Abstract
Reported cases of stroke attributed to calcified cerebral emboli are rare. The response to thrombolytic therapy in these cases compared to other causes of stroke is uncertain. We present five new cases with a discussion on the natural history and treatment of stroke secondary to calcified cerebral emboli. In our case series three patients were managed conservatively, while one patient received intravenous thrombolysis and one patient underwent intra-arterial mechanical thrombectomy.
Key words
stroke, thrombectomy, embolism
Introduction
Reported cases of calcific cerebral emboli are limited but have become more prevalent in the literature in recent years. It is unclear whether the current best management for stroke secondary to non-calcified thrombus is also the optimum treatment for patients with stroke secondary to calcified cerebral emboli. We present five cases to review the (i) imaging findings (ii) clinical management and (iii) response to therapy including intra-venous thrombolysis and intra-arterial thrombectomy in patients with stroke secondary to calcified cerebral emboli.
Often considered a rare cause of stroke, there is now a suggestion that calcified emboli may be a more common aetiology than previously thought. In an institutional review of all stroke Computed Tomography (CT) scans over a period of twelve months, Walker et al. found the prevalence of calcified cerebral emboli to be 2.7% [1]. The same review also found that 27% of cases identified on review were misdiagnosed on non-contrast CT (NCCT) as various alternative pathologies and a further 9% overlooked on initial interpretation [1]. One case report also noted that the initial NCCT was reported as normal [2]. While the majority of cases present as acute non-resolving neurological deficit, one case report described the unusual presentation of multiple episodes over two days of expressive aphasia and right arm and leg weakness, each episode lasting 10-20 minutes and spontaneously resolving [2].
Case reports to date reveal two distribution patterns of calcific emboli at presentation. The most common is a central calcific embolus within the major intracranial vasculature including the internal carotids, A1, P1, M1 or M2 segments. Less commonly, multiple punctate calcific densities within the pial arteries are seen, referred to as the ‘salted pretzel sign’ [3,4]. Initial imaging protocol in this patient group at presentation usually includes non-contrast CT with head and neck CT angiography. This enables visualization of central and pial calcific emboli and can identify potential sources of calcific emboli. Magnetic Resonance Imaging (MRI) allows for a more complete evaluation of the infarcted brain parenchyma. In cases of peripheral pial calcific emboli, non-contrast CT may greatly underestimate the volume of brain parenchyma at risk and hence MRI imaging with diffusion weighted sequences is recommended.
We present five new cases and a review of the natural history and the response to thrombolytic therapy in stroke attributed to calcified cerebral emboli in the literature to date. In our case series an initial non-contrast CT brain was performed, followed by appropriate adjunctive imaging guided by the clinical circumstances. Three patients were managed conservatively, while one patient received intravenous thrombolysis and intra-arterial mechanical thrombectomy was performed for one patient.
Case series
Case 1
A 72-year-old lady presented with aphasia and left sided hemiparesis, on a background of a transient neurological deficit one month previously. Her National Institute of Health Stroke Scale (NIHSS) [5] score was 17 at presentation. CT-brain and CT intracranial angiogram demonstrated an occluded right M1 segment secondary to a large calcified embolus, a new finding compared to previous CT (Figures 1a & 1b) Intravenous thrombolytic therapy was not administered due to the presence of subarachnoid haemorrhage on CT one month previously. The patient was transferred for angiogram and clot retrieval 75 minutes after symptom onset. Three passes of a Trevo ProVue Retreiver and three passes of a Solitaire Revascularization Device were required for recanalization of the vessel. (Figures 1c & 1d) Radiograph of the removed embolus was acquired following the procedure confirming calcium content. (Figure 1e) There was minimal clinical improvement, NIHSS score was 15 at both one hour and twenty-four hours post procedure. Follow-up non-contrast CT brain at 24 hours showed an evolving large infarct within the right middle cerebral artery (MCA) vascular territory.
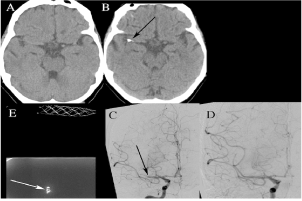
Figure 1. (A,B) CT-brain and CT angiogram demonstrate an occluded right M1 segment secondary to a large calcified embolus. (C,D) Angiogram demonstrating recanalization of the vessel following passage of a solitaire revascularization device. (E) Screening of the removed embolus post procedure confirms calcium content.
Case 2
A 63-year-old gentleman presented with right sided headache and left sided hemiparesis. His NIHSS score was 13 on admission. CT-brain demonstrated multiple calcific emboli within the pial arteries in the posterior right MCA territory. CT angiogram showed high grade stenosis of the proximal right internal carotid artery and patent proximal segments of the right MCA. The patient was treated with standard intravenous thrombolytic therapy. NIHSS four hours post treatment was 11. Follow-up non-contrast CT-brain at 24 hours demonstrated established infarcts in the right parietal and frontal lobes (Figures 2a & 2b, black arrows) and with an adjacent small volume convexal subarachnoid haemorrhage (Figure 2b, white arrow). Multiple calcific emboli were again demonstrated within the pial arteries in the posterior right MCA territory, unchanged from initial non-contrast CT performed on presentation. MRI brain showed diffusion weighted imaging (DWI) and apparent diffusion coefficient (ADC) map signal changes consistent with multi-focal infarction in the right parietal and frontal lobes. (Figures 2c & d) Susceptibility artefact was evident on gradient echo sequences due to the right parietal haemorrhage. (Figure 2e) There was significant residual neurological deficit at three month follow up.
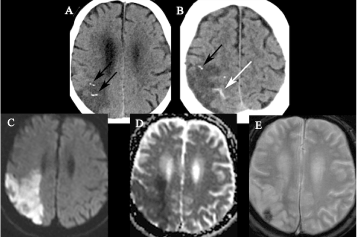
Figure 2. (A,B) Non-contrast CT brain performed 24 hours after presentation with left sided hemiparesis demonstrates established infarcts in the right parietal and frontal lobes (black arrows) and with an adjacent small volume convexal subarachnoid haemorrhage (white arrow). (C,D) MRI brain shows DWI and ADC map signal changes consistent with multi-focal infarction within the right parietal and frontal lobes. (E) Gradient echo sequences show evidence of susceptibility artefact due to the right parietal haemorrhage.
Case 3
An 81-year-old lady presented with acute dysphasia and right sided weakness. CT-brain showed loss of grey white matter interface and sulcal effacement within the left temporal lobe with an adjacent pial artery calcific embolus. (Figure 3a, arrow) The patient did not receive intravenous thrombolytic therapy as she presented outside the time window for treatment. Follow-up MRI demonstrated a region of acute infarction within the left temporal lobe with restricted diffusion. (Figures 3b & 3c) At three month follow-up the patient had significant residual neurological deficit with disability.
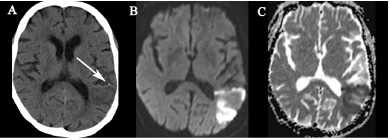
Figure 3. (A) CT-brain demonstrating loss of grey white matter interface and sulcal effacement within the left temporal lobe with an adjacent pial artery calcific embolus (arrow). (B,C) Follow-up MRI demonstrates a region of acute infarction within the left temporal lobe with restricted diffusion.
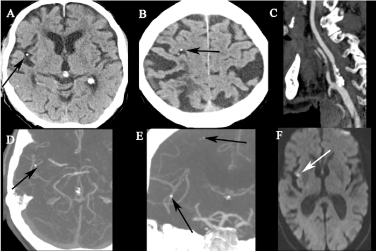
Figure 4. (A,B) CT-brain demonstrates a calcific emboli within the right M2 vessels and within the pial arteries of the right frontal lobe. (C,D,E) CT angiogram demonstrates heavily calcified atheroma at the origin of the right internal carotid artery and calcific emboli present within the distal contrast enhanced arteries. (F) Follow-up MRI demonstrates a small punctate site of restricted diffusion within the insular cortex adjacent to the calcific embolus within the right M2 vessel.
2021 Copyright OAT. All rights reserv
Case 4
An 81-year-old male presented with acute left hemiparesis and dysarthria. His NIHSS score at presentation was 7. CT-brain showed calcific emboli within the right M2 vessels and within the pial arteries of the right frontal lobe. (Figures 4a & 4b) CT angiogram showed heavily calcified atheroma at the origin of the right internal carotid artery, considered the likely source of the emboli. (Figure 4c-e) The patient did not receive intravenous thrombolytic therapy as the time of symptom onset was unclear. Within one hour of presentation there was an improvement in the patient’s neurological deficit and his NIHSS score decreased to 1. Follow-up MRI showed a small punctate site of restricted diffusion within the insular cortex corresponding to the calcific embolus within the right M2 vessel (Figure 4e).
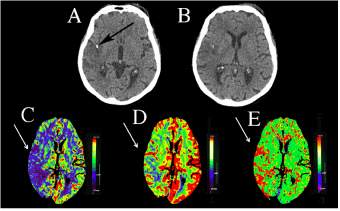
Figure 5. (A,B) CT demonstrates a calcified embolus within the right M2 vessel and evolving infarction within the right frontal lobe. (C - E) Perfusion CT including colour map of (C) Cerebral Blood Flow which is decreased in the right MCA distribution, (D) Cerebral Blood Volume which is reduced in the right MCA territory and (E) Mean Transit Time (MTT) which demonstrates prolonged MTT. A large perfusion defect is demonstrated in the right MCA territory.
Case 5
An 84-year-old female presented with a ‘wake-up’ left hemiparesis and a NIHSS score of 12. CT-brain showed a calcified embolus within the right M2 vessel and an evolving infarction within the right frontal lobe (Figures 5a & b). The patient did not receive intravenous thrombolytic therapy as CT perfusion confirmed an established infarct. (Figures 5c-e) The patient has a persistent neurological deficit.
Discussion
Possible sources of emboli identified include aortic valve calcification, mitral annulus calcification, calcified aortic arch and carotid atherosclerotic plaque [6]. Rare sources of embolism are brachiocephalic and vertebral artery atherosclerotic plaques [1]. It was previously speculated that the majority of cases of calcified cerebral emboli were non-spontaneous, provoked by mechanical interruption of the embolic source [7].However, in a statistical analysis of 70 cases which included 22 cases identified in an institutional review and 48 published case reports, it was found that 86% of cases were spontaneous while 9% followed left heart catheterization, 3% followed carotid artery manipulation, and 3% followed cardiopulmonary resuscitation [1]. All five cases presented in our case series had no provoking mechanical or traumatic event.
Intravenous thrombolytic therapy is widely used in the treatment of acute ischaemic stroke but there are limited reports of its use in stroke caused by calcified cerebral emboli, with mixed outcomes [8]. 9 of 22 new cases published by Walker, et al. received intravenous thrombolysis [1]. Of those, four were reported to have had symptomatic improvement. There are a further seven cases identified in our review describing thrombolysis use in stroke cause by calcified emboli. Three cases report a fatal outcome [6,9,10]. In one of these cases a post treatment NCCT showed a new calcified density in the basilar artery [10]. The remaining four cases resulted in symptomatic improvement [10-13]. Two of these showed distal migration of the embolus on follow up imaging [1,10]. The other two cases showed fragmentation of the embolus with dispersion of the fragments to the pial arteries [11,13]. A 63 year old male who presented acutely to our institution with left hemiparesis (Case 2) is the first reported case treated with intravenous thrombolysis where the patient presented with multiple peripheral pial artery calcific emboli on the initial CT brain. However, the patient’s functional outcome following treatment was disappointing. No peripheral embolic dispersion or fragmentation was identified on follow up NCCT, however given the initial relatively peripheral distribution of emboli, unchanged vascular distribution was not unexpected.
A 72 year old female who presented with a left hemiparesis and aphasia (Case 1), is to the best of our knowledge, the first described case to date of intra-arterial thrombectomy in acute stroke secondary to a calcified cerebral embolus. There was excellent technical success, the calcified embolus was removed intact and the vessel was successfully canalized. Despite this, the clinical outcome was disappointing and the post procedure NIHSS score remained unchanged at 15. Follow-up CT imaging showed an established infarct within the right MCA vascular distribution.
Conclusion
The reported outcomes in literature to date regarding use of intravenous thrombolytic therapy in cases of calcific cerebral emboli are mixed. Interestingly all four case reports of successful outcomes post intravenous thrombolysis report the dispersion of central calcific emboli into the peripheral pial vessels or distal migration of the embolus on post thrombolysis CT brain.
On CT imaging calcified emboli are identified in large or medium central vessels or peripherally within the pial arteries giving the ‘salted pretzel sig’ [3]. Diffusion weighted MRI can show acute infarction in the vascular distribution of the affected vessel.
Our experience of thrombectomy demonstrates the ability to successfully recanalize a vessel occluded with a calcified embolus. Unfortunately in our presented case of mechanical thrombectomy the neurological deficit was unchanged. Reported clinical outcomes, following the use of intravenous thrombolytic therapy have been mixed. Migration of central emboli or fragmentation and dispersion to the pial arteries on post thrombolysis CT brain appears to correlate with a favourable clinical outcome. Currently there is limited published literature; in the first instance it is imperative to establish the true prevalence of symptomatic acute calcific emboli in acute stroke patients, to establish risk factors and the natural history and to establish the efficacy of conventional management of acute stroke in this cohort. We suggest that intra-arterial mechanical thrombectomy is a valid treatment option for stroke syndromes secondary to acute calcified emboli. Further research in this area is necessary to establish the optimum treatment algorithm for this unique syndrome, and the role for mechanical thrombectomy which we suggest is likely a pivotal component.
References
- Walker BS, Shah LM, Osborn AG (2014) Calcified cerebral emboli, a "do not miss" imaging diagnosis: 22 new cases and review of the literature. AJNR Am J Neuroradiol 35: 1515-1519. [crossref]
- Gearry RB, Sharr JP, Avery SF (2005) Spontaneous calcific cerebral embolus. Australas Radiol 49: 154-156. [crossref]
- Christian BA, Kirzeder DJ, Boyd J, Laing J, Gash J (2009) Showered calcific emboli to the brain, the 'salted pretzel' sign, originating from the ipsilateral internal carotid artery causing acute cerebral infarction. Stroke 40: 319-321. [crossref]
- Deng Q, Zhang Y, Ding H, Dong Q, Fu J (2015) Calcific emboli originating from the brachiocephalic trunk causing acute cerebral infarction and worm-like calcification in the right middle cerebral artery. J Clin Neurosci 22: 889-890. [crossref]
- Brott T, Adams HP Jr, Olinger CP, Marler JR, Barsan WG, et al. (1989) Measurements of acute cerebral infarction: a clinical examination scale. Stroke 20: 864-870 [crossref]
- Gokhale S, Lahoti S, Rojas R, Caplan LR (2013) Tissue plasminogen activator and calcific emboli. JAMA Neurol 70: 642-643. [crossref]
- Vernhet H, Torres GF, Laharotte JC, Tournut P, Bierme T, et al. (1993) Spontaneous calcific cerebral emboli from calcified aortic valve stenosis. J Neuroradiol 20: 19-23. [crossref]
- The National Institute of Neurological Disorders and Stroke rt-PA Stroke Study Group (1995) Tissue plasminogen activator for acute ischemic stroke. N Engl J Med 333: 1581–1588. [crossref]
- Halloran JI, Bekavac I (2004) Unsuccessful tissue plasminogen activator treatment of acute stroke caused by a calcific embolus. J Neuroimaging 14: 385-387. [crossref]
- Kissela BM, Kothari RU, Tomsick TA, Woo D, Broderick J (2001) Embolization of calcific thrombi after tissue plasminogen activator treatment. J Stroke Cerebrovasc Dis 10: 135-138. [crossref]
- Gschwind M, Binaghi S, Zekeridou A, Michel P (2013) Neurological picture. Dispersion and 'salted pretzel sign' from thrombolysis of a spontaneous calcified embolus in an acute stroke. J Neurol Neurosurg Psychiatry 84: 111-112. [crossref]
- Acha O, Novo F, Espina B, Marco E, Rebollo M, et al. (2009) Spontaneous cerebral calcific embolus from the aortic arch. Clin Neurol Neurosurg 111: 862-863. [crossref]
- Kavanagh EC, Fenton DM, Heran MK, Lapointe JS, Nugent RA, et al. (2006) Calcified cerebral emboli. AJNR Am J Neuroradiol 27: 1996-1999. [crossref]





