The use of magnetic resonance imaging (MRI) to visualize lesions in multiple sclerosis in the spinal cord dates back to the 1980s. In general, medullary sclerosis is associated with brain lesions but as many as 20% of patients with spinal lesions do not have intracranial lesions. In the spinal cord, we observe lesions of both the white and gray matter. MRI shows abnormalities in 95% of patients with multiple sclerosis [1-3]. The MRI imaging protocol for the spinal cord must include T1-weighted images, STIR (Short-TI Inversion Recovery), T2 with contrast medium [4]. MRI is extremely important for diagnosis as indicated by the McDonald criteria, introduced in 2001 and periodically reviewed (last revision proposal in 2017-Table 1). MRI is used to demonstrate lesion dissemination over time (DIT) and space (DIS) [5,6].
Table 1. 2017 Proposed Revisions to the McDonald Criteria: Attack Onset.
Number of lesions with
objective clinical evidence |
Additional data needed for a diagnosis of multiple sclerosis |
≥ 2 clinical attacks† |
≥ 2 (DIS) |
None* |
≥ 2 clinical attacks† |
1 |
None* or clear-cut historical evidence of a previous attack involving a lesion
in a distinct anatomical location (DIS) |
≥ 2 clinical attacks† |
1 |
Dissemination in space (DIS) demonstrated by an additional clinical attack
implicating a different CNS site or by MRI |
1 clinical attack† |
≥ 2 (DIS) |
Dissemination in time (DIT) demonstrated by an additional clinical attack or by
MRI or demonstration of CSF-specific oligoclonal bands ** |
1 clinical attack† |
1 |
Dissemination in space (DIS) demonstrated by an additional clinical attack
implicating a different CNS site or by MRI and Dissemination in time (DIT)
demonstrated by an additional clinical attack or by MRI OR demonstration of
CSF-specific oligoclonal bands** |
†Attack, relapse, exacerbation, and (when it is the first episode) clinically isolated syndrome are synonyms. See clinically isolated syndrome and relapse for descriptions
*No additional tests are required to demonstrate dissemination in space and time. In addition, spinal cord MRI or CSF examination should be considered in patients with insufficient clinical and MRI evidence supporting multiple sclerosis, with a presentation other than a typical clinically isolated syndrome, or with atypical features
** The presence of CSF-specific oligoclonal bands does not demonstrate dissemination in time per se but can substitute for the requirement for demonstration of this measure. |
2017 MCDonald criteria for demonstration of dissemination in space and time by MRI in a patient with a
clinically isolated syndrome |
DIS: Dissemination in space can be demonstrated by one or more T2-hyperintense lesions* that are characteristic of multiple sclerosis in two or more of four areas of the CNS: periventricular, cortical or juxtacortical, and infratentorial
brain regions, and the spinal cord. |
DIT: Dissemination in time can be demonstrated by the simultaneous presence of gadolinium-enhancing and
non-enhancing lesions at any time or by a new T2-hyperintense or gadolinium-enhancing lesion on follow-up MRI, with reference to a baseline scan, irrespective of the timing of the baseline MRI. |
The magnetic resonance criteria currently recognized for DIS are the following:
- At least one T2 lesion in at least 2 of the 4 positions considered characteristic for MS (juxtacortical, periventricular, infratentorial and spinal cord)
- If brain imaging fails to demonstrate DIS, imaging of the spinal cord may be useful in demonstrating a minimum weighted lesion in T2 of 3 mm or larger, less than 2 vertebral bodies in length and involving less than cordial cross-section.
The magnetic resonance criteria for DIT are as follows:
- A new T2-weighted and/or gadolinium-enhancing lesion on follow-up MRI, with reference to baseline magnetic resonance regardless of initial imaging time
- Simultaneous presence of asymptomatic gadolinium lesions and non-modifying lesions at any time
Radiologically isolated syndrome (RIS)
The RIS describes cases in which McDonald's imaging criteria respond to DIT and DIS but lack corresponding clinical symptoms. The exact meaning is still the subject of debate. Some authors have suggested that this may represent an earlier stage of isolated clinical syndrome (CIS). In an extensive review of the RIS literature, Granberg et al. [7] emphasize that about two thirds of cases show a radiological progression and a third one develops clinical symptoms at 5-year follow-up. They also point out that clinical conversion is more likely when cervical cord lesions are present. However, further studies are needed to determine patient stratification and to define the role of RIS-modifying drugs [7].
Computerized tomography (CT)
Since the CT scan has poor sensitivity, detection, assessment and characterization of lesions and patterns of MS improvement are limited in this manner. Consequently, with the progress of MRI, the evaluation of the spinal cord by axial CT scan has been abandoned. In CT scans, large and similar lesions can occasionally mimic a neoplasm, and characterizing them can be difficult. Primary and secondary spinal cord neoplasms (astrocytomas, ependymomas), infection, transverse myelitis, acute infarction, sarcoidosis and systemic lupus erythematosus can simulate demyelinating MS plaques on CT scans.
Magnetic Resonance
Magnetic resonance far exceeds the CT scan in the ability to detect intramedullary disease; MRI is currently used for the first-line survey of spinal MS. The plaques, depending on the age, are visualized as areas of lower signal intensity in T1, can take various forms and generally do not occupy more than two vertebral segments in length [8,9] (Figure 1).
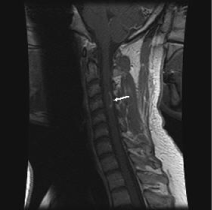
Figure 1. Sagittal image, weighed in T1, following the contrast of the gadolinium showing a arciform strengthening along the edge of the plate, typical of demyelination.
Most plaques appear hyperintense in T2-weighted images. The spinal cord may also appear locally enlarged in case of active disease while chronic lesions often show spinal cord atrophy [10]. The spinal cord may have a focal swelling which is usually an active disease. Larger active lesions may have extensive edema with associated cord expansion. In contrast, chronic lesions often demonstrate focal cordial atrophy. Spinal injuries usually coexist with more severe concomitant brain plaques. As many as 20% of MS spinal injuries are isolated. Spinal cord shrinkage due to atrophy is present in 10% of patients with medullary involvement [11] (Figure 2 and Figure 3).
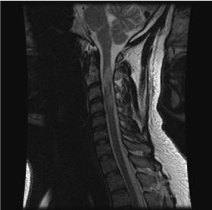
Figure 2. Sagittal image, T2-weighted image showing areas of hyperintensity of the signal in the cervical spinal cord and in the bridge.
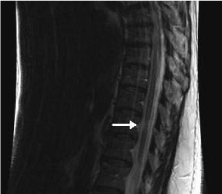
Figure 3. Sagittal image, T2-weighted image showing a focal area of atrophy of the spinal cord in a patient with longstanding multiple sclerosis.
Spinal cord swellings found in multiple sclerosis can simulate a neoplasm. FLAIR (Fluid Attenuation Inversion Recovery)sequences have a lower sensitivity than spin-echo sequences due to the relevance of spinal cord demyelinating lesions [12-14].
Contrast-enhancing multiple sclerosis lesions with MRI are important markers of active inflammation in the diagnostic work-up and in disease monitoring. Contrast-enhancing MS lesions have lower proton density and shorter relaxation times compared with nonenhancing lesions [15,16]. Gadolinium-based contrast agents may induce the presence of nephrogenic systemic fibrosis (NSF), also known as fibrosing nephrogenic dermopathy (NFD). The disease occurred in patients with moderate to end-stage renal failure after receiving a gadolinium-based contrast agent to improve MRI or MR angiographic (MRA) scans. NSF/NFD is a debilitating and sometimes fatal disease. Features include red or dark spots on the skin; burning, itching, swelling, hardening and tension of the skin; yellow spots on the white of the eyes; joint stiffness with difficulty in moving or straightening arms, hands, legs or feet; deep pain in the bones or in the ribs of the hip; and muscle weakness.
Differential diagnosis
2021 Copyright OAT. All rights reserv
The differential diagnosis is mainly posed with
- Primary or metastatic neoplasms of the spinal cord: in this case the presence of cysts or hemorrhages orientates towards these pathologies
- Acute disseminated encephalomyelitis (ADEM): in this pathology, with a monophasic course, the presence of brain lesions is concomitant, there is an improvement of the neuroradiological picture in the following periods and in the anamnesis there is usually an infectious process in the previous weeks
- Sarcoidosis: there is generally an involvement of the pious mother
- Transverse myelitis
- NMOSD (neuromyelitisoptica spectrum disorder): AQP4-IgG, MOG IgG; MRI: Longitudinally extensive optic nerve lesion with or without extension into optic chiasm, longitudinally extensive spinal cord lesions extending over three or more vertebral segments, brain lesions in diencephalon, dorsal midbrain, periependymal regions
- Infarction: generally present at the thoracic level with initial involvement of the spinal area of the anterior spinal artery (anterior gray matter)
- Vasculitis: evaluate the clinical features and the history. Multiple lesions are often present
- Radiation myelitis: typically doses above 4,000 cGy with a 1 to 3-year latency period are required
- Arterio-venous fistula: generally at thoraco-lumbar level over the age of 50 with a history of rachialgia. There is an interest in a very long segment
- Galler S, Stellmann JP, Young KL, Kutzner D, Heesen C, et al. (2016) Improved Lesion Detection by Using Axial T2-Weighted MRI with Full Spinal Cord Coverage in Multiple Sclerosis. AJNR Am J Neuroradiol 37: 963-969. [Crossref]
- Cohan S, Chen C, Baraban E, Stuchiner T, Grote L (2016) MRI utility in the detection of disease activity in clinically stable patients with multiple sclerosis: a retrospective analysis of a community based cohort. BMC Neurol 16: 184. [Crossref]
- Lukas C, Knol DL, Sombekke MH, Bellenberg B, Hahn HK, et al. (2015) Cervical spinal cord volume loss is related to clinical disability progression in multiple sclerosis. J Neurol Neurosurg Psychiatry 86: 410-418. [Crossref]
- Thompson AJ, Banwell BL, Barkhof F, Carroll WM, Coetzee T, et al. (2018) Diagnosis of multiple sclerosis: 2017 revisions of the McDonald criteria. Lancet Neurol 17: 162-173. [Crossref]
- Traboulsee A, Simon JH, Stone L, Fisher E, Jones DE, et al. (2015) Revised Recommendations of the Consortium of MS Centers Task Force for a Standardized MRI Protocol and Clinical Guidelines for the Diagnosis and Follow-Up of Multiple Sclerosis. AJNR Am J Neuroradiol 37: 394-401. [Crossref]
- http://c.ymcdn.com/sites/www.mscare.org/resource/collection/9C5F19B9-3489-48B0-A54B-623A1ECEE07B/mriprotocol2009.pdf
- Kearney H, Miller DH, Ciccarelli O (2015) Spinal cord MRI in multiple sclerosis--diagnostic, prognostic and clinical value. Nat Rev Neurol 11: 327-338. [Crossref]
- Granberg T, Martola J, Kristoffersen-Wiberg M, Aspelin P, Fredrikson S (2013) Radiologically isolated syndrome--incidental magnetic resonance imaging findings suggestive of multiple sclerosis, a systematic review. Mult Scler 19: 271-280. [Crossref]
- Agosta F, Absinta M, Sormani MP, Ghezzi A, Bertolotto A, et al. (2007) In vivo assessment of cervical cord damage in MS patients: a longitudinal diffusion tensor MRI study. Brain 130: 2211-2219. [Crossref]
- Filippi M (2000) Enhanced magnetic resonance imaging in multiple sclerosis. Mult Scler 6: 320-326. [Crossref]
- Filippi M, Bozzali M, Horsfield MA, Rocca MA, Sormani MP, et al. (2000) A conventional and magnetization transfer MRI study of the cervical cord in patients with MS. Neurology 54: 207-213. [Crossref]
- Stevenson VL, Gawne-Cain ML, Barker GJ, Thompson AJ, Miller DH (1997) Imaging of the spinal cord and brain in multiple sclerosis: a comparative study between fast FLAIR and fast spin echo. J Neurol 244: 119-124. [Crossref]
- Rocca MA, Horsfield MA, Sala S, Copetti M, Valsasina P, et al. (2011) A multicenter assessment of cervical cord atrophy among MS clinical phenotypes. Neurology 76: 2096-2102. [Crossref]
- Stevenson VL, Gawne-Cain ML, Barker GJ, Thompson AJ, Miller DH (1997) Imaging of the spinal cord and brain in multiple sclerosis: a comparative study between fast FLAIR and fast spin echo. J Neurol 244: 119-124. [Crossref]
- Poonawalla AH, Hou P, Nelson FA, Wolinsky JS, Narayana PA (2008) Cervical spinal cord lesions in multiple sclerosis: T1-weighted inversion-recovery MR imaging with phase-sensitive reconstruction. Radiology 246: 258-264. [Crossref]
- Blystad I, Håkansson A, Tisell J, Ernerudh Ö, Smedby P, et al. (2016) Quantitative MRI for Analysis of Active Multiple Sclerosis Lesions without Gadolinium-Based Contrast Agent. AJNR Am J Neuroradiol 37: 94-100. [Crossref]
Editorial Information
Editor-in-Chief
Kazuhisa Nishizawa
Teikyo University
Article Type
Commentary
Publication history
Received date: February 15, 2018
Accepted date: February 22, 2018
Published date: February 26, 2018
Copyright
© 2018 Lerza MC, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation
Lerza MC, Gargiulo MG, Busillo A, Somma MR, Montella S, et al. (2018) MRI in the multiple sclerosisspinal cord lesions. Biomed Res Clin Prac 3: DOI: 10.15761/BRCP.1000156
Corresponding author
Anna Capasso
Department of Pharmacy, University of Salerno, Salerno, Italy
E-mail : bhuvaneswari.bibleraaj@uhsm.nhs.uk

Figure 1. Sagittal image, weighed in T1, following the contrast of the gadolinium showing a arciform strengthening along the edge of the plate, typical of demyelination.

Figure 2. Sagittal image, T2-weighted image showing areas of hyperintensity of the signal in the cervical spinal cord and in the bridge.

Figure 3. Sagittal image, T2-weighted image showing a focal area of atrophy of the spinal cord in a patient with longstanding multiple sclerosis.
Table 1. 2017 Proposed Revisions to the McDonald Criteria: Attack Onset.
Number of lesions with
objective clinical evidence |
Additional data needed for a diagnosis of multiple sclerosis |
≥ 2 clinical attacks† |
≥ 2 (DIS) |
None* |
≥ 2 clinical attacks† |
1 |
None* or clear-cut historical evidence of a previous attack involving a lesion
in a distinct anatomical location (DIS) |
≥ 2 clinical attacks† |
1 |
Dissemination in space (DIS) demonstrated by an additional clinical attack
implicating a different CNS site or by MRI |
1 clinical attack† |
≥ 2 (DIS) |
Dissemination in time (DIT) demonstrated by an additional clinical attack or by
MRI or demonstration of CSF-specific oligoclonal bands ** |
1 clinical attack† |
1 |
Dissemination in space (DIS) demonstrated by an additional clinical attack
implicating a different CNS site or by MRI and Dissemination in time (DIT)
demonstrated by an additional clinical attack or by MRI OR demonstration of
CSF-specific oligoclonal bands** |
†Attack, relapse, exacerbation, and (when it is the first episode) clinically isolated syndrome are synonyms. See clinically isolated syndrome and relapse for descriptions
*No additional tests are required to demonstrate dissemination in space and time. In addition, spinal cord MRI or CSF examination should be considered in patients with insufficient clinical and MRI evidence supporting multiple sclerosis, with a presentation other than a typical clinically isolated syndrome, or with atypical features
** The presence of CSF-specific oligoclonal bands does not demonstrate dissemination in time per se but can substitute for the requirement for demonstration of this measure. |
2017 MCDonald criteria for demonstration of dissemination in space and time by MRI in a patient with a
clinically isolated syndrome |
DIS: Dissemination in space can be demonstrated by one or more T2-hyperintense lesions* that are characteristic of multiple sclerosis in two or more of four areas of the CNS: periventricular, cortical or juxtacortical, and infratentorial
brain regions, and the spinal cord. |
DIT: Dissemination in time can be demonstrated by the simultaneous presence of gadolinium-enhancing and
non-enhancing lesions at any time or by a new T2-hyperintense or gadolinium-enhancing lesion on follow-up MRI, with reference to a baseline scan, irrespective of the timing of the baseline MRI. |



