Purpose: Arterial waveform-derived dynamic variables, such as stroke volume variation (SVV), are useful for predicting fluid responsiveness in perioperative patients. Since specific catheters and machines must be used for measuring SVV, we tried to find a novel indicator that correlates with SVV values and can be calculated using usual monitoring equipment.
Methods: A total of 50 patients admitted to the ICU, both intubated (n=25) and non-intubated (n=25), were enrolled in this prospective observational study. Intubated patients received mechanical ventilation under pressure controlled ventilation (PCV) or continuous positive airway pressure /pressure support ventilation (CPAP/PSV) modes. SVV values were used for assessment of cardiac preload. Besides SVV, central venous pressure (CVP) and peripheral venous pressure (PVP) were simultaneously recorded. In each patient, 10 data pairs were randomly acquired at 1 hour or longer intervals.
Results: Regression analysis showed a slightly weaker but substantial association between PVP and CVP (Pearson's correlation coefficient r=0.91, p<0.0001). However, we found that PVP-CVP difference values correlated with SVV (r=0.64, p<0.0001). Moreover, we were able to predict whether SVV was normal (≤ 10%) or high (>10%) from PVP-CVP difference values with high accuracy. This prediction was not affected by the respiratory mode.
Conclusion: The difference between PVP and CVP might serve as a novel indicator of the adequacy of cardiac preload that does not require specific equipment, such as the FloTrac/Vigileo Monitor®.
central venous pressure, peripheral venous pressure, stroke volume variation, fluid responsiveness, intensive care unit
The main purpose of fluid therapy during severe invasive situations, such as after major surgery and in patients with sepsis, is to maintain tissue perfusion by optimizing intravascular volume. However, since there are only few methods to precisely measure cardiac preload in real time at the patient’s bedside, physicians often subjectively determine the volume of fluid required based on their experiences. On the other hand, several objective indicators to estimate cardiac preload have been developed and used, such as central venous pressure (CVP) and stroke volume variation (SVV). In general, the performance of dynamic indicators including SVV is reportedly better than that of static indicators [1-3]. For example, SVV can predict fluid responsiveness with both high sensitivity and specificity [4-6]. However, since special catheters and expensive machines, such as the FloTrac/Vigileo Monitor® system, must be used to measure SVV, not all facilities can perform this measurement. This consideration prompted us to estimate cardiac preload by measuring and comparing two static indicators, peripheral venous pressure (PVP) and CVP.
PVP reportedly correlates highly with CVP under stable hemodynamic conditions, and the fluctuations in these values occur in parallel. Indeed, the correlation coefficient between them was greater than 0.9 in many previous studies [7-10]. Conversely, the correlation coefficient between them is much smaller under unstable hemodynamic conditions [11]. Based on these evidences, we conducted a pilot study and noticed the possibility that the relationship between PVP and CVP values could depend on cardiac preload. We then hypothesized that we could predict fluid responsiveness by calculating the difference between PVP and CVP. To test this hypothesis, we compared the values of PVP - CVP difference between normovolemic and hypovolemic patients.
Ethical approval was obtained from the Ethics Committee of Gunma University, and written informed consent was collected from all patients. The study was registered with the UMIN Clinical Trials Registry (UMIN000014235)
This study included 50 patients who were prospectively enrolled and followed up from June 2014 to April 2015. The patients included postoperative patients and those admitted to the ICU as emergency cases. The inclusion criteria were patients >20 years old who gave consent for study participation. Patients with existing central, peripheral, and arterial lines with FloTrac® sensor monitoring kits (Edwards Life Sciences, Irvine, CA) or lines that were placed as part of their routine clinical care were selected. Exclusion criteria were the existence of cardiac diseases (myocardial infarction, unstable angina, and valvular disease) and atrial fibrillation. Patients with artificial arterio-venous fistulas for hemodialysis were also excluded. In addition, patients who required blood purification or use of a heart lung machine were excluded. Intubated patients were mechanically ventilated using pressure controlled ventilation (PCV) or continuous positive airway pressure and pressure support ventilation (CPAP/PSV) modes. Additionally, more than 5 cmH2O PEEP was added in both ventilatory settings. Mechanical ventilation was provided by either Bennett 840 (Puritan Bennett, CA, USA) or Engström Carestation (GE Healthcare, Dublin, Ireland) ventilators. Non-intubated patients were completely free from the ventilator, and breathed by themselves.
Investigation protocol
The distal port of a 12-Fr, triple-lumen central venous catheter (CVC, Covidien, Dublin, Ireland) or that of a 4.5-Fr, double-lumen peripherally inserted central catheter, (PICC, Covidien, Dublin, Ireland) was used to measure CVP. The CVC was inserted via the right internal jugular vein or the femoral vein, whereas the PICC was inserted via the brachial vein. Chest radiography was used to confirm placement of the tip of the CVC or PICC in the thoracic cavity. A 22-G arterial cannula was inserted into the radial artery for continuous monitoring of arterial pressure and a peripheral intravenous catheter (either 20 or 22 G) was placed in a distal forearm vein. All catheters were connected to a continuous pressure monitoring kit (Argon Medical Device, Plano, TX). The patients were placed in the supine position with their arms outstretched by their side. CVP and PVP were sequentially measured using a 3-way stop-cock device such that a single, common transducer was utilized for both measurements. The pressure transducer was positioned at the level of the right atrium and zeroed to atmospheric pressure just before the first reading in each patient. The waveforms of CVP and PVP were confirmed to show a display before every measurement. If the CVP and PVP wave traces were not satisfactory, tubing connections were checked and the line was flushed until a clear waveform appeared. Fluids and drugs were not administered through the lines used for pressure measurements. The FloTrac® arterial pressure sensor monitoring kit and the Vigileo monitor® (Edwards Life Sciences, Irvine, CA) were used to monitor arterial pressure and measure SVV for evaluating cardiac preload. CVP and PVP values were recorded in the end-expiratory phase. The concentrations of inspired and expired carbon dioxide were measured by capnometry (Intellivue, Royal Philips, Amsterdam) to confirm the respiratory cycle in intubated patients. We observed the movement of the thorax in non-intubated patients for the same purpose. A total of 10 data pairs were noted for each patient, as described in previous studies [10,12]. CVP and PVP were recorded as the mean of single values approximated to the nearest 1 mmHg. Data pairs were recorded under stable conditions, with an interval of at least 1 h for three consecutive measurements.
Statistical analysis
A priori power calculations for α=0.05 and an effect size of 0.5 (based on a two-tailed t test to calculate the mean PVP-CVP value) resulted in a sample size estimate of 34 for an 80% power. The effect size estimate was derived from our pilot study, which demonstrated that the mean values of PVP-CVP difference and standard deviation were 1.5 and 3 mmHg, respectively.
Data are expressed as mean ± standard deviation unless otherwise specified. Pearson’s correlation analysis was performed to examine the correlation coefficient. Mann-Whitney U test was used to compare the value of PVP-CVP difference between the normal-SVV state and high-SVV state. ROC curve analysis was used to determine the cutoff value of PVP-CVP difference. One-way ANOVA, unpaired t-test, or Mann-Whitney U test was used for comparisons between groups. All statistical analyses were performed using GraphPad Prism 6 software (GraphPad Software Inc, La Jolla, CA). P<0.05 was considered to indicate a significant difference.
The characteristics of the patient population are summarized in Table 1. Of the 50 patients enrolled in this study, 21 subjects were postoperative patients, and 29 patients were admitted to the ICU as emergency cases. A total of 500 data pairs were collected. Ventilator settings in intubated patients were not changed during the measurement period. Data pairs were acquired when the patients were in a stable condition, with a respiratory frequency of 20 breaths/min or less, and systolic blood pressure of 80 mmHg or more.
Table 1. Characteristics of the subjects. Data are represented as mean ± standard deviation. ARDS: Acute respiratory distress syndrome; CVC: Central venous catheter; PICC: Peripherally inserted central catheter.
Gender (M / F) |
37 / 13 |
Age (years) |
68 ± 12 |
Height (cm) |
161 ± 9 |
Body weight (kg) |
59 ± 12 |
Reason for ICU admission |
Postoperative |
Liver resection |
5 |
Complete esophageal resection |
13 |
Pheochromocytoma resection |
2 |
Total resection of pelvic organs |
1 |
Emergency admission |
Sepsis |
10 |
Respiratory insufficiency (ARDS) |
4 |
Aspiration pneumonitis |
2 |
Bacterial pneumonia |
4 |
Diabetic ketoacidosis |
1 |
Trauma |
2 |
Burns |
1 |
Transient ischemic attacks |
1 |
Tumor lysis syndrome |
1 |
Gastrointestinal perforation |
1 |
Accidental hypothermia |
1 |
Aortic dissection |
1 |
CVC insertion site |
Internal jugular vein |
26 |
Subclavian vein |
1 |
Femoral vein |
9 |
PICC insertion site |
Brachial vein |
14 |
Peripheral catheter sizes |
20 gauge |
7 |
22 gauge |
43 |
First, we investigated the relationship between CVP and PVP. The mean (± S.D) value of CVP was 7.9 ± 2.8 mmHg, whereas the mean PVP value was 8.5 ± 2.8 mmHg. Although the CVP and PVP value correlated with each other (r2=0.83, P<0.0001), as shown in Figure 1, the correlation coefficient was not as high as the values in the previous studies that targeted different groups of patients (r2 ≥ 0.9) [12-14].

Figure 1. Linear regression plot of central venous pressure (CVP) vs. peripheral venous pressure (PVP) with calculation of Pearson’s correlation coefficient. The solid line represents the linear regression. Pearson’s correlation coefficient was 0.91. The number of measurements was 500. All points are not visible because of overlap. The symbol colors indicate the number of data included in each point. Light blue, green, yellow, red, and gray indicate that 1-5, 6-10, 11-15, 16-20, and ≥ 21 data points, respectively, are included at each point.
For testing our hypothesis that cardiac preload can be assessed using CVP and PVP values, we plotted the values of PVP-CVP difference against SVV values and observed that PVP-CVP difference values tended to decrease as SVV values increased (Figure 2A, r2=0.41, P<0.0001). Besides SVV, the values of PVP-CVP difference were plotted against other hemodynamic variables, namely cardiac index (CI, Figure 2B) and stroke volume index (SVI, Figure 2C). Neither CI nor SVI correlated with the values of PVP-CVP difference (r2=0.03 and 0.04, respectively).
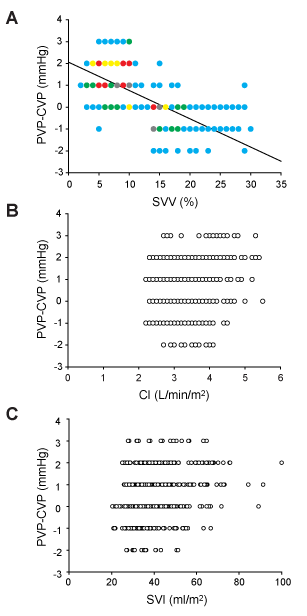
Figure 2. Relationship between the PVP-CVP difference value and (A) stroke volume variation (SVV), (B) cardiac index (CI), and (C) stroke volume index. The number of measurements for each parameter was 500. All points are not seen because of overlap in A. The symbol colors indicate the number of data points included in each point. Light blue, green, yellow, red, and gray indicate that 1-5, 6-10, 11-15, 16-20, and ≥ 21 data points are included at each point. The data points in B and C are not shown in color, because the number of points completely overlapping in these figures was much smaller. The solid line in A represents the linear regression. Pearson’s correlation coefficient was 0.64.
Next, we compared the values of PVP-CVP difference in normal- and high-SVV states. The threshold to divide SVV values into normal and high states was set at 10% [6,15-17]. The value of SVV was always ≤ 10% in the 10 measurements each made in 20 patients, while the corresponding value was always >10% in 12 patients. In the remaining 18 patients, the value of SVV was either ≤ 10% or >10% depending on the timing of measurements. As a result, the value of SVV was ≤ 10% and >10% at 304 and 196 time points, respectively. The CVP value in the normal-SVV state was smaller than that in the high-SVV state (7.6 ± 2.8 vs. 8.5 ± 2.8 mmHg, Mann-Whitney U test, P<0.001), whereas the PVP value in the normal-SVV state was greater than that in the high-SVV state (8.9 ± 2.8 vs. 7.8 ± 2.8 mmHg, Mann-Whitney U test, P<0.001). Consequently, the magnitude of the relationship between CVP and PVP values was reversed between normal- and high-SVV states. This discordance between CVP and PVP was reflected by the following results: PVP-CVP difference values were greater with normal-SVV state (SVV ≤ 10%) than in high-SVV state (SVV >10%, Figure 3A, Mann-Whitney U test, P<0.001). From another point of view, PVP-CVP difference tended to show positive values in the normal-SVV state, whereas PVP-CVP difference tended to show negative values in high-SVV state (Figure 3A).
Finally, we performed ROC curve analysis to determine the cutoff value of PVP-CVP difference to distinguish the normal- and high-SVV states. When the cutoff value was set at 0.5 mmHg, using the PVP-CVP difference value we were able to predict whether SVV was normal or high with high accuracy in any respiratory mode (Figure 3B, Table 2).
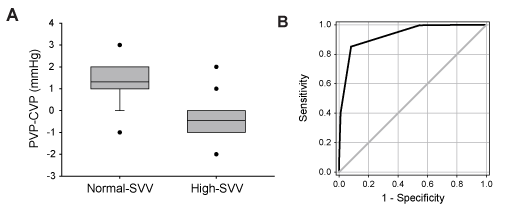
Figure 3. A) Comparison of PVP-CVP difference values in normal-SVV (≤ 10%) and high- SVV (>10%) groups. There was a significant difference between the groups (Mann-Whitney U test, p<0.001). The ends of the boxes define the 25th and 75th percentiles, with a line at the mean and error bars defining the 10th percentile. The error bar indicating the 10th percentile is not shown in the right box, because the values of 10th and 25th percentiles were identical. Error bars indicating the 90th percentile are not shown in both boxes, because the values of 75th and 90th percentiles were identical in both groups. The dots indicate outliers. B) Receiver operating characteristic curve analysis of PVP-CVP difference for predicting cardiac preload. All cases were included in the analysis. The predictive ability of PVP-CVP difference values (black line) was much better than random (grey line). The area under the curve was 0.93.
Table 2. Results of ROC curve analysis. CI: confidence interval; AUC: area under the curve; PCV: pressure control ventilation; CPAP/PSV continuous positive airway pressure/pressure support ventilation.
Respiratory mode |
Cutoff |
Sensitivity |
95% CI |
Specificity |
95% CI |
AUC |
95% CI |
All cases |
0.5 |
0.85 |
[0.80 - 0.89] |
0.92 |
[0.87 - 0.95] |
0.93 |
[0.91 - 0.95] |
PCV |
0.5 |
0.84 |
[0.75 - 0.90] |
0.97 |
[0.90 - 1.00] |
0.94 |
[0.90 - 0.97] |
CPAP/PSV |
0.5 |
0.95 |
[0.83 - 0.99] |
0.92 |
[0.79 - 0.98] |
0.96 |
[0.92 - 1.00] |
Non-intubated |
0.5 |
0.84 |
[0.77 - 0.89] |
0.87 |
[0.78 - 0.93] |
0.92 |
[0.88 - 0.95] |
The hemodynamic and respiratory variables, usage of sedative medicines, and severity score in 3 groups of patients stratified according to respiratory mode are shown in Table 3. There were no differences between patient groups for most items, including SVV, except for CVP, PVP, FiO2, and RASS score.
Table 3: Hemodynamic and respiratory variables, usage of sedative medicines, and severity score of the patients. Numbers in parentheses in the first row and rows for sedative data indicate the number of patients in each group. Data are represented as mean ± standard deviation. One-way ANOVA was used for comparison among groups unless otherwise mentioned. §: unpaired t-test; †: one-way ANOVA on ranks; ‡: Mann-Whitney U test; PCV: pressure control ventilation; CPAP/PSV: continuous positive airway pressure/pressure support ventilation; BP: blood pressure; CI: cardiac index; SVI: stroke volume index; SVV: stroke volume variation; PEEP: positive end expiratory pressure; EtCO2: end-tidal CO2; SpO2: arterial oxygen saturation by pulse oximetry; FiO2: fraction of inspiratory oxygen; RASS; Richmond agitation-sedation scale; APACHE II: Acute Physiological and Chronic Health Enquiry II; SOFA: Sequential Organ Failure Assessment; ND: not detected, N/A: not applicable.
|
|
All (50) |
PCV (17) |
CPAP/PSV (8) |
Non-intubated (25) |
P value |
Hemodynamics
parameters |
Systolic BP (mmHg) |
117 ± 16 |
111 ± 10 |
124 ± 17 |
120 ± 18 |
0.08 |
Diastolic BP (mmHg) |
57 ± 9 |
53 ± 10 |
58 ± 7 |
59 ± 9 |
0.07 |
Heart rate (bpm) |
84 ± 15 |
82 ± 15 |
91 ± 21 |
83 ± 13 |
0.33 |
CI (L/min/m²) |
3.3 ± 0.6 |
3.2 ± 0.7 |
3 ± 0.5 |
3.4 ± 0.6 |
0.06 |
SVI (ml/m²) |
41 ± 10 |
40 ± 10 |
35 ± 12 |
44 ± 10 |
0.08† |
SVV (%) |
11.1 ± 5.1 |
10.8 ± 4.8 |
13.6 ± 7.2 |
10.5 ± 4.5 |
0.48† |
PVP-CVP (mmHg) |
0.6 ± 0.8 |
0.4 ± 0.6 |
0.4 ± 0.7 |
0.8 ± 0.9 |
0.14† |
CVP (mmHg) |
7.8 ± 2.6 |
9.2 ± 2.5 |
9.1 ± 1.9 |
6.5 ± 2.2 |
<0.01† |
PVP (mmHg) |
8.5 ± 2.6 |
9.7 ± 2.6 |
9.5 ± 2.2 |
7.3 ± 2.2 |
<0.01† |
Respiratory
status |
Respiratory rate (bpm) |
10 ± 2.3 |
10 ± 1.2 |
10 ± 1.2 |
9 ± 1.5 |
0.14† |
Tidal volume (ml/kg) |
8.9 ± 0.5 |
8.9 ± 0.5 |
8.9 ± 0.3 |
ND |
0.93§ |
PEEP |
6 ± 1.9 |
6.1 ± 1.9 |
6 ± 1.8 |
N/A |
0.90‡ |
EtCO2 |
32.8 ± 2.5 |
32.7 ± 2.4 |
33.2 ± 2.6 |
ND |
0.63§ |
SpO2 |
98.6 ± 1.1 |
98.5 ± 1.0 |
98.1 ± 1.3 |
98.8 ± 1.1 |
0.31† |
FiO2 |
0.39 ± 0.11 |
0.47 ± 0.12 |
0.44 ± 0.08 |
0.33 ± 0.06 |
<0.0001† |
Sedatives |
RASS |
-2.5 ± 1.2 |
-3.5 ± 0.8 |
-3.3 ± 0.7 |
-1.6 ± 0.7 |
<0.0001† |
Propofol (mg/kg/hr) |
1.73 ± 0.72 (13) |
1.79 ± 0.65 (10) |
1.34 ± 0.97 (3) |
N/A |
0.35§ |
Precedex (μg/kg/hr) |
0.51 ± 0.2 (14) |
0.51 ± 0.22 (5) |
0.49 ± 0.12 (5) |
0.53 ± 0.24 (4) |
0.96 |
Fentanyl (μg/kg/hr) |
0.9 ± 2.62 (34) |
0.97 ± 0.34 (13) |
0.81 ± 0.36 (4) |
0.86 ± 0.39 (17) |
0.61 |
Severity scores |
APACHE II |
15.2 ± 7.5 |
14.9 ± 5.9 |
12.6 ± 2.6 |
16.2 ± 9.3 |
0.57† |
SOFA |
7.5 ± 3.8 |
7.8 ± 3.6 |
7.1 ± 1.1 |
7.5 ± 4.5 |
0.87† |
In the current study, we found that CVP and PVP values weakly correlated with each other in critically ill patients. Moreover, the value of PVP-CVP difference correlated with SVV, but not with CI or SVI. As a result, it was possible to accurately predict whether the patient was likely to have normal or high SVV by calculating PVP-CVP difference values.
Although PVP is not a commonly measured parameter, it has the advantage that it requires only a peripheral route for its measurement. Numerous studies performed in patients under general anesthesia showed that PVP can be used as a substitute for CVP [9-12]. This was the first study to investigate the relationship between CVP and PVP in critically ill patients. The relatively weak correlation between CVP and PVP in this study may have resulted from the wide variability in patient background factors and hemodynamic parameters. Several studies have demonstrated that the difference between CVP and PVP varies with the value of CVP [10,12,18]. Likewise, we found that the magnitude of the relationship between CVP and PVP was reversed between normal- and high-SVV states. Although the mechanisms that underlie this phenomenon are unknown, a plausible cause is changes in peripheral venous tone by cardiac preload. Further study will be necessary to clarify this mechanism.
This study examined only one regression model, i.e., a=1 and b=-1 in the framework of multivariate linear regression model: SVV=a*PVP + b*CVP +e, because we intended to emphasize convenience in clinical use. However, accuracy of prediction may be slightly improved in future through optimization of the multivariate linear regression models.
Although dynamic arterial waveform-based variables, including SVV, seem to be good predictors of volume responsiveness, robust evidence suggested that its effectiveness is limited under spontaneous breathing even in intubated patients [2,3]. In particular, the predictive value of SVV was reportedly poor in patients ventilated with a tidal volume of less than 8 ml/kg [19-21]. This indicates that SVV measurements could have several such limitations, as with other parameters. Hence, several alternative strategies have been developed, such as assessment of the hemodynamic effect of passive leg raising, a tidal volume challenge, lung recruitment maneuvers, and end-expiratory occlusion [3,22]. Nonetheless, since measurement of SVV is noninvasive and convenient, it is still accepted in many facilities.
There were no differences in SVV values among patient groups with different breathing conditions in the current study (Table 3). Previous reports showed that SVV was useful for predicting fluid responsiveness even in septic patients under spontaneous respiration [23,24]. Other studies that evaluated spontaneously breathing patients suggested that slow patterned breathing might improve the discriminative value of SVV [25,26]. Indeed, the average respiratory rate was 10 breaths/min and tidal volume was 8.9 ml/kg in this study (Table 3). Perhaps because the patients breathed relatively slowly and deeply under sedation, the accuracy of SVV could be guaranteed even under spontaneous breathing. For these reasons, although we did not measure fluid responsiveness, it seems reasonable to assume that SVV, which was used as the target variable in this study, would have served as a useful indicator of fluid responsiveness in our setting. As shown in Table 3, both CVP and PVP values were smaller in the non-intubated patient group. This was likely to have resulted from the lack of PEEP in these patients. Light sedation in non-intubated patients was reflected by the high RASS scores in them. The low FiO2 in non-intubated patients can be explained by good oxygenation in these patients. These differences seemed not to affect the relationship between CVP and PVP among the patient groups in this study.
Two different sizes (20 and 22 G) of peripheral venous catheters were utilized in the present study, which could possibility have affected the PVP values. However, this was not the case (8.5 ± 2.2 mmHg vs. 8.5 ± 2.7 mmHg). These results are consistent with previous reports showing that size of peripheral catheter does not affect the measurement of PVP [27,28].
This study has certain limitations. We did not directly test whether the value of PVP-CVP is actually applicable as an indicator of fluid responsiveness. Comparison of PVP-CVP difference values before and after fluid therapy in the same patient would be a good way to address this issue in future.
In conclusion, we found that PVP-CVP difference values correlate with SVV values in critically ill patients regardless of respiratory mode. These results suggest that PVP -CVP difference might serve as a novel indicator to guide fluid therapy.
All procedures performed in this study involving human participants were in accordance with the ethical standards of Gunma University Hospital and with the 1964 Helsinki declaration and its later amendments. Ethical approval was also obtained from the hospital.
Written informed consent was obtained from all patients enrolled in this study.
This work was supported by JSPS KAKENHI Grant Number 15K10533. We wish to thank the staff members of the Intensive Care Unit, Gunma University Hospital, for helping with data acquisition.
- Toyoda D, Fukuda M, Iwasaki R, Terada T, Sato N, et al. (2015) The comparison between stroke volume variation and filling pressure as an estimate of right ventricular preload in patients undergoing renal transplantation. J Anesth 29: 40-46. [Crossref]
- Chew MS, Åneman A (2013) Haemodynamic monitoring using arterial waveform analysis. Curr Opin Crit Care 19: 234-241. [Crossref]
- Monnet X, Marik PE, Teboul JL (2016) Prediction of fluid responsiveness: an update. Ann Intensive Care 6: 111. [Crossref]
- Biais M, Bernard O, Ha JC, Degryse C, Sztark F (2010) Abilities of pulse pressure variations and stroke volume variations to predict fluid responsiveness in prone position during scoliosis surgery. Br J Anaesth 104: 407-413. [Crossref]
- Michard F, Teboul JL (2000) Using heart-lung interactions to assess fluid responsiveness during mechanical ventilation. Crit Care 4: 282-289. [Crossref]
- Cannesson M, Musard H, Desebbe O, Boucau C, Simon R, et al. (2009) The ability of stroke volume variations obtained with Vigileo/FloTrac system to monitor fluid responsiveness in mechanically ventilated patients. Anesth Analg 108: 513-517. [Crossref]
- Desjardins R, Denault AY, Belisle S, Carrier M, Babin D, et al. (2004) Can peripheral venous pressure be interchangeable with central venous pressure in patients undergoing cardiac surgery? Intensive Care Med 30: 627-632.
- Munis JR, Bhatia S, Lozada LJ (2001) Peripheral venous pressure as a hemodynamic variable in neurosurgical patients. Anesth Analg 92: 172-179. [Crossref]
- Anter AM, Bondok RS (2004) Peripheral venous pressure is an alternative to central venous pressure in paediatric surgery patients. Acta Anaesthesiol Scand 48: 1101-1104. [Crossref]
- Hoftman N, Braunfeld M, Hoftman G, Mahajan A (2006) Peripheral venous pressure as a predictor of central venous pressure during orthotopic liver transplantation. J Clin Anesth 18: 251-255. [Crossref]
- Kumar D, Ahmed SM, Ali S, Ray U, Varshney A, et al. (2015) Correlation between central venous pressure and peripheral venous pressure with passive leg raise in patients on mechanical ventilation. Indian J Crit Care Med 19: 648-654. [Crossref]
- Kim SH, Park SY, Cui J, Lee JH, Cho SH, et al. (2011) Peripheral venous pressure as an alternative to central venous pressure in patients undergoing laparoscopic colorectal surgery. Br J Anaesth 106: 305-311. [Crossref]
- Hadimioglu N, Ertug Z, Yegin A, Sanli S, Gurkan A, et al. (2006) Correlation of peripheral venous pressure and central venous pressure in kidney recipients. Transplant Proc 38: 440-442. [Crossref]
- Memtsoudis SG, Jules-Elysse K, Girardi FP, Buschiazzo V, Maalouf D, Sama AA, Urban MK (2008) Correlation between centrally versus peripherally transduced venous pressure in prone patients undergoing posterior spine surgery. Spine (Phila Pa 1976) 33: E643-E647. [Crossref]
- Monnet X, Rienzo M, Osman D, Anguel N, Richard C, Pinsky MR, Teboul JL (2005) Esophageal Doppler monitoring predicts fluid responsiveness in critically ill ventilated patients. Intensive Care Med 31: 1195-1201. [Crossref]
- Reuter DA, Felbinger TW, Schmidt C, Kilger E, Goedje O, et al. (2002) Stroke volume variations for assessment of cardiac responsiveness to volume loading in mechanically ventilated patients after cardiac surgery. Intensive Care Med 28: 392-398. [Crossref]
- Berkenstadt H, Margalit N, Hadani M, Friedman Z, Segal E, et al. (2001) Stroke volume variation as a predictor of fluid responsiveness in patients undergoing brain surgery. Anesth Analg 92: 984-989. [Crossref]
- Cave G, Harvey M (2008) The difference between peripheral venous pressure and central venous pressure (CVP) decreases with increasing CVP. Eur J Anaesthesiol 25: 1037-1040. [Crossref]
- Reuter DA, Bayerlein J, Goepfert MS, Weis FC, Kilger E, et al. (2003) Influence of tidal volume on left ventricular stroke volume variation measured by pulse contour analysis in mechanically ventilated patients. Intensive Care Med 29: 476-480. [Crossref]
- De Backer D, Heenen S, Piagnerelli M, Koch M, Vincent JL (2005) Pulse pressure variations to predict fluid responsiveness: influence of tidal volume. Intensive Care Med 31: 517-523. [Crossref]
- Lansdorp B, Lemson J, van Putten MJ, de Keijzer A, van der Hoeven JG, et al. (2012) Dynamic indices do not predict volume responsiveness in routine clinical practice. Br J Anaesth 108: 395-401. [Crossref]
- Michard F (2017) Toward Precision Hemodynamic Management. Crit Care Med 45: 1421-1423. [Crossref]
- Lanspa MJ, Grissom CK, Hirshberg EL, Jones JP, Brown SM (2013) Applying dynamic parameters to predict hemodynamic response to volume expansion in spontaneously breathing patients with septic shock. Shock 39: 155-160. [Crossref]
- Yoshihara F, Kishida M, Ogawa K, Nishigaki T, Nakasaki H, et al. (2017) High Stroke Volume Variation Is an Independent Predictor for Decreased Blood Pressure During Hemodialysis. Ther Apher Dial 21: 166-172. [Crossref]
- Zöllei E, Bertalan V, Németh A, Csábi P, László I, et al. (2013) Non-invasive detection of hypovolemia or fluid responsiveness in spontaneously breathing subjects. BMC Anesthesiol 13: 40. [Crossref]
- Juri T, Suehiro K, Tsujimoto S, Kuwata S, Mukai A, et al. (2017) Pre-anesthetic stroke volume variation can predict cardiac output decrease and hypotension during induction of general anesthesia. J Clin Monit Comput. [Crossref]
- Sahin A, Salman MA, Salman AE, Aypar U (2005) Effect of catheter site on the agreement of peripheral and central venous pressure measurements in neurosurgical patients. J Clin Anesth 17: 348-352. [Crossref]
- Tugrul M, Camci E, Pembeci K, Al-Darsani A, Telci L (2004) Relationship between peripheral and central venous pressures in different patient positions, catheter sizes, and insertion sites. J Cardiothorac Vasc Anesth 18: 446-450. [Crossref]



