The present work proposes an original approach to study the effectiveness of antisense intervention in the brain via the association of molecular biological applications and In vivo neuro-pharmacological analysis. In particular, direct influence of specific antisense oligonucleotide of GABAA receptors upon physiological parameters such as sleep duration as well as presynaptic and post synaptic activities via In vivo and in real time electrochemical analysis of neurotransmitters and cell firing in discrete rat brain areas have been monitored.
GABAA receptors antisense, in vivo electrochemistry & electrophysiology, molecular biology, rat brain
The influence of GABA upon sleep is well known [1]. In particular, the role of the GABAA receptors is well established [2]. The present experiments have been undertaken to observe i] the feasibility of monitoring In vivo eventual direct influence of specific antisense oligonucleotide of GABAA receptors upon physiological parameters such as sleep duration as well as ii] presynaptic and post synaptic activities via electrochemical analysis of neurotransmitters and cell firing. While sleep recording were performed via classic polygraphic measurements [3], the electrochemical original approach has been performed by means of the association of voltammetric and electrophysiologic measurements at a single microsensor as described previously [4,5].
As first target for antisense treatment, the GABA A ALPHA-2 subunit has been chosen. The microinjection of the respective antisense oligonucleotide [obtained from Thermo Fisher Scientific U.S.A.] into the medial preoptic area of the rat brain caused a significant increase in sleep time. The selectivity of such effect was confirmed by the observation that no effect was observed when treating with GABA A ALPHA-5 subunit (Figure 1).
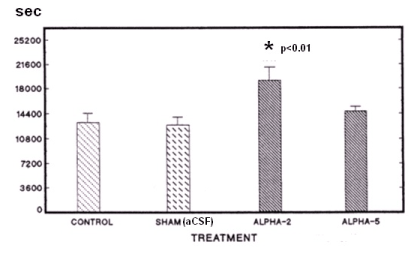
Figure 1. Effect of ALPHA-2 or ALPHA-5 subunit on pentobarbital sleep-time here measured in seconds: the sleep evaluation method was based on prolongation of pentobarbital-induced sleeping time as described [6,7]. Briefly, various groups of animals were given no treatment (control, n=5) a single dose of the vehicle aCSF (SHAM, 1μl, n=5), or the subunits (15 μg, n=5 each). After 30 min, pentobarbital (40 mg/Kg, IP) was injected to induce sleep. Each rodent was considered asleep if stayed immobile and lost its righting reflex when positioned on its back.
Successively, in order to evaluate the putative effect(s) of antisense treatment at both presynaptic (voltammetry) and post-synaptic (electrophysiology) levels, a single carbon fibre micro electrode [6,7] was stereotactically implanted in the CA3. region of the hippocampus of anaesthetised rats (urethane, 1.2 mg/kg i.p. n=5) as described earlier [8].
After a period of control measurements, a single injection of 15 μg antisense (in 1 μl of aCSF) was performed in situ, close to the micro biosensor. This was followed by:
i) a large increase of the extracellular catecholaminergic peak (dopamine release [Crespi 1990, 2002] and of the peptidergic voltammetric signal with maximum levels 2.5-3 hours later, then remaining at high levels until the end of the experiment which lasted more than 4 hours.
ii) a minor, transient increase of the indole signal that was reaching 200% of control values within 50 min after injection (Figure 2).
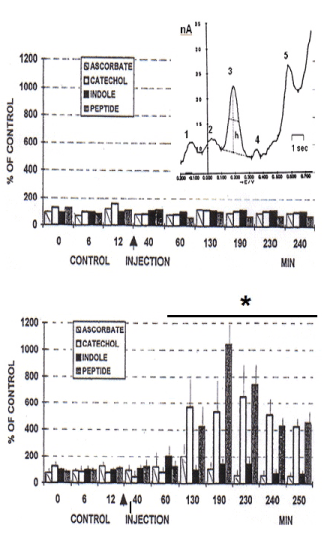
Figure 2. In vivo voltammetry performed with micro carbon fibre electrodes in the hippocampus [CA3] of anaesthetized rats treated [arrow] with 1 μl aCSF (TOP: n=5 control group) or 15 μg ALPHA2 antisense (BOTTOM n=5). Each DPVoltammetric measurement analyzing the 4 signals was lasting approximately 30 sec and repeated every 5 min, in the interval period the micro biosensor was shifted to electrophysiological measurement of cell firing [8]. The DPVoltammogram shows the peaks recorded each scan i.e. Peak 1 = ascorbate; peak 2 = catechols, peak 3 = 5-OH-indoles, peak 4 = HVA, peak 5 = neuropeptides [9,10].
iii) None of the biochemical effects were seen after injection of aCSF (vehicle) alone.
The concomitant electrophysiological monitoring indicated a parallel to biochemical changes of neurotransmitter activities increase of the cell firing as shown in Figure 3.
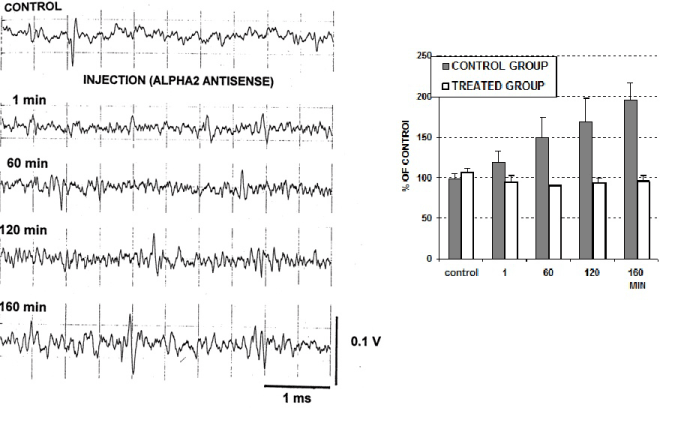
Figure 3. Electrophysiological recordings in the CA3. region performed with the single micro-biosensor used also for voltammetric analysis as described [8-12] in a single rat [left] and in the control group (1 μl aCSF, n=5) or in the rats treated with 15 μg antisense (n=5).
In all the experiments, values were expressed as mean ± SEM. Statistical analysis was performed using one way analysis of variance (ANOVA) followed by Tamhane’s T2 post-hoc test. Differences were considered significant at *p < 0.01.
The data gathered in these experiments indicate a direct In vivo effect of oligonucleotide antisense upon neurotransmitter activities as well as cell firing. In particular it appears that the action of blocking the expression of a GABA A receptor subunit via treatment with a selective oligonucleotide antisense can be confirmed via real time monitoring of selective changes of neurotransmitter activities and cell firing. Thus, these results indicate that the effects of antisense treatment on sleep time are not necessarily due to changes within the GABAergic system alone, but might as well involve changes in the release of other neurotrasmitters relevant for sleep regulation e.g. serotonin [13]. lndeed, in the experiment performed with the ALPHA2 antisense the indole signal was reaching 200% of control values 50 min after treatment (Figure 2).
To our knowledge this is the first time that the effectiveness of antisense intervention in the brain is demonstrated In vivo as its direct selective influence on “classic” neurotransmitter systems was monitored via electrochemical in situ analysis in discrete brain areas in real time. This data proposes the usefulness of the association of molecular biological applications and In vivo neuro-pharmacological analysis for a better understanding of the In vivo characteristics and anatomical localization of GABAA receptor subtypes: this will contribute to rationale drug development. For instance, the feasibility of changing the position of the electrode within one animal brain multiple times, would allow to screen the projection areas of the antisense injection site for changes in cell firing and-transmitter release resulting in a substantial reduction of the extremely time consuming of classic in vitro methodologies to determine the exact location of the receptor knockout. Positive “In vivo areas” could then be further anatomically evaluated by immunocytochemistry and in situ hybridization.
In conclusion, the presented preliminary data obtained by the novel approach of molecular biology and In vivo neuro-electro-biochemical methods indicate the feasibility of a link between molecular biological applications and the classical neuropharmacological analysis. This will enlarge our understanding of processes regulated by ligand gated ion channels at the molecular, as well as at the cellular-receptorial levels, thus it appears highly promising for the purpose of opening up new avenues of ethical pharmacology.
- Gottesmann C (2002) GABA mechanisms and sleep. Neuroscience 111: 231-239. [Crossref]
- Mehta AK, Ticku MK (1999) An update on GABAA receptors. Brain Res Rev 29: 196-217. [Crossref]
- Oishi Y, Takata Y, Taguchi Y, Kohtoh S, Urade Y, et al. (2016) Polygraphic Recording Procedure for Measuring Sleep in Mice. J Vis Exp: e53678. [Crossref]
- Crespi F (1989) Advances in the voltammetric measurement of neurotransmitter release and metabolism In vivo. J Neurochem 52: S19.
- Crespi F (1990) In vivo voltammetry with micro-biosensors for analysis of neurotransmitter release and metabolism. J Neurosci Methods 34: 53-65. [Crossref]
- Breese GR, Cott JM, Barrett R. Cooper BR, et al. (1975) Effects of Thyrotropin-Releasing Hormone (TRH) on the Actions of Pentobarbital and Other Centrally Acting Drugs. J Pharmacol Exp Ther 193: 11–22. [Crossref]
- Ghorbani A, Rakhshandeh H, Sadeghnia HR (2013) Potentiating Effects of Lactuca sativa on Pentobarbital-Induced Sleep. Iran J Pharm Res 12: 401-406. [Crossref]
- Crespi F, England T, Ratti E, Trist DG (1995) Carbon fibre micro-electrodes for concomitant In vivo electrophysiological and voltammetric measurements: no reciprocal influences. Neurosci Lett 188: 33-36. [Crossref]
- Crespi F (2011) Influence of Neuropeptide Y and antidepressants upon cerebral monoamines involved in depression: an In vivo electrochemical study. Brain Res 1407: 27-37. [Crossref]
- Crespi F (1991) In vivo voltammetry: detection of neuropeptides with micro carbon fibre biosensors: possible selective detection of somatostatin. Anal Biochem 194: 69-76. [Crossref]
- Crespi F (2010) SK channel blocker apamin attenuates the effect of SSRI fluoxetine upon cell firing in dorsal raphe nucleus: A concomitant electrophysiological and electrochemical In vivo study reveals implications for modulating extracellular 5-HT. Brain Res 1334: 1-11. [Crossref]
- Crespi F (2002) In vivo voltammetry and concomitant electrophysiology at a single biosensor to analyse ischaemia, depression and drug dependence. J Neurosci Methods 119: 173-184.
- Watson CJ, Baghdoyan HA, Lydic R (2010) Neuropharmacology of Sleep and Wakefulness. Sleep Med Clin 5: 513-528. [Crossref]
2021 Copyright OAT. All rights reserv



