Abstract
Diabetic retinopathy (DR) a leading cause of blindness in diabetes mellitus (DM). Impaired oscillatory potentials (OPs) in the electro-retinogram (ERG) and visual dysfunction are commonly seen in DM. Streptozotocin (STZ)-induced diabetic rats showed obvious vacuolation and many swollen mitochondria in the retinal ganglion cells of the retina, and reduced amplitudes of b-waves and oscillatory potentials (OP). Hence, in this study, the effects of lutein and zeaxanthin isomers (L/Zi) on visual function and carbohydrate metabolism in STZ induced diabetic rats evaluated. DM induced in overnight in fasted animals by a single intra-peritoneal injection of STZ (30 mg/kg) in 0.1 M citrate buffer, pH 4.5. Fasting blood glucose levels was measured at 72 h after STZ injection. Animals having blood glucose levels >150 mg/dL are considered as diabetic rats. Twenty nine (29) Male Wistar strain (WNIN) rats (2 months old; mean body weight: 213 ± 14 g) divided into four groups [Group I, Control (standard diet); Group II, DM (STZ induced); Group III: Group II + regular lutein and zeaxanthin isomers [L/Zi, lutein, 0.5%]; and Group IV [Group II + soluble lutein and zeaxanthin isomers [SL/Zi, 0.5%]. All animals housed in each cage maintained on their respective diets for 12 weeks and drinking water provided ad libitum throughout the study period. Daily food intake and weekly body weights, fasting glucose and HbA1c levels assessed. Electro-retinogram performed for all treatments. L/Zi treatment minimized deterioration of b-wave/a-wave ratio and improved oscillatory potential in Group III (442.6) and Group IV (561.9) compared to Group II (334.2). Group IV had shown much difference in sum of OPs compared to Group II and reduced HbA1c levels observed in Group III and VI. Group IV significantly prevented reduction in total retinal thickness better than that of Group III as shown by H & E staining qRT PCR, western blot and immunohistochemistry studies showed lowered expression of VEGF and PDGF (stimulates vasculogenesis and angiogenesis in the retina) in Group IV when compared to group-D. Group IV prevented loss of rhodopsin and nerve growth factor proteins as assessed by qRT PCR and immunofluorescence. Increased expression of stress proteins like glial fibrillary acidic protein (GFAP) and hypoxia-inducible factor 1-alpha (HIF-1A) observed in Group IV more effectively than Group III. These results show that lutein has great potential in preventing diabetes-induced retinal degeneration.
Key words
lutein, zeaxanthin isomers, age-related macular degeneration, macular pigment, electro-retinogram, rhodopsin, NGF, GFAP, HIF-1A, VEGF
Abbreviations
STZ: Streptozotocin; RL/Zi: Regular Lutein/Zeaxanthin isomers; D: Diabetic rats; Ops: Oscillatory potentials; Rho: Rhodopsin; NGF: Nerve growth factor; HIF-1-α: Hypoxia-inducible factor 1-alpha; VEGF: Vascular, Endothelial growth factor; GFAP: Glial fibrillary acidic protein; T1DM: Type 1 diabetes mellitus; T2DM: Type 2 diabetes mellitus ; CVD: Cardiovascular disease; IR: Insulin resistance; Nrf2: Nuclear factor (erythroid-derived 2)-like 2; SL/Zi: Soluble Lutein/ Zeaxanthin isomers; ERG: Electeroretinogram; HbA1c: Glycated haemoglobin; INL: Inner nuclear layer; OPL: Outer plexiform layer; ONL: Outer nuclear layer; ROS: Reactive oxygen species; PDGF: platelet-derived growth factor
Introduction
Based on recent Center for Disease Control [CDC] report prevalence of diabetic retinopathy (DR) was observed in one-third of adults over age 40 years with diabetes mellitus (DM) and greater than one-third of DR prevalence observed in African-Americans and Mexican-Americans [1]. Diabetic retinopathy (DR) is damage to the retina caused by complications of diabetes, which can eventually lead to blindness (Figure 1). About one in three people with diabetes have diabetic retinopathy and estimated costs is $ 500 million annually. It is estimated from 2010 to 2050, the number of Americans with diabetic retinopathy is expected to nearly double, from 7.7 million to 14.6 million. Hispanic Americans are expected to see the greatest increase in cases, rising more than three-fold from 1.2 million to 5.3 million. In U.S. the rate of DR was increased by 89%. Current estimation of diabetes is 366 million people worldwide and over 126.6 million people have DR in 2010 and which may potentially increase to 191.0 million people by 2030, and most vision-threatening diabetic retinopathy (VTDR) may increase from 37.3 million to 56.3 million in U.S. [2,3].
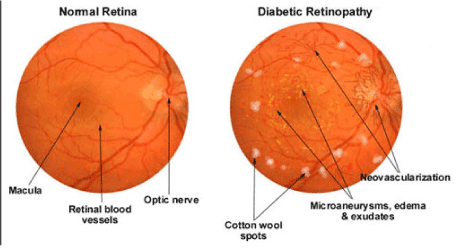
Figure 1. Normal vs Diabetic Retinopathy.
The STZ rat model of diabetes used for decades and is one of the most commonly used and has been reproducible models has been of diabetes mellitus (DM, Figure 2). It is almost like human DM in reference to its structural, functional and complications. It is an easy model to study the mechanism of action of DM for potential therapeutic and nutritional therapies.
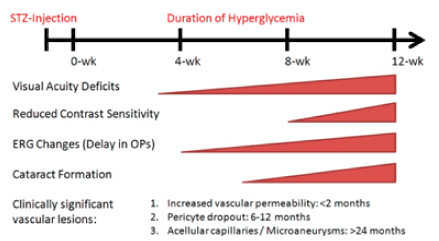
Figure 2. Clinically signficant changes in vision based on published reports on STZ induced Diabetic Rat Model.
Source: Investigative Ophthalmology & Visual Science February 2013, Vol.54, 1370-1377.
Lutein and zeaxanthin are isomers (L/Zi) that differ by the location of a single double bond. Zeaxanthin exist as 3 stereoisomeric forms; (3R, 3’R)-zeaxanthin and (3R, 3’S)-zeaxanthin (also called as meso-zeaxanthin) are the predominant forms present in the macula of the retina, while small amounts of (3S, 3’S)-zeaxanthin have also been detected (Figure 3). Humans are unable to synthesize lutein and zeaxanthin; thus, these nutrients are obtained from natural dietary sources or from supplementation.
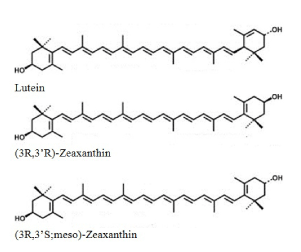
Figure 3. Macular Carotenoid Structures.
Supplementation with lutein/zeaxanthin has been shown to increase circulating and tissue levels of xanthophylls. However, variability in their bioavailabilities has been reported, and proposed to be related to such factors as the matrix of the formulation (e.g., presence of fat), the form in which they administered (i.e., free versus esterified) and interactions with other nutrients. Lutein and zeaxanthin are potent antioxidants, and act as filters of high-energy blue light. Thus, xanthophylls (macular carotenoids) are protective against photo-induced oxidative damage, particularly in highly exposed tissues such as the skin and eyes. In preclinical studies, supplementation with lutein and zeaxanthin reduced generation of reactive oxygen species (ROS) and inflammatory responses resulting from ultraviolet (UV) exposure. Lutein and zeaxanthin isomers are capable of quenching ROS such as singlet oxygen, and are effective scavengers of free radicals [4-7]. Free radicals and other ROS can damage deoxyribonucleic acid (DNA), proteins and lipids, and ultimately affect the integrity and stability of subcellular structures, cells, and tissues [4].
In vivo studies conducted in primates indicated that dietary intake of carotenoids is a critical determinant of concentrations in the retina. Monkeys fed xanthophyll-deficient diets depleted of macula pigment [8-10] and this was reversed with lutein or zeaxanthin supplementation [9,11]. Mice with diabetes have decreases in body weight and increases in blood glucose. As a result, retinal ganglion cells and the inner nuclear layer (INL) undergo apoptosis in this animal model. Results show that lutein prevents reactive oxygen species formation in diabetic mice and rats. Reactive oxygen species (ROS) in the retina is measured using dihydroethidium and visual function is evaluated by electro-retinograms. The decreased amplitude of the oscillatory potentials in the diabetic mice reversed by administration of lutein [12,13]. Nutritional supplementation prevents diabetic retinopathy, and maintains normal retinal function, mitochondrial homeostasis and inflammatory mediators [14]. Thus, the study was designed to investigate the protective ability of SL/Zi on diabetic retinopathy changes, oxidative stress and inflammation in an STZ animal model of DM.
Methodology
Experimental design
Male Wistar strain (WNIN) rats (2 months old; Average BW of 213 ± 14 g) were obtained from the National Center for Laboratory Animal Sciences, National Institute of Nutrition, Hyderabad, India (NCLAS, NIN). Animals were maintained at NCLAS, NIN and kept for acclimatization in experimental room for two weeks. A novel water-soluble L/Zi (SL/Zi, UltraSoL Lutemax 2020 [ 20% Xanthophylls]) formulation and regular L/Zi (RL/Zi, 78% Xanthophylls, Lutemax 2020) were used in the study (OmniActive Health Technologies Ltd., Thane, India) for testing treatments effect on retinal health and diabetes risk factors. Table 1 provides dose for each treatment and diet (nutrient composition). Diabetes was induced in overnight fasted animals by a single intraperitoneal injection of STZ (30 mg/kg) in 0.1 M citrate buffer, pH 4.5. Another set of rats, which received only vehicle, served as the control (Group I; n=12). Fasting blood glucose levels were measured 72 h after STZ injection. Blood glucose levels >150 mg/dL considered diabetic and rats divided into four groups (Group I-IV, Table 1). All the animals were housed in each cage were maintained on their diets for 12 weeks and drinking water was provided ad libitum. Daily food intake, body weights and fasting glucose levels were recorded. Electro-retinogram (ERG) performed to all rats and HbA1C levels estimated. At the end of 12 weeks rats were euthanized and retinas harvested for histological and morphological changes.
Table 1. Composition of diets for different treatments.
Groups |
Treatments |
Diet |
I |
Control Group, N=6 |
AIN 93 |
II |
STZ DM Group, N=9 |
AIN 93 |
III |
Group II + Regular lutein/Zeaxanthin isomers (RL/Zi), N=6 |
AIN 93 with regular lutein 0.5 % |
IV |
Group II + Soluble lutein/Zeaxanthin isomers (SL/Zi), N=8 |
AIN 93 with soluble lutein 0.5 % |
Electro-Retinogram (ERG) analysis
Diabetic retinopathy (DR) is characterized by retinal function disturbances and microvascular complication of DM. Retinal function was assessed by electro-retinogram (ERG). Diabetes results in ischemia and apoptosis in different retinal cell layers results in changes in the functions of the retina. Oscillatory potentials (OPs) were more affected in DM than a- or b- waves. OPs represent the functional aspects of inner retinal layers, ganglion cell layer and inner plexiform layer.
Animals were dark-adapted for overnight and ready for the ERG procedure under dim red illumination. The pupils of the rats were dilated with atropine eye drops. The ground electrode was a sub-cutaneous needle in the tail, and the reference electrode an ear clip electrode. The active contact lens electrodes were placed on the cornea. The recordings were performed with a UTAS Visual Diagnostic System. The responses differentially amplified with a gain of 1,000 using alternating current-coupled UBA-4204 Amplifier. A flash stimuli of -2 to 8 dB was delivered via a with BigShot™ Ganzfeld System (LKC Technologies; Gaithersburg, MD, USA). The oscillatory potentials were extracted from the wave form and the sum of all OPs calculated.
Histopathology
The eyeballs from selected animals were collected in fixative 4 % paraformaldehyde solution in separately labeled vials. Samples were kept at room temperature for 24-48 hrs followed with replacing the fixative with 20 mM sodium phosphate buffer. Buffer was replaced with fresh buffer once weekly until histopathological processing. Tissues were embedded in paraffin and sections taken in microtome. Coated slides were used for immunohistochemistry and immunofluorescence, because uncoated slides were used for H & E staining.
Quantitative real time PCR
RNA isolated from the rats and quantifies mRNA of various genes including nerve growth factor (NGF), glial acidic fibrallary protein (GFAP), vascular endothelial growth factor (VEGF), rhodopsin, hypoxia-inducible factor (HIF) and platelet-derived growth factor (PDGF) in the retina of experimental rats by means of quantitative real-time PCR [9-11].
Immunohistochemistry
Retinas were fixed in 4% paraformaldehyde in sodium phosphate buffer (pH 7.2), followed by embedding and sectioning for paraffin using standard protocols. Three retinas were used for each group. Immunolocalization of GFAP, VEGF and rhodopsin were carried out on 4 μM thick paraffin sections of rat retinas. The protein localized by staining in the retina sections by addition of DAB solution containing H2O2 or fluorescent tagged antibodies. Slides were observed under a microscope (# LMD 6000, Leica Microsystems, Germany). The complete retinal morphology including thickness of various layers were studied in detail in the retinal sections.
Immunoblotting: In addition to immunohistochemistry, quantified protein levels of VEGF, GFAP and rhodopsin in the retinas of experimental rats using immunoblotting. Housekeeping protein β-actin used as a loading control. Quantification of band intensity was reported using Image J software.
Statistical analysis
The data was expressed as mean ± SEM. Results were analyzed for statistical significance by one way ANOVA followed by Dunnett’s multiple comparison tests for comparing all the groups with control group. Between group significance was checked by two tailed unpaired t-test. All analyses were performed with significance set at P less than 0.05.
Results
Body weights and average daily food intake
The diabetic control rats had significantly reduced body weight (p<0.001) compared to age matched non-diabetic control rats. The diabetic control rats consumed much (p<0.001) more food than the non-diabetic control rats (Figure 4A & 4B). Although the amount of food consumed by the diabetic rats treated with RL/Zi and SL/Zi was less compared to that consumed by the diabetic control rats, there was no significant difference. Despite the increased food intake, the body weight of diabetic (D) animals decreased when compared with age matched non diabetic controls (C) (P<0.001).
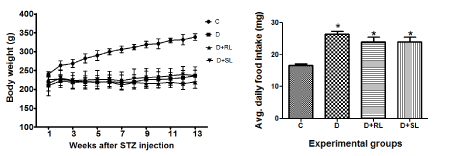
Figure 4. A body weight and 4B average daily food intake.
Fasting blood glucose
The plasma glucose concentrations of the diabetic control rats was higher than those of the non-diabetic control rats throughout the experiment. A small decrease in fasting plasma glucose levels was observed in groups treated with SL/Zi but no significant effect of treatment was observed on plasma glucose in diabetic rats (Figure 5A).
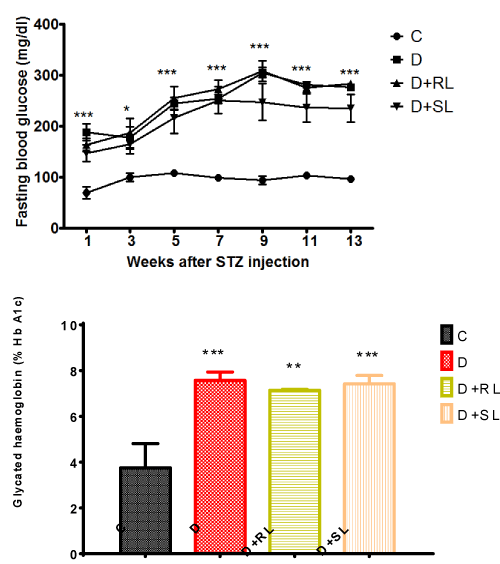
Figure 5. A and 5B S_L/Zi Decreased Fasting Blood Glucose and HbA1c over Diabetes group.
Glycated haemoglobin (HbA1c)
Glycated haemoglobin (HbA1c) is a better indicator than fasting blood glucose levels of the glycemic status in diabetics. A significant increase in HbA1c levels was observed in diabetic animals (>6.5 %). Treatment with SL/Zi decrease HbA1c percentage moderately but statistically not significant (Figure 5B). These results correlated with the fasting blood glucose pattern between groups and no significant changes observed with RL/Zi.
Diabetic retinopathy
Electro-radiograph
In diabetic rats (D) the amplitude of OPs was reduced (334.2 µV) compared to normal control (C) animals. Ingestion of L/Zi resulted in lowering the reduction in OP amplitudes suggested by sum of Ops compared with RL/Zi (442.6), SL/Zi (561.9). SL/Zi normalised the implicit times (Figure 6).
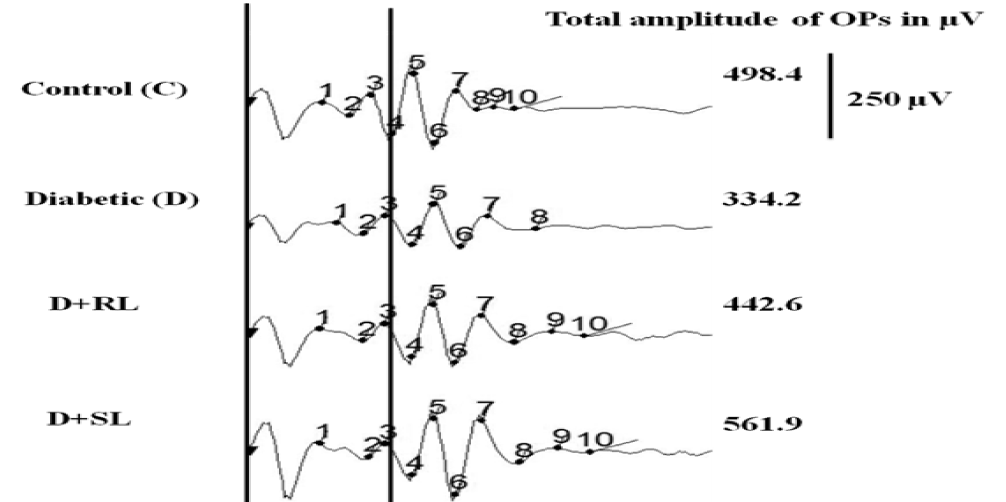
Figure 6. S_L/Zi increased total amplitude Ops over diabetes group.
H & E Staining
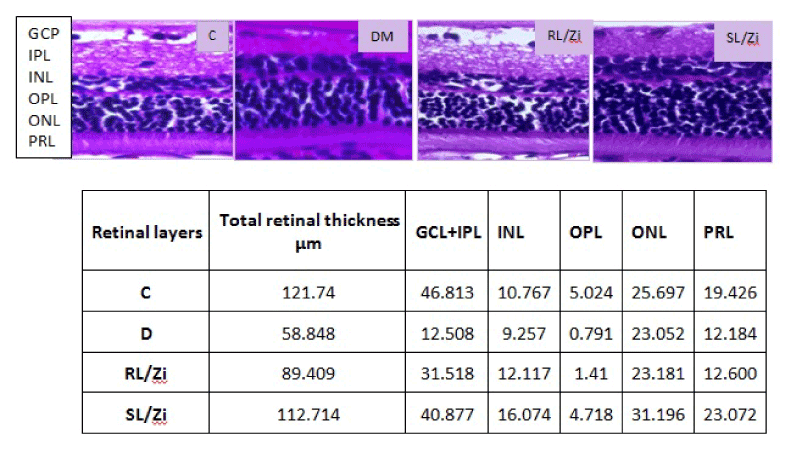
In control rats (C), all retinal layers were intact with maximum thickness of retina and noted with dense inner nuclear layer (INL) and distinguishable separation of outer plexiform layer (OPL) between INL and outer nuclear layer (ONL). In contrast, retinas of diabetic rats (D) showed much reduced total retinal thickness and was marked by less dense ONL and almost merged INL and ONLs. The greatest reduction in thickness was in the inner plexiform layer (IPL), inner nuclear layer (INL) and ONL. Treatment with L/Zi prevented gross morphological changes to a significant extent in diabetic retina. Soluble L/Zi treatment increased total retinal thickness compared with other groups. Soluble L/Zi enhanced ganglion cell layer (GCL), inner plexiform layer (IPL), inner nuclear layer (INL), outer plexiform layer (OPL), outer nuclear layer (ONL), and photo receptor layer (PRL) thickness than other treatment groups. However, soluble L/Zi is more effective than regular L/Zi, indicated by dense ONLs. Morphological changes in the retina was associated with functional impairment as well as temporal changes in retinal gene expression in diabetic rats and L/Zi prevented decline in the expression of rhodopsin and nerve growth factor which have vital role in maintaining the health of the retina. L/Zi prevented over expression of HIF1α, GFAP and VEGF involved in stress and angiogenesis (Figure7).
Representative wave forms of OPs from individual animals of different groups and total amplitudes.
Figure 7. Diabetes induced gross structural alterations (Retinal layers’ Thickness, µm).
GCL: ganglion cell layer; IPL: inner plexiform layer; INL: inner nuclear layer; OPL: outer plexiform layer; ONL: outer nuclear layer; PRL: photo receptor layer.
Immunofluorescence
Rhodopsin (Rho)
Rhodopsin is a biological pigment in photoreceptor cells of the retina responsible for the first events in the perception of light Immunofluorescence imaging of Rho protein showed its decreased expression in diabetic rat retina in comparison to normal control rat retina. Treatment with RL/Zi and SL/Zi prevented loss of Rho protein expression in diabetic retina indicated by intensed Rho positive fluorescence. Furthermore, RL/Zi and SL/Zi resulted in preventing loss of Rho protein expression in rat retina in diabetic animals (Figure 7A).
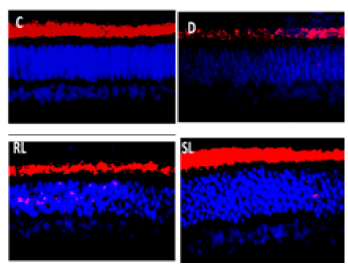
Figure 7A. Rhodopsin enhanced with SL/Zi.
Nerve growth factor (NGF)
Immunofluorescence imaging of NGF protein showed its decreased expression in diabetic rat retina in comparison to normal control rat’s retina (Figure 7B). Treatment with RL/Zi and SL/Zi prevented loss of NGF protein expression in diabetic retina indicated by intense NGF positive fluorescence. The mRNA levels of NGF in the retinal of various experimental groups shown in Figure 7B.
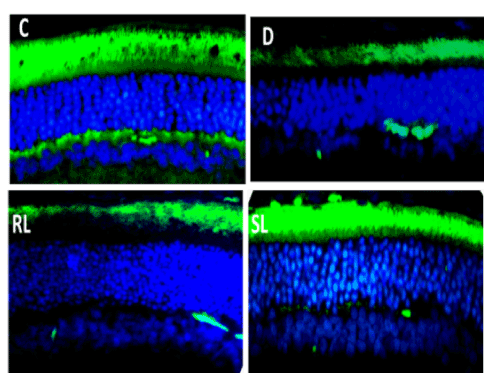
Figure 7B. SL/Zi prevented loss of NGF.
Hypoxia-inducible factor 1-alpha (HIF-1-alpha)
HIF1α is expressed at low oxygen2021 Copyright OAT. All rights reservl role in cellular and systemic responses to hypoxia. Immunofluorescence imaging of HIF1 α protein expression increased in diabetic rat’s retina in comparison to normal control rats (Figure 7C). Treatment with RL/Zi and SL/Zi decreased protein expression in diabetic retina indicated by lower HIF1 α positive fluorescence indicating the prevention of hypoxia in the retinas.
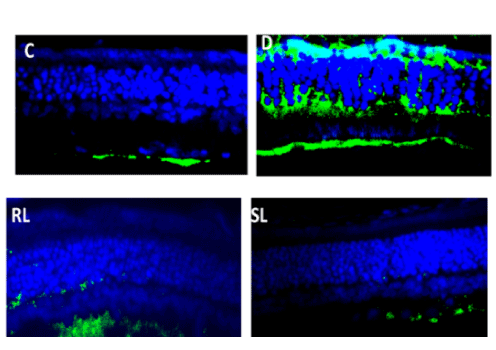
Figure 7C. SL/Zi decreased Hif1-alpha.
Glial fibrillary acidic protein (GFAP)
GFAP involved in many important central nervous system processes, including cell communication, cell strength and shape and the functioning of the blood brain barrier. In diabetic rats the mRNA levels of GFAP levels were up regulated drastically. Treatment with SL/Zi prevented its increase, there was no significant effect of RL/Zi treatment (Figure 7D).
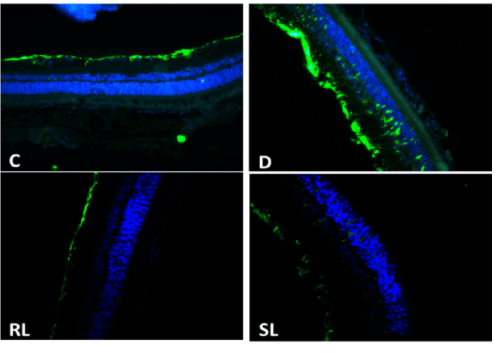
Figure 7D. GFAP down regulated with SL/Zi.
Vascular endothelial growth factor (VEGF)
Vascular endothelial growth factor is (VEGF) a signal protein produced by cells that stimulates vasculogenesis and angiogenesis. When VEGF is overexpressed, it can lead to disease condition. Western blotting for VEGF indicated up regulation of VEGF expression (Figure 7E) in diabetic retina. Treatment with RL/Zi did not affect VEGF over expression in diabetic retina but treatment with SL/Zi reduced VEGF expression.
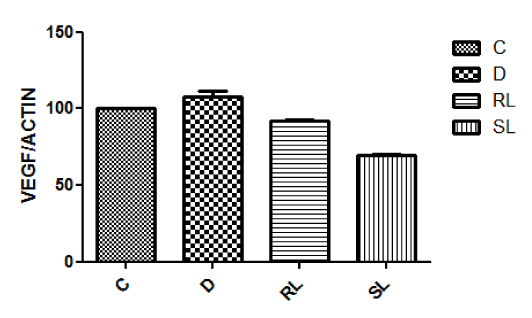
Figure 7E. Vascular Endothelial Growth Factor decreased with SL/Zi treatment.
Discussion
L/Zi supplementation reduces oxidative stress and damage to the retinal mitochondria, and regulates VEGF and inflammatory mediators increased in diabetes. Thus, L/Zi supplementation, which is now being tested for diabetes-related visual dysfunction, appears to have potential to inhibit the development of diabetic retinopathy. Muriach et al. [15] reported the effects of lutein on diabetic retinal changes in alloxan-induced diabetic mice. These mice showed an increase of malondialdehyde (MDA, a marker for lipid peroxidation) and nuclear factor κB (NFκB) levels, along with a decrease in glutathione (GSH) levels and glutathione peroxidase (GPx) activities in the retina. Electroretinography (ERG) b-wave amplitude decreased in diabetic mice. Lutein [70% purity, 0.2 mg/kg body weight (wt), administered by stomach tube] was used as daily treatment started on day 4 after alloxan injection and lasted until the end of the experiment. While lutein treatment did not alter the hyperglycemic status of alloxan diabetic mice, supplementation of lutein restored levels/activities of NFκB, MDA, GSH, and GPx in the retina. ERG b-wave amplitude was also restored to normal after lutein treatment, suggesting that although the high blood glucose levels were not normalized, biochemical and functional changes in the diabetic retina were improved by supplementation of lutein.
Carotenoids are powerful antioxidants, and by scavenging free radicals, they protect the cells from the damage caused by free radicals [16-17]. Antioxidant properties of carotenoids are routinely linked with their beneficial effects on chronic diseases including diabetes, and the uptake of lutein and zeaxanthin in the retina is decreased in diabetes [18].
Our results suggest L/Zi supplementation prevents the development of diabetic retinopathy by protecting DNA from undergoing damage, and thus preventing the initiation of the self-propagating cycle. NGF is an important regulator of retinal development and also plays a key role in regulating survival, growth, and functional maintenance of retinal ganglion cells, photoreceptors and other retinal neurons. Immunofluorescence imaging of NGF protein showed its decreased expression in diabetic rat retina in comparison to normal control rat retina.
Rhodopsin is a biological pigment in photoreceptor cells of the retina responsible for first events in perception of light. RL was unable to prevent the loss of Rho protein effectively in diabetic rat retina. Furthermore, SL is more effective. Hypoxia-inducible factor 1-alpha (HIF-1-alpha) was expressed at low oxygen concentrations and plays an essential role in cellular and systemic responses to hypoxia. Immunofluorescence imaging of HIF1A protein showed its increased expression in diabetic rat retina in comparison to normal control rat retina in various layers of the retina. Treatment with RL/Zi and Sl/Zi showed decreased protein expression in diabetic retina indicated by lower HIF1A positive fluorescence indicating prevention of hypoxia in the retinas.
Glial fibrillary acidic protein (GFAP) involved in many important central nervous system processes, including cell communication, cell strength and shape and the functioning of the blood brain barrier. SL found to be more effective. In diabetic rats the mRNA levels of GFAP are up regulated drastically, while treatment with RL andSL significantly prevented its increase but there is no significant effect of RL treatment. Among these three SL is showed to have more effective.
VEGF, a hypoxia-induced factor is considered as major growth factors in the development of diabetic retinopathy [19-20] Diabetes-induced increase in VEGF and cell permeability during early stages of diabetic retinopathy, and angiogenesis. RL and SL inhibited VEGF expression. Antioxidants administration protects retina from increases in VEGF [21] and attenuates increase in retinal VEGF [22]. Retinal VEGF remains normal in diabetic rats treated with nutrients. This clearly implies that carotenoids protect increase in VEGF, both in the early stages and later stages of diabetic retinopathy.
NFkB a redox-sensitive transcriptional factor, controls transcription of DNA, and activate pro-inflammatory genes. Diabetes activates NF-kB in retina and vascular cells, and act as pro-apoptotic [23,24]. L/Zi supplementation on diabetic retinopathy was mediated via inhibition of both inflammation and mitochondrial damage (Figure 8). Saskai et al. [25] reported levels of STAT3 activation, downstream of inflammatory cytokine signals, and reactive oxygen species (ROS), which are both upregulated during EIU, were reduced by lutein. Reactive oxygen species (ROS) levels in the retina increased degenerative changes in retina. L/Zi protects retina against diabetic damage because of its antioxidant properties. Supplementation of L/Zi significantly decreased elevation of lipid peroxide, oxidative modified DNA, nitrotyrosine, iNOS, VEGF, and ICAM-1; and levels of electron transport complex III increased to normal. The nature defense system against oxidative stress, Mn superoxide dismutase (SOD), and GSH decreased significantly in diabetic retinas [26].
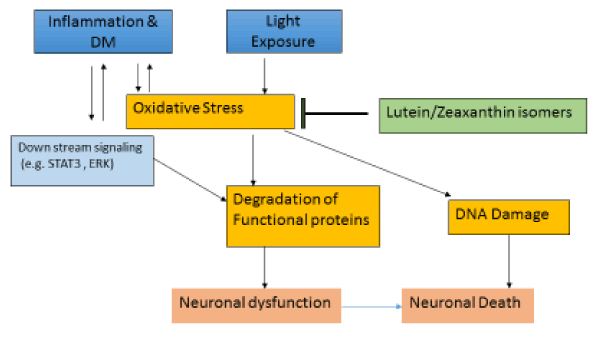
Figure 8. Potential mechanism of action of L/Zi.
Our data show SL/Zi supplementation protects neuronal cells and vascular cells, and inhibits development of retinopathy. SL/Zi supplementation ameliorates increase in inflammatory mediators and maintaining mitochondria homeostasis, and protecting the retina from self-propagating vicious cycle of mitochondrial damage. SL/Zi supplementation appears an inexpensive adjunct therapy to inhibit retinal dysfunction, and to be onset of blinding disease. SL/Zi ameliorates oxidative stress, and prevent/retard diabetic retinopathy. In addition, LZi preserve retinal structural and functional abnormalities associated with diabetes.
Acknowledgements
We wish to thank National Institute of Nutrition. The study was sponsored by OmniActive Health Technologies Ltd., India.
Conflict of Interest
DJ, JS and VJ are employees of OmniActive Health Technologies.
Note: Presented part of the data at International Carotenoid Society, Utah, 2014; Ophthalmology 2014.
References:
- http://www.cdc.gov/visionhealth/pdf/factsheet.pdf (retrieved on 05/08/2016)
- Zheng Y, He M, Congdon N (2012) The worldwide epidemic of diabetic retinopathy. Indian J Ophthalmol 60: 428-431. [Crossref]
- IDF Diabetes Atlas (2011). 5th ed. Brussels, Belgium: International Diabetes Federation. International Diabetes Federation (retrieved on 05/08/2016)
- Stahl W, Sies H (2002) Carotenoids and protection against solar UV radiation. Skin Pharmacol Appl Skin Physiol 15: 291-296. [Crossref]
- Stahl W, Sies H (2003) Antioxidant activity of carotenoids. Mol Aspects Med 24: 345-351. [Crossref]
- Sies H, Stahl W (2003) Non-nutritive bioactive constituents of plants: lycopene, lutein and zeaxanthin. Int J Vitam Nutr Res 73: 95-100. [Crossref]
- Loane E, Kelliher C, Beatty S, Nolan JM (2008) The rationale and evidence base for a protective role of macular pigment in age-related maculopathy. Br J Ophthalmol 92: 1163-1168. [Crossref]
- Malinow MR, Feeney-Burns L, Peterson LH, Klein ML, Neuringer M (1980) Diet-related macular anomalies in monkeys. Invest Ophthalmol Vis Sci 19: 857-863. [Crossref]
- Neuringer M, Sandstrom MM, Johnson EJ, Snodderly DM (2004) Nutritional manipulation of primate retinas, I: effects of lutein or zeaxanthin supplements on serum and macular pigment in xanthophylls-free rhesus monkeys. Invest Ophthalmol Vis Sci 45: 3234-3243. [Crossref]
- Johnson EJ, Neuringer M, Russell RM, Schalch W, Snodderly DM (2005) Nutritional manipulation of primate retinas, III: Effects of lutein or zeaxanthin supplementation on adipose tissue and retina of xanthophyll-free monkeys. Invest Ophthalmol Vis Sci 46: 692-702. [Crossref]
- Barker FM II, Snodderly DM, Johnson EJ, Schalch W, Koepcke W, et al. (2011) Nutritional manipulation of primate retinas, V: effects of lutein, zeaxanthin, and n-3 fatty acids on retinal sensitivity to blue-light-induced damage. Invest Ophthalmol Vis Sci 52: 3934-3942. [Crossref]
- Tang L, Zhang Y, Jiang Y, Willard L, Ortiz E, et al. (2011) Dietary wolfberry ameliorates retinal structure abnormalities in db/db mice at the early stage of diabetes. Exp Biol Med (Maywood) 236: 1051-1063. [Crossref]
- Hu BJ, Hu YN, Lin S, Ma WJ, Li XR (2011) Application of Lutein and Zeaxanthin in nonproliferative diabetic retinopathy. Int J Ophthalmol 4: 303-306. [Crossref]
- Kowluru RA, Zhong Q, Santos JM, Thandampallayam M, Putt D, et al. (2014) Beneficial effects of the nutritional supplements on the development of diabetic retinopathy. Nutr Metab (Lond) 11: 8. [Crossref]
- Muriach M, Bosch-Morell F, Alexander G, Blomhoff R, Barcia J, et al. (2006) Lutein effect on retina and hippocampus of diabetic mice. Free Radic Biol Med 41: 979-984. [Crossref]
- Whitehead AJ, Mares JA, Danis RP (2006) Macular pigment: a review of current knowledge. Arch Ophthalmol 124: 1038-1045. [Crossref]
- McNulty H, Jacob RF, Mason RP (2008) Biologic activity of carotenoids related to distinct membrane physicochemical interactions. Am J Cardiol 101: 20D-29D. [Crossref]
- Lima VC, Rosen RB, Maia M, Prata TS, Dorairaj S, et al. (2010) Macular pigment optical density measured by dual-wavelength autofluorescence imaging in diabetic and nondiabetic patients: a comparative study. Invest Ophthalmol Vis Sci 51: 5840-5845. [Crossref]
- Aiello LP (2005) Angiogenic pathways in diabetic retinopathy. N Engl J Med 353: 839-841. [Crossref]
- Aiello LP, Brusell SE, Clermont A, Duh E, Ishii H, et al. (1997) Vascular endothelial growth factor-induced retinal permeability is mediated by protein kinase c in vivo and suppressed by an orally effective beta isoform-selective inhibitor. Diabetes 46: 1473-1480. [Crossref]
- Obrosova IG, Minchenko AG, Marinescu V, Fathallah L, Kennedy A, et al. (2001) Antioxidants attenuate early up regulation of retinal vascular endothelial growth factor in streptozotocin-diabetic rats. Diabetologia 44: 1102-1110. [Crossref]
- Kowluru RA, Zhong Q, Santos JM, Thandampallayam M, Putt D, et al. (2014) Beneficial effects of the nutritional supplements on the development of diabetic retinopathy. Nutr Metab (Lond) 11: 8. [Crossref]
- Kowluru RA, Koppolu P, Chakrabarti S, Chen S (2003) Diabetes-induced activation of nuclear transcriptional factor in the retina, and its inhibition by antioxidants. Free Radic Res 37: 1169-1180. [Crossref]
- Romeo G1, Liu WH, Asnaghi V, Kern TS, Lorenzi M (2002) Activation of nuclear factor-kappaB induced by diabetes and high glucose regulates a proapoptotic program in retinal pericytes. Diabetes 51: 2241-2248. [Crossref]
- Sasaki M, Ozawa Y, Kurihara T, Noda K, Imamura Y, et al. (2009) Neuroprotective effect of an antioxidant, lutein, during retinal inflammation. Invest Ophthalmol Vis Sci 50: 1433-1439. [Crossref]
- Kowluru RA, Menon B, Gierhart DL (2008) Beneficial effect of zeaxanthin on retinal metabolic abnormalities in diabetic rats. Invest Ophthalmol Vis Sci 49: 1645-1651. [Crossref]













