We present the immunopathogenesis of Lymphomatoid Papulosis associated with mild immunosuppression. The foundation of our observation was based on case reports discussing the development of LyP while concurrently using immune modulators. The medically altered immune state is mirrored by the hormone-induced changes of pregnancy. Furthermore, we explain how the natural progression of pregnancy facilitates a pathologic transformation of LyP in rare individuals. This is only the second reported case of pregnancy induced LyP. To clarify the findings, we include a basic review of Lymphomatoid Papulosis with key points of immunology related to both pathology and pregnancy.
A 35-year-old Caucasian female G4P4 at week one post partum with no significant past medical history was referred to our clinic for evaluation and treatment of right thigh dermatitis. Reported history suggested a recurrent dermatitis in the same location of which she believed the papules had been larger during the previous flare. Physical exam confirmed the lesions were localized to the distal right thigh with a grouping of four 2-3 mm erythematous macules, the larger papule with scales (Figure 1). Initial presentation and history placed prurigo nodularis as a top differential. A biopsy was agreed upon and later demonstrated the presence of CD4 cells, greater than 50% CD30 positive (Figure 2). A diagnosis of Lymphomatoid Papulosis (LyP) was suggested from the pathology, which correlated to her clinical picture. For further confirmation, gene rearrangement studies were requested from the biopsy.
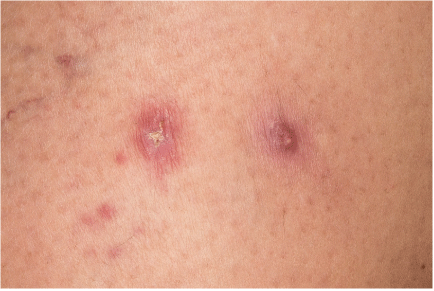
Figure 1. Right thigh image demonstrating erythematous macules and papules with scale and central ulceration.
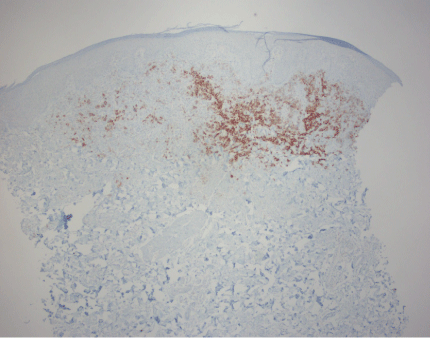
Figure 2. CD30 Immunohistochemical stain at 40X
The patient returned to clinic for Grand Rounds and further evaluation two weeks later. The patient denied systemic symptoms including fatigue, fever, chills or additional skin lesions. Additional medical history revealed an enlarged thyroid under investigation by her obstetrician. The skin lesion was predominantly unchanged with the previous biopsy site healing well. A second biopsy was obtained, which failed to demonstrate the abundance of CD4 cells. Subsequently, the original biopsy gene study resulted demonstrating a clonal T-Cell Receptor Beta (TCRB) with a clonal size of 263BP and 319BP. Clinical presentation followed by the regression of disease and prior positive biopsy confirmed the diagnosis of LyP, specifically subgroup C (Figures 3 and 4).
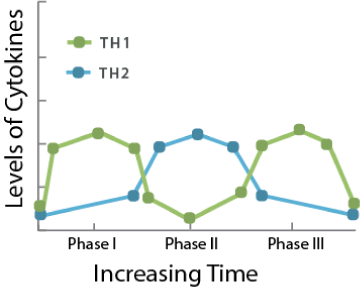
Figure 3. Immune Response during Pregnancy. Note the predominance of Th1 at the early and late phases and Th2 during the middle phase.
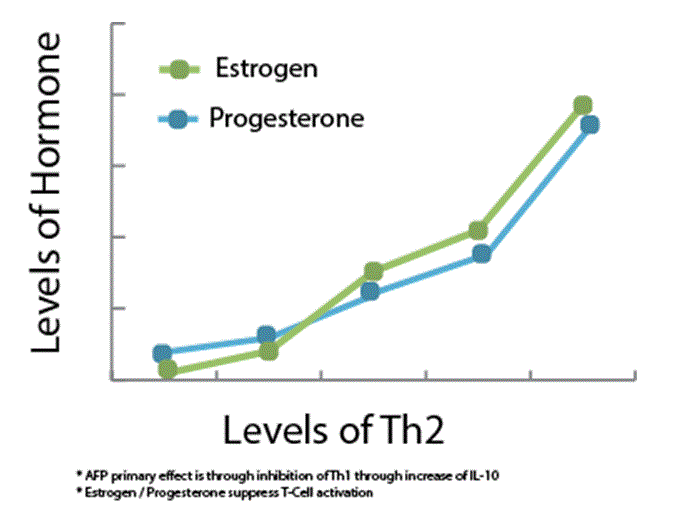
Figure 4. Hormonal Induced Immune Suppression. Estrogen and Progesterone play the key roles, while hCG and AFP also contribute as supportive roles.
The patient and 4)group Clowed by the regression of disease and prior positive biopsy confirmed the Ultrasound images of the thyroid noted a 5.1 cm nodule in the left lobe. The patient was later evaluated by fine needle aspiration and found negative for thyroid malignancy.
LyP during pregnancy has only been documented once previously. In Yamamotognancy has only been documented once previously.ly.found negatiLyP eruption [1]. Similarly, our patient noted a localized papulosquamous eruption during her recent pregnancy. Her prior dermatologic eruptions may or may not have been associated with other gestations; the timing and duration of prior eruptions were unclear. For this reason, our patient likely had an undiagnosed or chronic LyP that was unmasked during pregnancy.
LyP is a rare disease with an incidence less than 2:1,000,000 people. Its a rare disease with an incidence less than 2:1,000,000 people.le.ns may or may not have been associated with other gers to over a decade [2,3]. While LyP is self-limiting, 10-year survival rate of 100% [3], there is a strong correlation of LyP with the development of other lymphomas [4]. Approximately 40% of LyP may progress to another form of lymphoproliferative disorder with a predominance to mycosis fungoides (MF) [4]. Additional significant associations for LyP have been found with endocrinopathies and thyroid nodules [1,5].
Histologically, LyP can be further subcategorized into 4 groups. Group A is noted to be of mixed infiltrate with a large population of predominately atypical CD30 positive cells. Group B has smaller atypical T lymphocytes and convoluted nuclei with a similar histologic presentation to MF. Group C, as noted in our patient, is often found to have large groupings of CD30 positive suggestive of an anaplastic lymphoma. Lastly, in more recent literature, the fourth subtype has been presented. Type D, where the presentation is histologically related to epidermotropic CD8 positve T-Cell lymphoma and CD30 negative [6].
Looking past the histopathological diagnosis, the immunopathogenesis of LyP may assist in unifying several key case reports in the literature. Three cases have been published describing therapies that invoked or exacerbated LyP through the use of an immune modulator: infliximab (TNF-imab (TNF- of arituximab (anti-CD20 in B-cells), and fingolimod (inhibit cytotoxic CD8 T-cells) [7-9]. These cases cover LyP correlated with pathologic autoimmune response [7,9] and by immunosuppressive treatment for Chronic Lymphocytic Leukemia [8]. The immunological changes occurring in the above cases can be compared with the changes associated in pregnancy. We will cover the expected immune response in more detail to clarify pregnancy inducing LyP [1].
Often times the mechanism behind clinical presentation is misunderstood. Wong et al clarifies key aspects of immunopathogenesis with similar cutaneous lymphoproliferative disorders such as Cutaneous T-Cell Lymphomas (CTCL), specifically Mycosis Fungoides (MF) and Sezary Syndrome (SS) [10]. His research proposes that a progenitor T-cell (MF/SS) is capable of proliferation under the right conditions. These pathological processes induce shifts within the Th1/Th2 system; similar changes are appreciated through hormonal modulation of the immune system during pregnancy.
In MF/SS, the progenitor T-Cell within the skin demonstrates normal to increased expressions of inflammatory Th1 Cytokines during the early phases. The key cytokines of IL-2, IFN-ithin the skin demonstrates normal to increased ex The increased levels of IL-2 up-regulate CD28 further allowing T-cell proliferation. As the malignancy advances, the malignant CD4 T-cell lines predominate causing a shift from the Th1 pro-inflammatory state to a Th2 anti-inflammatory state. Increasing levels of IL-4, IL-5, IL-10, and IL-13 govern this transition. The increasing levels of IL-10, and decrease in IFN- CD4 T-cell lcytotoxic system further enable the shift towards Th2 predominance. The reduced anti-tumor response allows the malignancy to evade the immune system [10].
Similarly, the early immune phase of pregnancy begins with an increase in the Th1 dominant system. The associated pro-inflammatory cytokines allow for implantation and early development. As the pregnancy continues, a shift towards the Th2 system is favored allowing for a period of permissive immunotolerance of an allogenic fetus to the maternal system [11-13]. Pregnancy immune suppression is guided by increased levels of gonadotropins, particularly progesterone, estrogen, hCG, and AFP. The excess estrogen and progesterone favor the Th2 pathway by potentially suppressing T-cell activation and inducing effector T-cell apoptosis [11,12]. The role of T-regulatory cells in cancer pathology is divided due to its ability for both propagation and destruction of malignancies. Although the evidence is conflicting towards the exact mechanism, it is agreed that progesterone and estrogen are involved in the regulation of regulatory T-cells during pregnancy [12]. Given the general shift of the Th2 system and immunotolerance during middle phases of pregnancy, T-regulatory cells are likely to be up-regulated during pregnancy allowing for propagation of LyP T-cells [13]. As mentioned previously, hCG and AFP are increased during this period. The role of hCG is reduction of IFN-FN-tion of [12] which are involved in tumor suppression via apoptosis and cytotoxic mediated tumor destruction. AFP further assists with the suppression of TNF-α. Simultaneously, hCG also increases levels of IL-10 [12], further suppressing the cytoxic Th1 pathway. With our current understanding, it can be appreciated that faulty T-cells in conjunction with immunosuppression facilitates induction of LyP in rare individuals. By manipulating mediators of tumor suppression the immune system permits development of the fetus. It is during the same time where the pathological propagation, such as LyP, manifests. As the pregnancy hormones normalize after childbirth, so does the Th1/Th2 system. At this point, as indicated by our patient, the immune system is able to resume normal function and suppress activity of pathology such as LyP [14].
In addition to immune suppression, as in therapy for treatment of many dermatologic diseases, pregnancy is also a time of immunologic change. While the majority of pregnancies do not present with pathology, it is important to remember that this period of altered immune function may be an opportunity for disease to emerge. Consideration for biopsy should be addressed in select patients that present with recurrent or persistent dermatological processes. While LyP may be benign, a small percentage may go on to develop lymphoma, endocrinopathies or pathology that may require additional patient education and monitoring.
The views expressed in this article are those of the author(s) and do not necessarily reflect the official policy or position of the Department of the Navy, Department of Defense or the United States Government.
We are military service members or employees of the Federal Government. This work was prepared as part of our official duties. Title 17 U.S.C. 105 provides that ‘Copyright protection under this title is not available for any work of the United States Government.’ Title 17 U.S.C. 101 defines a United States Government work as a work prepared by a military service member or employee of the United States Government as part of that person’s official duties.
- Yamamoto O, Tajiri M, Asahi M (1997) Lymphomatoid papulosis associated with pregnancy. Clin Exp Dermatol 22: 141-143. [Crossref]
- Wang HH, Lach L, Kadin ME (1992) Epidemiology of lymphomatoid papulosis. Cancer 70: 2951-2957. [Crossref]
- Wieser I, Oh CW, Talpur R, Duvic M (2016) Lymphomatoid papulosis: Treatment response and associated lymphomas in a study of 180 patients. J Am Acad Dermatol 74: 59-67. [Crossref]
- de la Garza Bravo MM, Patel KP, Loghavi S, Curry JL, Torres Cabala CA, et al. (2015) Shared clonality in distinctive lesions of lymphomatoid papulosis and mycosis fungoides occurring in the same patients suggests a common origin. Hum Pathol 46: 558-569. [Crossref]
- Sanchez NP, Pitte2021 Copyright OAT. All rights reservann RK (1983) The clinicopathologic spectrum of lymphomatoid papulosis: study of 31 cases. J Am Acad Dermatol 8: 81-94. [Crossref]
- Cardoso J, Duhra P, Thway Y, Calonje E (2012) Lymphomatoid papulosis type D: a newly described variant easily confused with cutaneous aggressive CD8-positive cytotoxic T-cell lymphoma. Am J Dermatopathol 34: 762-765. [Crossref]
- McCurdy O, McCormack C, Ritchie D, Prince HM (2014) Exacerbation of lymphomatoid papulosis during rituximab therapy. Australas J Dermatol 55: e1-3. [Crossref]
- Samaraweera AP, Cohen SN, Akay EM, Evangelou N (2016) Lymphomatoid papulosis: A cutaneous lymphoproliferative disorder in a patient on fingolimod for multiple sclerosis. Mult Scler 22: 122-124. [Crossref]
- Spires N, McGibbon D (2009) Lymphomatoid papulosis improving on hormone-replacement therapy. Clin Exp Dermatol 34: 635-636. [Crossref]
- Wong HK, Mishra A, Hake T, Porcu P (2011) Evolving insights in the pathogenesis and therapy of cutaneous T-cell lymphoma (mycosis fungoides and Sezary syndrome). Br J Haematol 155: 150-166. [Crossref]
- Enninga EA, Holtan SG, Creedon DJ, Dronca RS, Nevala WK, et al. (2014) Immunomodulatory effects of sex hormones: requirements for pregnancy and relevance in melanoma. Mayo Clin Proc 89: 520-535. [Crossref]
- Schumacher A, Costa SD, Zenclussen AC (2014) Endocrine factors modulating immune responses in pregnancy. Front Immunol 5: 196. [Crossref]
- Mor G (2006) Immunology of pregnancy. Georgetown, Tex, New York: Landes Bioscience/Eurekah.com; Springer Science+Business Media.
- Williams T, Tsien B (2010) The physical space of science: the Neurosciences Institute and Skirkanich Hall. Cell Cycle 9: 28-31. [Crossref]




