields [1]. At present, there are a variety of biomaterials and processes to heal the damaged tissues and replace the biological tissues. Articular cartilage, skin, nerves, bone and partial organ tissue healing and reconstruction are possible in present and have a huge importance for majority people who have special needs because of organ failure or tissue loss. To solve the issues presented before, different bio composites have been investigated during the past several years, as substitute materials for damaged or diseased tissues from the human body. The composites are used for medical field because provide several unique mechanical properties such as: toughness, strength, stiffness and fatigue resistance and the costs are relatively low [2]. The composite materials are composed of a bulk continuous phase, matrix, and one or more scattered, non-continuous phases, as reinforcement, improving the matrix mechanical and thermal properties. The interface matrix/ reinforcement materials are designed to have the best properties of each of the component materials. The composites are based on the following concept: the matrix accepts the load over a large surface area and move it to the reinforcement phase, which, being distinct, changes the mechanical properties of the composite, whether it is stiffness, fatigue resistance, strength or toughness [3-5].
The biomedical composites properties are strongly influenced by various factors such as:
- reinforcement size, shape and size distribution;
- bioactivity of the reinforcement (or the matrix);
- volume percentage;
- reinforcement properties;
- distribution of the reinforcement in the matrix;
- Matrix properties (grain size, molecular weight etc.);
- Reinforcement-matrix interfacial state.
Beside these factors, properties of the materials which are included in composite play an important role. Moreover, factors like composite architecture (the reinforcement percentage, orientation and distribution) and also, extremely important is reinforcement-matrix bonding condition. The biological and mechanical performance of biocomposites can be adapted to meet various clinical requirements by carefully controlling these factors.
The importance of these materials is in the fact that there are various matrix materials and reinforcement types which can be combined in many different ways to obtain a new material with the desired properties. The classification of the composites can be based on its matrix material (ceramic-matrix composites, polymer-matrix composites and metal-matrix composites- the last type are less used for biomedical applications and are mainly used for high-temperature applications) or based on reinforcement components (particulates, short or continuous fibers) which can be also polymers, ceramics or metals [6,7].
Types of matrix for biomedical composites
Bioceramics: Hydroxyapatite (HA), with the chemical formula Ca10(PO4)6(OH)2, is frequently used in orthopedic applications procedures due to its crystallographic and chemical similarity to the human bones. Hydroxyapatite has been used in the biomedical field for two decades because they accelerate the bone regeneration and repair. HA stands by its excellent biocompatibility and its osteoconductive properties. Despite of its bioactivity and its osteoconductive characteristics, HA cannot be used as a load bearing implant because of its poor mechanical properties. HA is usually used in tissue engineering in the form of powder, dense body or porous structure. However, the most used form of hydroxyapatite is as a bioactive coating on total hip prostheses.
Another compound from the calcium phosphate family used for medical applications is the tricalcium phosphate (TCP), with the chemical formula Ca3(PO4)2, which is remarkable due to its bioresorbable properties. TCP is used for bone repair in the form of calcium phosphates cements, granules or ceramic blocks. However, hydroxyapatite and also tricalcium phosphate are weak because of their poor mechanical properties and as a fact they cannot be used on their own as major load-bearing implants in the human body [8-10].
Another bioactive bioceramics that have been used for bone tissue engineering are bioglass and apatite-wollastonite (A-W) glass–ceramic. Bioglass is a class of bioactive glasses that contain SiO2, Na2O, CaO and P2O5 in different proportions. Bioglass (45S5 Bioglass) is remarkable by its ability to bond to both hard and soft tissues [10]. The main disadvantage of this class of biomaterials is low fracture toughness due to an amorphous two-dimensional glass network mechanical weakness. Another characteristic which makes it unfit for major load-bearing applications is the inadequate bending strength, which is in the range of 40–60 MPa. If heat treatment is applied, a suitable glass can be changed into glass–crystal composite containing crystalline phase(s) of controlled sizes and contents.
The new glass ceramic can have higher mechanical properties than the parent glass as well as than sintered crystalline ceramics. The chemical composition of the porous apatite-wollastonite (A-W) glass ceramic is 4.6% MgO, 44.7% CaO, 34.0% SiO2, 16.2% P2O5, and 0.5% CaF2, and the crystallised glass-ceramic consisted of 28% residual glass, 38% apatite [Ca10(PO4)6(O, F2)], and 34% wollastonite (SiO2•CaO). The apatite-wollastonite (A-W) glass ceramic is obtained by the conventional melt-quench method and their bioactivity is superior to the sintered HA. Thus, A-W glass–ceramic has excellent mechanical properties and has been used in the medical field for vertebrae and iliac prostheses and as intervertebral spacers [11].
Polymers: Polymers are a large class of natural or synthetic materials with different properties that are used in tissue engineering. The natural polymers class includes proteins (collagen, fibrinogen, gelatin and keratin), polysaccharides (cellulose, chitin, chitosan and amylose) or polynucleotides (ribonucleic acid, deoxyribonucleic acid). There are many biocompatible and bio-stable polymers, however only a few of them are used for bone matrices. Usually, the composite materials based on biodegradable polymeric matrix such as poly(lactic acid) (PLA), poly(ε-caprolactone) (PCL) and polyhydroxybutyrates (PHBs) are used in order to obtain composite materials for bone substitutes, but polymers such as polysulfone (PSU) and polyether ether ketone (PEEK) was used for this purpose.
Poly (ε-caprolactone) (PCL) is a biodegradable polymer with a very good biocompatibility. Also, the low degradation rate of PCL and its mechanical properties give biocomposites with PCL several advantages for application in tissue engineering. Poly (L-lactide) is a semi-crystalline polymer, called as ‘green biopolymer’ and recognized for biomedical applications due its good biocompatibility, biodegradability and acceptable mechanical properties [12,13].
Types of fillers for biomedical composites
The intensive production of various composites using different filler showed a huge volume of information on properties of composite materials for the appropriate research field. The reinforcement materials are generally used as continuous fibers, whiskers or particles, from the shape point of view. The filler type has a huge importance in the final composite properties such as biodegradability, conductivity, stiffness, strength, and color [13,14].
In comparison to particles or whiskers, the diameters of the fibers are higher (5 to 50 μm). When the diameters are smaller, the strength of the fiber is elevated due to a reduction in surface flaws. The properties of composite materials are also influenced by the orientation, the length and the volume fraction of fibers [15-17]. Usually, materials are stronger and stiffer in the fibrous form than in the other form. This fact explains the choice of the scientists for using fibers in composite materials design, especially when they are used for structural applications, where they are the main load-carrying component. Whiskers have their aspect ratio higher (>100) and smaller diameters (around 10 μm) than continuous fibers, being fibers made of single crystals. They have very high strengths but also high manufacturing cost. Unlike the continuous fiber composites, the short ones are less efficient in use and in attaining of a desired orientation. They can also come very close to obtaining their theoretical strength and are limited in processing and design possibilities. Natural fibers such as collagen, cotton, wool, and silk, have various properties and present many processing challenges. The collagen fibers were successfully used in tissue engineering products for ligament and skin regeneration. In the biomedical field carbon fibers which are as strong as glass fiber are used in manufacturing external prosthetic components in order to improve the fatigue resistance [18,19]. Through polymers, aramid fibers with very precise orientation such as Kevlar are used for orthopedic applications due to their high resistance to impact fracture. For the cardiology field Teflon and polyester (Dacron) fibers are used for the production of flexible vascular prostheses. Polylactide and polyglycolide and their copolymers are used to make fiber composites with high adsorption properties [16,17].
The particles used as reinforcement material enhance the mechanical properties such as hardness and toughness. In the case of polymer based composites, by adding the ceramic particles, controlled properties such as conductivity, biodegradability, and dimensional stability. Particulate reinforcement is randomly disseminated in a matrix, which leads to isotropic composites. Depending on their shape, particles can influence the mechanical properties of the composite materials. Spherical particles are less helpful than platelet - or flake like particles in including stiffness. In the biomedical field, the particulate reinforcement is used for ceramic matrices for bone and dental applications. The most used particles form is hydroxyapatite, but they have low mechanical properties and can be used more as a bioactive component than as a reinforcement component.
The determination of the mechanical properties of a composite is based on the physical characteristics (size, shape, size distribution) of the reinforcement. The reinforcement is usually assumed to have a spherical shape due to mathematical modeling of the mechanical behavior of a particulate composite, in the ideal case. In reality, the particulate filler may have an acicular, platy or irregular shape [18].
Clinical applications and some limitations of currently used biocomposites
The method which combines the results of various studies for providing conclusions about the efficacy of a treatment modality is named meta-analysis. One of the purposes of this paper is to perform an analysis of the scientific literature in order to study the effect of adding magnesium powder as filler for biomedical composites by combining information from multiple studies. Based on the scientific literature analysed [19-37], we present an overview about the existing studies about biomedical composites reinforced with magnesium in Table 1. Magnesium is the second most abundant element inside human tissues and the fourth most abundant positively charged ion in the human body (approximately 20-28 g Mg). Magnesium plays important roles in the human body, thus it is involved in neuromuscular transmission and activity and muscle relaxation, in supporting a healthy immune system, in the regulation of carbohydrate and glucose metabolism [36,37]. Magnesium and their alloys appear to have a great potential as a biodegradable metallic biomaterial for manufacturing different orthopedic and cardiovascular applications [38]. Magnesium has the advantage of the ultra-low density (1.74 g/cm3), it has a Young's modulus close to that of bone (3–20 GPa), high specific ratio of weight rather than other metallic biomaterials which are of particular importance in osteosynthesis applications [39]. Also, what makes it suitable for its use in the biomedical field are its mechanical properties which are similar to those of the human bone on higher than polymers or polymer-based composites currently used for manufacturing bioresorbable orthopedic implants [40]. This combination of remarkable properties and rate of biodegradation in physiological environments have encouraged scientists to study Mg-based biodegradable materials for potential clinical applications, especially for orthopedic applications [41,42]. In vivo studies have shown tolerable cytotoxicity of magnesium based implants [43]. The process of degradation which appears on the bone-magnesium alloy interface has been studied by Witte et al. and they analysed bone formation nearby biodegradable implants made by different magnesium alloys (WE43, AZ31, AZ91, LAE442) in comparison with currently degradable polymers used currently in clinical practices. A better deposition rate of mineral close to degradable implants and higher osteoblast activity compared to a degradable polymer implant has been observed. The magnesium ions positively influence the activity of enzymes which manage the nucleic acids and the synthesis of biological nucleic acids [12,13]. More recently, the scientists have proved that modification of ceramics-based composites by adding magnesium powder enhance bioactivity and stimulates vascularization [16]. Witte use magnesium alloy-based metal matrix composites improved with hydroxyapatite particles, while Cifuentes added magnesium particles into polymer matrix obtaining biomedical composites with potential use for orthopedic applications due to their good osseointegration and to reduced inflammatory response [17]. The biomedical composites reinforced with magnesium powder are known as promising materials to provide higher strength implants compared to unreinforced metals [44-46]. However, the main disadvantages such as high degree of shrinkage during solidification of molten magnesium, its rate of corrosion in physiological and high chloride environments and high chemical reactivity especially at elevated temperatures have reduced the use of pure Mg [46,47].
Table 1. Biomedical composites reinforced with magnesium base filler (overview of the published studies)
Composite type |
Application |
Properties |
Year |
Author |
Ref |
Polymer matrix |
Reinforcement material |
|
|
|
|
|
Collagen |
HA |
Orthopedics |
Accelerate osteogenesis
Improve resistance to degradation |
2004 |
Xie
et al. |
[19],[20],[21] |
TCP |
Scaffolds for bone regeneration
|
Improve cellular attachment, viability, proliferation, and activity and mechanical properties |
2009 |
Moreau
et al. |
[22] |
Mg+TCP+ Mg+10% TCP |
Orthopedics |
Improve biocompatibility, rate of degradation and mechanical properties |
2013 |
Antoniac
et al. |
[23] |
PLLA |
HA |
Tissue engineering scaffold
|
Enhance osteoinductivity, osteoconductivity and mechanical properties |
2004 |
Kasuga
et al. |
[25],[26] |
TCP |
Tissue engineering scaffold
|
Improved mechanical properties |
2001 |
Xiong
et al. |
[27],[28],
[29] |
Mg particles |
Orthopedics |
Enhance the osseointegration;
Reduce the inflammatory response |
2012 |
Cifuentes
et al. |
[30] |
PCL |
HA |
Boontharika Chuenjitkuntaworn,
Boontharika Chuenjitkuntaworn,
Orthopedics |
Increase the bone bonding to the implant and the mechanical properties |
2013 |
Boontharika Chuenjitkuntaworn,
Antoniac |
[24] |
TCP |
Protein or growth factor delivery |
Enhance cell growth |
2000 |
Hutmacher
et al. |
[31], [32], [33] |
Ceramic matrix |
Magnesium powder |
Bone tissue engineering
Regenerative medicine |
Reduce hydrogen evolution |
2016 |
Xiong
et al. |
[34] |
HA |
45S5 bioglass |
ZK30/ Mg
|
Orthopedics |
Enhance bone formation through stimulation of osteoblast
proliferation |
2012 |
Huan
et al. |
[35],[36] |
HA +TCP
|
Mg-5Sn |
Bone repair |
Better corrosion resistance |
2015 |
Wang
et al. |
[37] |
The corrosion reaction (Mg+2H
2O→Mg
2++2OH
−+H
2) accelerates the formation of hydrogen, which leads to a premature loss of mechanical strength and harmful hydrogen gas pockets, as well as local increase of alkalinity in the vicinity of magnesium-based implants [48]. Alloying is the best option to overcome these problems and to improve the mechanical properties and also, the corrosion resistance of metallic Mg with a reduced hydrogen evolution [49].
Since previous studies have demonstrated that magnesium endowment with bioactivity and simultaneously reducing the release rate of hydrogen can be achieved by the addition of bioactive ceramic fillers such as hydroxyapatite (HA) and bioactive glass into Mg matrix [50-52].
In their researches from 2007, Witte et al. introduced Mg-matrix composites as a new generation of orthopedic biomaterials, their role being to avoid any known or uncertain clinical side effect that often comes with common alloying elements associated with magnesium. The results obtained from studying the Mg-matrix composites have shown the improvements of degradation properties and the bioactivity, while the issues about their mechanical behavior along with degradation rate have not been approached [52-55].
The poly(α-hydroxyacid)s like PLA, poly(glycolic acid) (PGA), and polydioxanone (PDS) are the most used biodegradable polymers in the biomedical field. The special features of the PLA such as good biodegradability and biocompatibility make it potentially useful for applications in medical industry. The main disadvantage of various PLA materials is the low mechanical properties and therefore they could not meet the requirements of human bones. The main reinforced materials which have been investigated to solve these problems were titanium dioxide, calcium carbonate, bioglass, chitosan and magnesium, in form of powders. From these materials, magnesium powder appears to be the most attractive due to its high properties of biocompatibility, biodegradable character, and potential to increase the mechanical properties.
Various works support the hypothesis that the mechanical properties of the material of bone substitutes must be consistent with those of natural bone (Table 2) [9]. Another important type of materials used for obtaining the composites or scaffolds for bone tissue engineering are biodegradable polymer-based composites reinforced with different calcium phosphates (CaPs) due to the hydroxyapatite and other calcium phosphates chemical and crystal resemblance to the natural bone tissue. However, the use of the combination polymer- ceramic for load bearing applications with requested biodegradability is limited. Fortunately, we consider that reinforcing these composites with supplementary magnesium powder could offer a wide range of possibilities in this case. Usually, the bone substitutes need to have a good bone binding ability. As we mentioned above, by adding calcium phosphates or magnesium powder the bioactivity of a material can be improved and the osseointegration is stimulated, as shown by Cifuentes et al., who obtained a better osseointegration of the bone implant and a reduced inflammatory response by incorporating Mg particles into polymer matrix [56-61].
Table 2. Mechanical properties and porosity of the human bones.
Bone |
Modulus of elasticity [GPa] |
Porosity
[%] |
Tensile Strength
[MPa] |
Compressive Strength [MPa] |
Flexural strength
[MPa] |
Cortical |
12-18 |
5-13 |
50-151 |
130-180 |
135-193 |
Spongy |
0,1-0,5 |
50-90 |
1-5 |
4 - 12 |
- |
Table 3. The values of compression force and compressive strength of experimental samples.
Sample |
Compression Force
[N] |
Compressive Strength
[MPa] |
C1 |
127 |
1.62 |
C2 |
699 |
8.90 |
C3 |
628 |
8.00 |
The aim of this study was to obtain and characterize some collagen-based composites reinforced with Mg powder, hydroxyapatite and β-TCP, potentially used as bone substitutes.
Experimental collagen-based composites were obtained using the freeze-drying technology, according to the previously described protocol [62,63]. Each sample contains 1.2% collagen (percentage by weight/ quantitative percentage based on dry substance). The samples were crosslinked with 0.25% glutaraldehyde (crosslinking agent). The initial collagen concentration was 2.11% and was brought to a value of 1.2%. The initial pH value was 2.6 and was brought to pH=7.4 by adding 1M NaOH. An INOLAB pH-meter was used for determination of the pH. After which a gel with a concentration 1.2% collagen was obtained, the powder (Mg, respectively the mixt Mg/ β-TCP) was incorporated. We obtained gels samples with the concentration of 5% Mg and 5% Mg, 5% β-TCP (volume percentage; the report is made at the volume of the gel). The gels were poured into glass Petri boxes, with the diameter of 5-5.2 cm, after which they were lyophilized in a Martin Christ 24LSC lyophilizer, for 24 hours. After lyophilization, we obtained experimental samples in the form of spongy collagen-based matrices: collagen-Mg (5%Mg), collagen-Mg- β-TCP (5% Mg; 5% β-TCP). Also, we obtain a collagen sponge by the same method which was used as reference sample. The experimental samples with disc shape are shown in Figure 1.

Figure 1. Macroscopic view of samples of collagen-based composites reinforced with magnesium powder: a) C1: collagen (reference sample); b) C2: collagen-based composite reinforced with 5% Mg; c) C3: collagen-based composite reinforced with 5% Mg and 5% β-TCP.
The preliminarily characterization of the experimental collagen-based composites was made in order to analyse the chemical structure, filler distribution and integration into collagen matrix. The chemical structure was analyzed by FTIR spectroscopy in the near IR spectrum using a FTIR JASCO 6200 Spectrometer. Scanning Electron Microscopy (SEM) coupled with Energy Dispersive X-ray Spectroscopy (EDS) was used for the morphology analysis and identification of the filler presence into the collagen matrix. The equipment used was a Philips microscope XL 30 ESEM TMP type.
For mechanical tests, the experimental samples were obtained in cylinders with a diameter of 13 mm and a height of 10 mm. After lyophilisation, the samples have changed dimensions. These, in dry state, were tested at compression using an instron 8801 equipment. All tests were realized using the same operating parameters. The samples were subjected at compression with a loading speed of 0,5 mm/s. Compression tests were carried out on 5 samples for each one of types of composite material (n=5).
SEM-EDS results
According the experimental results obtained by SEM (Figure 2), we observe that the smooth surface that appear in the case of collagen sample began to modify with the incorporation of fillers (Mg, respectively Mg-β-TCP). The filler will induce a gradually rough surface and morphological modification but the composites will keep their porous structure. The irregular nature of the inorganic component and relatively inhomogeneous distribution of filler in the collagen matrix was observed.

Figure 2. SEM images of samples of collagen-based composites reinforced with magnesium powder. For the meaning of the sample codenames see legend to Figure 1.
The EDS analyses (Figure 3) put in evidence the magnesium and β-TCP presence as fillers in collagen matrix. The distribution of the magnesium particles is relatively uniform although some particles agglomerated during the process.
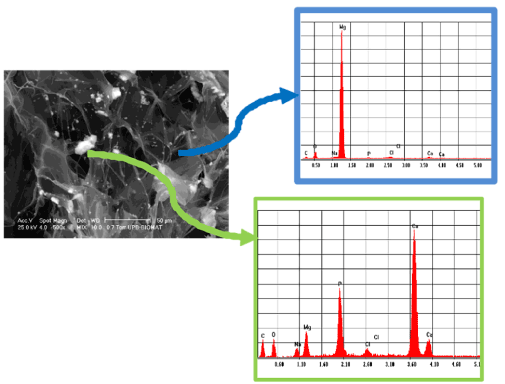
Figure 3. SEM image coupled with EDS analysis of sample C3, showing the presence of Mg and β-TCP particles and the good integration of the reinforcement materials in the collagen matrix. For the meaning of sample codename see legend to Figure 1.
FTIR results
The experimental spectra recorded by FTIR spectroscopy for all samples are shown in Figure 4. The FTIR pattern emphasize the characteristic bands for collagen and namely, the amide I band at 1644 cm-1 , the amide II at 1549 cm-1 and amide III at 1239 cm-1. The amide I band occurs due to the stretching vibration of carbonyl group (C=O), the amide II band is due to the strong stretching vibration of N-H bond coupled with stretching vibration of C-N and the amide III band is due to the stretching vibration of the C-N groups coupled with deformation vibration of N-H groups (Figure 4).
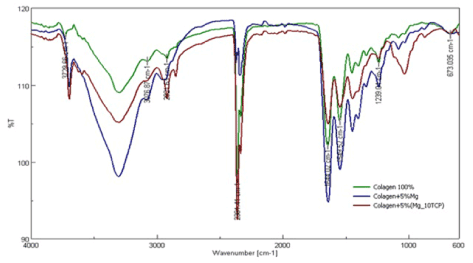
Figure 4. IR spectra corresponding to the samples described in Figure 1.
Along with these bands appear the characteristic bands for amide A at 3297 cm-1, amide B at 3076 cm-1 and absorption band from 1444 cm-1, due to vibration of the pyrrolidine ring of proline and hydroxyproline. In the FTIR spectra of the sample C3 is highlighted the characteristic band of the phosphate group from β-TCP at 1032 cm-1.
Determination of compressive strength
The experimental composites were subjected to a single press cycle, using a speed of 0.5 mm/min. The compressive strength was calculated with the following mathematical equation:
where:
F –compressive force,
A – cross-sectional area of the sample.
According to the results shown in F 3 and Figure 5, the values of the compression force for the experimental composite materials register high values compared to the control sample made just of collagen (Figure 5).
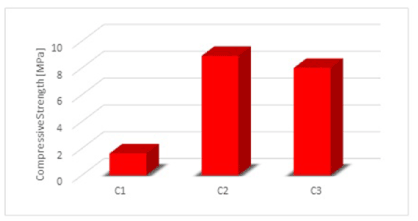
Figure 5. The variation of compression strength in the experimental composites. For the meaning of the sample codenames see legend to Figure 1.
From the mechanical compression tests results, we observe that even the highest value of the compression force was registered by sample C2, respectively the collagen-based composite reinforced with 5% Mg, the experimental composites have similar values and could be considered as potential materials for bone substitutes from the mechanical compression tests perspective.
2021 Copyright OAT. All rights reserv
Using freeze-drying technology, collagen-based composites reinforced with Mg powder, hydroxyapatite and β-TCP can be obtained as potential bone substitutes. This technology assures a better integration of the magnesium powder into collagen matrix. Also, the potential problems related to the hydrogen release when magnesium powder is into an aqueous medium because the magnesium powder is mixed in collagen gel before starting the liophylization process.
The collagen-based composites reinforced with Mg powder exhibit improved mechanical properties as compared to pure collagen-matrix obtained by the same method and these properties are very similar to the mechanical properties of spongy bone.
Further studies are due in order to avoid the concerns regarding the biocompatibility of these composites and to demonstrate that it is possible to increase and control the degradation rate by using Mg powder as filler in polymer-based composites.
Compliance with ethical standards
Not applicable.
The authors declare that there is no conflict of interest of a scientific or commercial nature. The authors have no relevant affiliations to, or financial support from any organization that may have a financial interest in the subject matter.
- Smith MJ, Smith DC, Bowlin GL, White KL (2010) Modulation of murine innate and acquired immune responses following in vitro exposure to electrospun blends of collagen and polydioxanone. J Biomed Mater Res A 93: 793-806.
- Salernitano E, Migliaresi C (2003) Composite materials for biomedical applications: a review. J Appl Biomater Biomech 1: 3-18. [Crossref]
- Hull D, Clyne TW (1996) An introduction to composite materials. 2nd ed. Cambridge: Cambridge University Press p. 208-236.
- Bonfield W, Grynpas MD, Tully AE, Bowman J, Abram J (1981) Hydroxyapatite reinforced polyethylene a mechanically compatible implant material for bone replacement. Biomaterials 2: 185-186.
- Wang M (2003) Developing bioactive composite materials for tissue replacement. Biomaterials 24: 2133-2151. [Crossref]
- Ess JW, Hornsby PR (1987) Twin-screw extrusion compounding of mineral filled thermoplastics: dispersive mixing effects. Plast Rubber Process Appl 8: 147–156.
- Wang M, Porter D, Bonfield W (1994) Processing, characterisation, and evaluation of hydroxyapatite reinforced polyethylene composites. Br Ceram Trans 93: 91–95.
- Aoki H (1994) Medical applications of hydroxyapatite. Tokyo, St. Louis: Ishiyaku EuroAmerica, p. 13-74.
- Hench LL, Wilson J, editors (1993) An introduction to bioceramics. 2nd ed. Singapore: World Scientific, p. 521-540.
- Elliott JC (1994) Structure and chemistry of the apatites and other calcium orthophosphates.1st ed. Amsterdam: Elsevier Science, p. 55-62.
- Hench LL (1991) Bioceramics: from concept to clinic. J Am Ceram Soc 74: 1487–1510.
- Ratner BD, Hoffman AS, Schoen FJ, Lemons JE (1996) editors. Biomaterials. San Diego: Academic Press, p. 193-199.
- Suuronen R, Pohjonen T, Hietanen J, Lindqvist C (1998) A 5-year in vitro and in vivo study of the biodegradation of polylactide plates. J Oral Maxillofac Surg 56: 604-614. [Crossref]
- Evans SL, Gregson PJ (1998) Composite technology in load-bearing orthopaedic implants. Biomaterials 19: 1329-1342. [Crossref]
- Bernardini D, Nasulewic A, Mazur A, Maier J. (2004) Magnesium and microvascular endothelial cells: a role in inflammation and angiogenesis. Front Biosci 10: 1177–1182.
- Witte F, Feyerabend F, Maier P, Fischer J, Stoermer M, (2007) et al. Biodegradable magnesium–hydroxyapatite metal matrix composites. Biomaterials 28: 2163-2174.
- Witte F, Fischer J, Nellesen J, Crostack HA, Kaese V, et al. (2006) In vitro and in vivo corrosion measurements of magnesium alloys. Biomaterials 27: 1013-1018. [Crossref]
- Witte F, Kaese V, Haferkamp H, Switzer E, Meyer-Lindenberg A, et al. (2005) In vivo corrosion of four magnesium alloys and the associated bone response. Biomaterials 26: 3557–3563.
- Xie J, Baumann MJ, McCabe LR (2004) Osteoblasts respond to hydroxyapatite surfaces with immediate changes in gene expression. J Biomed Mater Res A 71: 108-117. [Crossref]
- Argarate N, Olalde B, Atorrasagasti G, Valero J, Cifuentes SC et al. (2014) Biodegradable bi-layered coating on polymeric orthopaedic implants for controlled release of drugs. Mater Lett 132: 193–195.
- Cifuentes SC, Gavilán R, San Román A, Lieblich M, Benavente R, et al (2014) Effect of magnesium particle shape on in-vitro degradation kinetics of novel PLA/Magnesium composites. Eur Cell Mater 28: 70.
- Moreau JL, Sun L, Chow LC, Xu HHK (2011) Mechanical and acid neutralizing properties and inhibition of bacterial growth of amorphous calcium phosphate dental nanocomposite. J Biomed Mater Res B. 98: 80–88.
- Antoniac I, Vranceanu MD, Antoniac A (2013) The influence of the magnesium powder used as reinforcement material on the properties of some collagen based composite biomaterials. J Optoelectron Adv M 15: 667-672.
- Antoniac I (2014) Biodegradability of some collagen sponges reinforced with different bioceramics. Key Eng Mater 587: 179-184.
- Kasuga T, Ota Y, Nogami M, Abe Y (2001) Preparation and mechanical properties of polylactic acid composites containing hydroxyapatite fibers. Biomaterials 22: 19-23.
- Kasuga T, Maeda H, Kato K, Nogami M, Hata K (2003) Ueda M. Preparation of poly(lactic acid) composites containing calcium carbonate (vaterite). Biomaterials 24: 3247-3253.
- Xiong Z, Yan Y, Zhang R, Sun L (2001) Fabrication of porous poly(L-lactic acid) scaffolds for bone tissue engineering via precise extrusion. Scripta Mater 5: 773–779.
- Xiong Z, Yan Y, Chen L (2001) Two new Rapid Prototyping technologies for cell delivery scaffolds of bone tissue engineering. Chin J Mech Eng 12: 515–518.
- Xiong Z, Yan Y, Wang S, Zhang R, Zhang C (2002) Fabrication of porous scaffolds for bone tissue engineering via low-temperature deposition. Scripta Mater 46: 771–776.
- Cifuentes SC, Frutos E, González-Carrasco JL, Muñoz M, Multigner M et al. (2012) Novel PLLA/magnesium composite for orthopedic applications: A proof of concept. Mater Lett 74: 239–242.
- Rohner D, Hutmacher DW, Cheng TK, Oberholzer M, Hammer B (2003) In vivo efficacy of bone-marrow-coated polycaprolactone scaffolds for the reconstruction of orbital defects in the pig. J Biomed Mater Res B 66: 574–580.
- Hutmacher DW1, Schantz JT, Lam CX, Tan KC, Lim TC (2007) State of the art and future directions of scaffold-based bone engineering from a biomaterials perspective. J Tissue Eng Regen Med 1: 245-260. [Crossref]
- Hutmacher DW1 (2000) Scaffolds in tissue engineering bone and cartilage. Biomaterials 21: 2529-2543. [Crossref]
- Xiong G, Nie Y, Ji D (2016) Characterization of biomedical hydroxyapatite/magnesium composites prepared by powder metallurgy assisted with microwave sintering. Curr Appl Phys 16: 830–836.
- Huan ZG, Leeflang MA, Zhou J, Duszczyk J (2011) ZK30-bioactive glass composites for orthopedic applications: A comparative study on fabrication method and characteristics. Mater Sci Eng B 176: 1644–1652.
- Huan Z, Leeflang S, Zhou J, Duszczyk J (2010) Magnesium-based composites with improved in vitro surface biocompatibility. J Mater Sci Mater Med 21: 3163-3169. [Crossref]
- Dezfulia SN, Leeflanga S, Huan Z, Chang J, Zhoua J (2017) Fabrication of novel magnesium-matrix composites and their mechanical properties prior to and during in vitro degradation, J Mech Behav Biomed Mater. 67: 74–86.
- Khalajabadi SZ, Kadir MA, Izman S, Marvibaigi M (2016) The effect of MgO on the biodegradation, physical properties and biocompatibility of a Mg/HA/MgO nanocomposite manufactured by powder metallurgy method. J Alloys Compd 15: 266-280.
- Ham B, Zhang X (2011) High strength Mg/Nb nanolayer composites, Mater Sci Eng A 528: 2028–2033.
- Wang X, Li JT, Xie MY, Qu LJ, Zhang P et al. (2015) Structure, mechanical property and corrosion behaviors of (HA + ß-TCP)/Mg–5Sn composite with interpenetrating networks. Mater Sci Eng C. 56: 386-392.
- Mordike B, Ebert T (2001) Magnesium: Properties applications potential. Mater Sci Eng A 302: 37–45.
- Gupta M, Meenakshisundaram GK (2015) Insight into Designing Biocompatible Magnesium Alloys and Composites:Processing, Mechanical and Corrosion Characteristics; Springer: New York, NY, USA p. 1-15.
- Wang K (1996) The use of titanium for medical applications in the USA. Mater Sci Eng A 213: 134–137.
- Long M, Rack HJ (1998) Titanium alloys in total joint replacement – A materials science perspective. Biomaterials 19: 1621–1639.
- Cha PR, Han HS, Yang GF, Kim YC, Hong KH, et al. (2013) Biodegradability engineering of biodegradable Mg alloys: tailoring the electrochemical properties and microstructure of constituent phases. Sci Rep 3: 2367–2372.
- Salahshoor M, Guo Y (2012) Biodegradable Orthopedic Magnesium-Calcium (MgCa) Alloys, Processing, and Corrosion Performance. Materials 5: 135.
- Munitz A, Cotler C, Stern A, Kohn G (2001) Mechanical properties and microstructure of gas tungsten arc welded magnesium AZ91D plates. Mater Sci Eng A 302: 68–73.
- Witte F, Kaese V, Haferkamp H, Switzer E, Meyer-Lindenberg A, et al. (2005) In vivo corrosion of magnesium alloys and the associated bone response. Biomaterials 26: 3557-3563.
- Poinern GEJ, Brundavanam S, Fawcett D (2012) Biomedical Magnesium Alloys: A review of material properties, surface modifications and potential as a biodegradable orthopaedic implant. AJBE 2: 218-240.
- Huang Y, Liu D, Anguilano L, You C, Chen M (2015) Fabrication and characterization of a biodegradable Mg-2Zn-0.5Ca/1β-TCP composite. Mater Sci Eng C Mater Biol Appl 54: 120-132. [Crossref]
- Yu HJ, Wang JQ, Shi XT, Louzguine-Luzgin DV, Wu HK, et al, (2013) Ductile biodegradable Mg-based metallic glasses with excellent biocompatibility, Adv Funct Mater 23: 4793–4800.
- Witte F, Feyerabend F, Maier P (2007) Biodegradable magnesium-hydroxyapatite metal matrix composites. Biomaterials 28: 2163–2174.
- Witte F, Hort N, Vogt C, Cohen S, Kainer KU, et al. (2008) Degradable biomaterials based on magnesium corrosion. Curr Opin Solid State Mater Sci. 12: 63-72.
- Niiranen H, Pyhältö T, Rokkanen P, Kellomäki M, Törmälä P (2004) In vitro and in vivo behavior of self-reinforced bioabsorbable polymer and self-reinforced bioabsorbable polymer/bioactive glass composites, J Mater Sci Mater Med 69: 699–708.
- Li L, Ding S, Zhou C (2004) Preparation and degradation of PLA/chitosan composite materials. J Appl Polym Sci 91: 274–277.
- Wu YH, Li N, Cheng Y, Zheng YF, HanY (2013) In vitro study on biodegradable AZ31 magnesium alloy fibers reinforced PLGA composite. J Mater Sci Technol 29: 545–550.
- Vergnol G, Ginsac N, Rivory P, Meille S, Chenal JM, et al. (2016) In vitro and in vivo evaluation of a polylactic acid-bioactive glass composite for bone fixation devices. J Biomed Mater Re 104: 180–191.
- Cifuentes SC, Bensiamar F, Gallardo-Moreno AM, Osswald TA, González-Carrasco JL, et al. (2016) Incorporation of Mg particles into PDLLA regulates mesenchymal stem cell and macrophage responses. J Biomed Mater Res A 104: 866–878.
- Witte F, Feyerabend F, Maier P, Fischer J, Störmer M, et al. (2007) Biodegradable magnesium-hydroxyapatite metal matrix composites. Biomaterials 28: 2163–2174.
- Huang Y, Liu DB, Xia MX, Anguiliano L (2013) Characterization of an Mg-2Zn-1Ca-1ß-TCP composite fabricated by high shear solidification and ECAE. Mater Sci. 765: 813-817.
- He S, Sun Y, Chen M, Liu D, Ye X (2011) Microstructure and properties of biodegradable ß-TCP reinforced Mg-Zn-Zr composites, China. Trans Nonferrous Met Soc. 21: 814-819.
- Rusu LC, Nedelcu IA, Albu MG, Sonmez M, Voicu G, et al. (2015) Tetracycline loaded collagen/hydroxyapatite composite materials for biomedical applications. J Nanomater 1: 1-5.
- Albu MG, Titorencu I, Ghica MV (2011) Biomaterials Applications for Nanomedicine. 1st ed. Croatia:InTech p. 333–358.