The crystal of hydroxyapatite, Ca10 (PO4) 6 (OH) 2 has a characteristic that it varies easily depending on the acidic base environment around the enamel. The etymology of apatite is said to come from the Greek word meaning "to deceive" since it contains various elements in the Ca, PO4, and OH areas and changes appearance. In other words, the true character behind the demineralization and remineralization of apatite is the "deceptive" character of this crystal. Studies undertaken by this deceit have actually been carried out since the 1950's [1,2].
As shown by Ca10(PO4)6(OH)2+8H+→ 10Ca2+ + 6HPO42-+ 2H2O, while apatite variation due to H+ results in the elution of all ions, phosphoric acid ions go through a three stage dissociation of H2PO4-, HPO42-, PO43- due to H+ ions. The formula CaHPO4・2H2O out of that makes up brushite. In addition, Ca3(PO4)2 signifies whitlockite. Later studies caused this brush it and whitlockite to become known as caries crystals that are found in areas of decay [3]. This variation is reversible, and the aforementioned formulas progress to the left once H+. decreases. Hydroxyapatite is the most acid resistant among other calcium phosphates. However, while the solubility of hydroxyapatite itself is mostly zero at pH7, it is reported that elution occurs at pH4 for many minerals [4]. In short, the demineralization and remineralization of enamel is defined as occurring due to variation in the solubility of minerals within the solution (saliva). With tooth decay at a decline in the present day, understanding the variation occurring at subsurface enamel and performing remineralization treatment is becoming increasingly important in eliminating tooth decay altogether.
Although demineralization and remineralization at white spots are a response occurring at a space of around 200 to 300μm at the subsurface, this layer is abundant with F and Zn elements.
Enamel contains many trace elements, and after dividing enamel into three layers from the surface, turning each into powder and investigating the density of the trace elements, Brudevold and Soremark [5] reported that those with a high density at the surface were F, Zn, Pb, Fe, and Sn, those with mostly a uniform distribution were Sn and Cu, and those that increase in density at a deep level were CO2, Mg, and Na. It can be assumed that the abundance of F and Zn in the layer where subsurface demineralization occurs is in environments where these elements are easily captured in the process of remineralization when the environment improves even in the unlikely case that demineralization occurs.
The crystals fluorapatite and Ca10(PO4)6F2 form when F ions are replaced within the apatite crystal. The solubility of this crystal reportedly radically declines in accordance with an increase in the content of F [4]. F is an element which increases crystallinity, improves acid resistance, and actively works to form crystals through being captured by hydroxyapatite apatite together with Zn. Since the arrangement of elements in such enamel is common to the teeth of humans, dogs, and guinea pigs, it is concluded that it is determined by the progression of mineralization due to the tooth forming process and cell participation.
Although we cannot identify the mineral type using a μCT approach to search for the mineral content of enamel, results of scanning every 180μm of the tooth enamel of a sound bovine teeth up to 900μm (dentin-enamel junction) and comparing the density of all minerals demonstrated that the mineral content gradually decreased from the surface to deeper parts [6].
The principle behind detecting demineralization with QLF is based on the fact that a decreased intensity of dispersed fluorescence due to the morphological change of enamel crystals caused by demineralization is strongly correlated with the degree of demineralization measured with TMR (Transversal Microradiography). TRF requires that tissue be destroyed to be measured, but QLF has the benefit of being able to measure within the oral cavity without causing any damage. QLF cannot diagnose the relationship between the amount and depth of demineralization, however. As a result of soaking the enamel of a sound bovine tooth for 60 minutes in a pH5.5 solution and afterward performing a comparison between μCT and QLF measurements, it was shown that while the naked eye is unable to tell them apart from sound enamel (Figure 1), QLF shows the average demineralization depth (Figure 2). However, it is not understood at what depth and to what degree demineralization happens to enamel. Meanwhile, with μCT we can see that after slicing from the surface every 180μ, the Hoursfield unit value (mineral density) decreases the deeper the enamel depth (Figure 3). In this way, we see that while QLF perceives the demineralized portion of the enamel as one cluster and presents the total demineralized content, with μCT we can get a good 3D sensitive visualization of the lost mineral content.

Figure 1. Visual observation of the region demineralized for 60 minutes with a pH 5.5 solution.

Figure 2. QLF observation of the region demineralized for 60 minutes with a pH 5.5 solution.
ΔF: Average demineralization depth; ΔQ: Average demineralization amount; WS Area: Average demineralization area
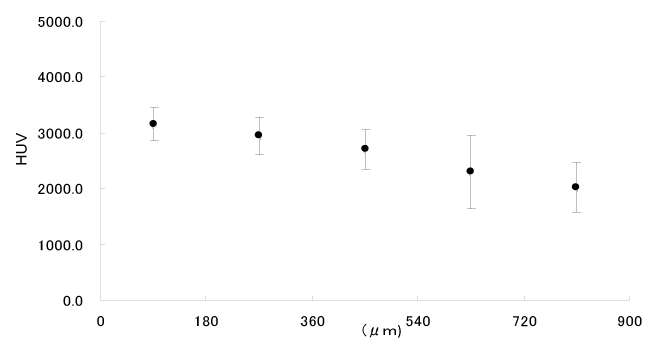
Figure 3. μCT observation of the region demineralized for 60 minutes with a pH 5.5 solution.
HUV: mineral density.
Although white spots often seen in a clinical setting display a subsurface demineralized state, we can consider them to be a fairly advanced beginning stage of decay. In other words, while white spots are affected by plaque over a long period of time and the surface evades demineralization with minerals obtained from saliva, there is a continual state of conflict between demineralization and remineralization at the subsurface. The aforementioned demineralization experiment which used bovine teeth was tooth erosion made by soaking it in a weak acid (pH5.5) solution for a short amount of time, but TMR confirms that this subsurface demineralization occurs even for decay caused by plaque [7]. This means that prevalent remineralization occurs when saliva and fluoride are provided in support to enamel. An area where white spots can be often observed within the oral cavity is the buccal surface of the premolars and the upper labial surface of the incisor, and a characteristic of this area is that it has the worst reach for saliva [8]. Thus, it is an environment with slow movement speed and low pH [9]. The carriers (minerals) for crystals at the enamel subsurface of such an area get caught or separate and are always moving around. Imagining such an image, why not try implementing remineralization treatment? The world is steadily progressing from scraping up enamel and packing it away to instead being romantic and microscopic.
- Gustafson (1957) The histpathology of caries of human dental enamel. Acta Odont Scand 15: 13-55.
- Darling AI (1958) Studies of the early lesion of enamel caries with transmitted light, polarized light and microradiography. Brit J 101: 289-329.
- Chow LC, Brown WE (1973) Reaction of dicalcium phosphate dehydrate with fluoride. J Dent Res 52: 1220-1227.
- Brown WE, Patel PR, Chow LC (1975) Formation of CaHPO4·2H2O from enamel mineral and its relationship to caries mechanism. J Dent Res 54: 475-481. [Crossref]
- Brudevold F, Soremark R (1967) Chemistry of the mineral phase of enamel from structural and chemical organization of teeth. Ed. By A.E.W.Miles, New York and London, Academic Press. Volume II, pp.247-277.
- Watanabe K, Nakamura T, Ogihara T, Ochiai Y, Watanabe S (2012 ) Longitudinal evaluation of mineral loss at the earliest stage of enamel demineralization using micro-computed tomography. Health 14: 334-340.
- Silverstone LM (1977) Remineralization phenomena. Caries Res 11: 59-84. [Crossref]
- Watanabe S (1992) Salivary clearance from different regions of the mouth in children. Caries Res 26:423-427. [Crossref]
- Dawes C, Watanabe S, Biglow-Lecomte P, Dibdin GH (1989) Estimation of the velocity of the salivary film at some different locations in the mouth. J Dent Re 68: 1479-1482. [Crossref]



