A wetland rice field provides a unique set of condition for biological nitrogen fixation. Firstly, the aquatic plant habitat gives suitable sites for the activity of autotrophic nitrogen-fixing blue-green algae. Secondly, the anaerobic condition in a submerged soil is suitable for heterotrophic nitrogen-fixing bacteria, the nitrogenase that is labile [1,2] investigated the possibility of making large quantities of BGA (blue-green algae) available for use as fertilizer. He inoculated soil with BGA and found gains in yield over untreated controls of 2% in the 1st year, 8% in the 2nd, 15% in the 3rd year, 20% in the 4th year and 11% in the 5th years. Watanabe then considered that fertilization with algae in equivalent to (NH4)2SO4 application of 29 Kg N ha-1. Using 15N, [3] have shown higher plants can take up nitrogen fixed by BGA within a few days. No quantitative information however, is available on the proportion of BGA nitrogen that is readily available for plant uptake and the proportion that becomes available only after an extended period of mineralization.
It is now well established that nitrogen fixation by BGA plays a vital role in the buildup and maintenance of soil fertility, but it is equally important to understand how, when and what ways the fixed-N is made available to the rice plants. Evidences on these aspects are still scanty and mostly hypothetical. For this reason, an attempt has been taken to evaluate and compare percent recovery of nitrogen by rice from supplied two chemical fertilizers and five BGA species.
The soil for the present experiment is highly productive known as Insch soil obtained from Murrials farm in Aberdeenshire of Scotland UK. Chemically, the soil had pH 5.96, organic carbon 7.95%, total nitrogen 0.28 %, cation exchange capacity 11.5 meq 100-1 g and sandy loam in texture. Because of limitation of choice the rice variety ‘MRI’ (variety no. IR22) of Malaysian agricultural development Institute was collected from School of Agriculture, Aberdeen University, UK was selected as indicator plant.
After viability test (98.3% germination was recorded), the 25 days old seedlings were transplanted to 4L capacity plastic pots. The experiment was laid out in a split plot design. The
experimental plots were divided into two blocks i.e., block I and block II. Each block was sub-divided into two sub-blocks i.e., sub-block I and sub-block II.and representing two replications in each sub-block. Each sub-block was again divided into 21 unit plots upon which the treatment was superimposed randomly. The total number of unit plots (pots, 21 X 4) was 84. There were three sources of nitrogen, namely urea (U), ammonium sulfate (As) and blue green algae (BGA) each at five rates. For the BGA each species was considered to be a rate (Table 1).
Table 1. For the five BGA spieces rate
Rate |
Fertilizers
(mg N pot-1) |
BGA |
1 |
30 mg |
Anabaena variabilies |
2 |
60 mg |
Anabaena cylindrica
|
3 |
90 mg |
Anabaena doliolum |
4 |
120 mg |
Nostoc muscorum
|
5 |
150 mg |
Plectonema boryanum |
Two types of control were prepared provided in this design. One control receiving no nitrogen (0) and the second control was inoculated with five species of BGA in each sub block without growing rice plants. Thus, there were 21 treatments and combinations. 84 plastic (21cm Х17cm) round pots were numbered consecutively. Each pot has drainage holes were closed with thick sticky tape. The pots were washed carefully and dried before use. 1800g air-dry soils were placed into each pot with capillary matting (Fyba mat) at the bottom. At the rate of 5 m mole P and K were added to each pot as solution of KH2PO4 . So the air dry soil was mixed with 20 ml of KH2PO4 solution with 150 ml water in a Kenwood mixer for the basic fertilizer dose of P and K The moist soil were transferred to the pots with light and even packing and 1500 ml of water were added to each pot. This forms a 2-cm depth of standing water over the soil surface.
pots were kept at constant temperature covering with polythene sheet. The pots were transformed after 5 days to the glass house and appropriate quantities of N fertilizer added and mixed. In case of BGA pots each inoculate was applied as a liquid suspension collected through batch culture. Four days after transplanting, when the seedlings become fully established, the depth of standing water was raised to 4.5 cm and maintained throughout the growing period. In the present study, the temperature was optimum (25-30°C) up to 56 days and then dropped in month November. In that month and especially on cloudy days’ fluorescent lights were used to supply adequate light. The pots were weeded by hand from time to time whenever necessary. The rice plants started tillering within 2 weeks after transplanting. Six weeks after transplanting, pots numbering 1-21 from block I and 43-63 from block II and twelve weeks after transplanting (harvesting period) from rest of pots 10g moist soil were taken and extracted with 2M KCL solution for NH4-N and NO3-N determination and another 10g samples was removed for determination of water content. The method of Bremner [4] was followed to extract and determination for NH4-N and NO3-N of the pot soils. There was little obvious difference between the two fertilizers, i.e. urea and ammonium sulphate on basic of their relative efficiency factors (on the basis of taking urea as 100) in (Table 2). These values also show the greater efficiency of fertilizer application compared to supply through blue-green algae but in BGA exceptionally A. variabilies that showed higher relative efficiency capacity among all fertilizers. Values for the apparent % recovery of nitrogen by rice plants are presented in (Table 3) and were obtained by subtracting the control offtake value from the value obtained for each rate of application and expressing those as a percentage of the N-applied. Recoveries were variable but low. Recovery of added-N was highest at the first harvest in case of chemical fertilizers that suggests that fertilizer-N was taken up by rice in preference of soil and BGA-N which contributed to plant uptake slowly and with respect to time. Normal recovery of fertilizer nitrogen applied to wetland rice crop seldom more than 30 to 40% and even with the best agronomic practices, rarely exceeds 60-68% [5]. Low efficiency of rice is related to ammonia volatilization, denitrification, leaching, ammonia fixation, fertilizer N in lowland immobilization and runoff [5]. Dry matter yield of rice as obtained by application of (a) five level of U and AS and (b) five BGA species at the 2nd harvest compare to control treatment presented in figure 1.
Table 2. Efficiency factor for different sources of N
N-sources |
Relative Efficiency Factor (urea 100) |
|
|
Dry Matter Yield |
N-offtake |
Control |
64 |
39 |
Urea |
100 |
100 |
Ammonium Sulphate |
103 |
105 |
BGA |
87 |
84 |
A. variabilies
A. cylindrica
A. doliolum
A. doliolum
P. boryanum |
108 |
123 |
85 |
82 |
87 |
92 |
72 |
59 |
81 |
62 |
Table 3. Apparent % recovery of N by rice through two chemical fertilizer and five BGA species.
N source |
First harvest |
Mean |
N Rate (mg) |
|
30 mg |
60 mg |
90 mg |
120 mg |
150 mg |
|
As, r= -0.840 |
60.84 |
28.04 |
16.54 |
9.04 |
15.4 |
25.91 |
U, r= -0.773 |
61.65 |
61.99 |
24.27 |
31.88 |
32.9 |
42.53 |
BGA, r= -0.588 |
A.varibilies |
A.cylindrica |
A.doliolum |
N.muscorum |
P.boryanum |
|
|
29.72 |
18.9 |
38.62 |
4.77 |
11.4 |
20.68 |
N source |
Second harvest |
Mean |
N Rate |
|
30 mg |
60 mg |
90 mg |
120 mg |
150 mg |
|
As, r=0.106*** |
19.5 |
17.68 |
27.16 |
25.08 |
17.31 |
21.35 |
U, r= -0.080 |
30.6 |
38.54 |
59.17 |
28.9 |
32.26 |
37.89 |
BGA. r= -0.833 |
A.variabilies |
A.cylindrica |
A.doliolum |
N.muscorum |
P.boryanum |
|
|
37.81 |
30.64 |
21.75 |
12.06 |
21.16 |
24.68 |
*** significant at 0.01%
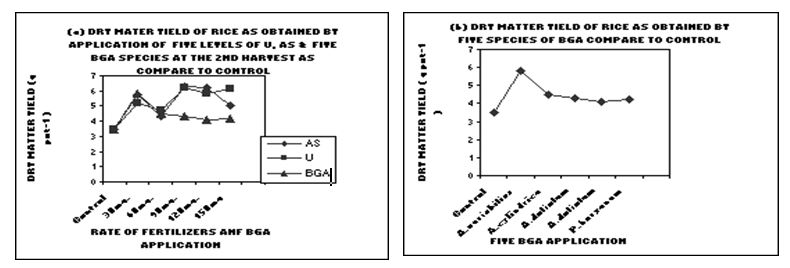
Figure 1. Dry matter yield of rice as obtained by application of (a) five levels of U and AS
and (b) five BGA species at the 2nd harvest compare to control treatment.
From correlation study it was found that only ammonium sulphate showed highly correlated value with different doses of fertilizers at 0.1% level (r = 0.106) at 2nd harvest . On the other hand, urea and all BGA species showed negative relationship [6].
- Roger PA, Santiago-Ardales S, Reddy PM, Watanabe I (1987) The abundance of heterocystous blue-green algae in rice soils and inocula used for application in rice fields. Biology and Fertility of Soils 5: 98-105.
- Watanabe A (1960) Proceedings of the Symposium on Algology. Indian Council of Agricultural Research, New Delhi.
- Miah MH, Stewart WDP (1985) Fate of nitrogen applied as Azolla and blue-green algae (cyanobacteria) in waterlogged rice soils – a 15N traces study. Plant and Soil 83: 363-370.
- Bremner JM (1965) Inorganic forms of nitrogen in “Methods of Soil analysis” (C.A.Black, Ed), Agronomy No 9,Chapter 84 (Part 2) pp.1179-127. Amer.Soc. Agron, Madison, Wisconsin.
- De Datta SK, Fillery LRP, Craswell ET (1983) Results from recent studies on nitrogen fertilizer efficiency in wetland rice. Outlook Agric 12: 125-134.
- Savant NK, De Datta SK (1982) Nitrogen transformation in wetland rice soils. Adv. Agron 35: 241-302.
