Abstract
Simpson-Golabi-Behmel syndrome (SGBS) is a rare inherited X-linked disorder which is characterized by craniofacial, cardiac, skeletal and renal anomalies. Prenatal somatic and visceral overgrowth which persists in the postnatal period is the most remarkable feature of this condition. The perinatal period is marked by risk of asphyxia, respiratory depression necessitating assisted ventilation, difficulty in maintenance of airway, risk of aspiration, transient metabolic disturbances like hypoglycemia, hypocalcemia, hyperbilirubinemia and feeding difficulty. Airway management during the neonatal period and subsequently anesthetic management during surgical correction of congenital anomalies is challenging. Abnormal airway anatomy inherent to SGBS warrants a meticulous preoperative assessment and perioperative management. We present the perinatal management, preoperative and intraoperative anesthetic management when the infant underwent reconstructive surgery for cleft lip and cleft palate. The perinatal period was marked by resuscitation at birth and subsequently requiring respiratory support, management for hypoglycemia, hyperbilirubinemia, early enteral feeding and anti-reflux measures. Intraoperatively inhalational anesthesia with spontaneous ventilation without neuromuscular blockade was used. Anticipation of a difficult airway with measures for tackling macroglossia, complete avoidance of neuromuscular blocking agents, avoiding excessive use of opioids and deep extubation form the tenets of anesthetic management in this case. Surgical closure of the cleft palate reduces the size of the oral cavity available for laryngoscopy and suctioning in addition to that due to the macroglossia, retrognathia and surgical packs. Thus re-intubation in these cases can be a daunting task which underlines the need to have a clear anesthetic plan.
Key words
Simpson-golabi-behmel syndrome, macrosomia, macroglossia, cleft palate
Introduction
Simpson-Golabi-Behmel syndrome (SGBS) is a rare inherited congenital disorder which is a constellation of craniofacial, cardiac, skeletal and renal anomalies. This syndrome is the result of mutations in an X-linked gene encoding glypican-3 or glypican-4 (GPC3 or GPC4) which is responsible for control of growth of embryonic mesodermal tissues. SGBS was initially described by Simpson et al. in 1975 [1]. In 1984 Golabi, Rosen and Behmel independently reported more cases [2,3]. Neri et al. put all these reports together and named the disorder ‘Simpson-Golabi-Behmel syndrome’ [4]. There are two clinical subtypes, the classical form (SGBS type I) due to mutations in GPC3 and a lethal form (SGBS type II) which is extremely rare [5,6]. The condition is characterized by pre and postnatal overgrowth, visceral, skeletal and neurological abnormalities. There is an increased risk of neoplasms of embryonal origin which usually appear by end of first decade of life [7]. Commonly seen anomalies in SGBS which can lead to airway problems include macroglossia, cleft lip and palate, retrognathia and short neck. Airway obstruction is the major problem during the anesthetic management of SGBS requiring appropriate preoperative planning. The principle of managing difficult airway in this situation is to maintain spontaneous ventilation until the airway is secured [8]. Till date there are few published case reports on the anesthetic management of SGBS [9]. We present the perinatal management, preoperative and intraoperative anesthetic management of a 1½ year old child with SGBS undergoing reconstructive surgery for cleft lip and cleft palate.
Case report
The index case, a 4.6 kg male neonate was delivered to a 26 year old primigravida by elective caesarean section (LSCS) at 38 weeks period of gestation. The indication for LSCS was fetal macrosomia. The infant was born to non-consanguineous couple from central Indian state following an uneventful antenatal period. The fetal biometry had consistently showed growth to be greater than the 97th centile and bilateral nephromegaly on ultrasound (Figure 1). There was no maternal diabetes and the fetal echo was normal. The birth weight of 4.6 kg, length of 55 cm and occipito-frontal circumference (OFC) of 38 cm were all > 97th centile. The baby was noted to have bilateral cleft lip and palate with a protruding premaxilla and deficient columella (Figure 2). Based on the constellation of macrosomia, bilateral cleft lip and palate and nephromegaly the neonate was identified to have Simpson-Golabi-Behmel fetal overgrowth syndrome. The baby did not cry at birth. Positive pressure ventilation (PPV) with Bag and mask was ineffective due to ineffective seal of the face mask and baby required placement of a size 1 laryngeal mask airway (LMA) which allowed effective PPV. Postnatally, neonatal intensive care unit (NICU) admission was remarkable for the difficulty in airway management and ventilation. The large midline cleft palate, retrognathia and macroglossia creating a Robin sequence resulted in difficulty in intubating and the large size made the bassinet of the radiant warmer relatively small and cramped. The baby required assisted ventilation for 3 days during which there was need for sedation to prevent agitation and asynchrony during periods of arousal. There were no episodes of hypoglycemia as the baby was managed with IV 10% Dextrose. Unconjugated hyperbilirubinemia was managed by intensive double surface phototherapy for 72 hours. The maximum total serum bilirubin was 19 mg/dl. During the admission in the postnatal ward, baby required oro-gastric tube feeding and anti-reflux measures. The kidney ultrasounds showed bilateral nephromegaly with the kidney size reported as 78 mm and 68 mm on the left and right side respectively while the liver size and span was normal. Postnatal echo showed a 4 mm secundum atrial septal defect (ASD). The baby was discharged to home on oral feeding using a long-teat feeding bottle with the family having been counseled and explained care, maintenance and hazards of feeding bottle. The infant was given a palatal obturator as a short term measure to allow oral feeding without the risk of oro-nasal reflux and aspiration pneumonia (Figure 3). Follow-up at 5 months age with CT abdomen for hepatoblastoma and renal tumours was negative showing only bilateral nephromegaly (Figure 4). The renal function tests were normal and serum alpha-fetoprotein (AFP) levels were not elevated. The infant continued to grow well and attained milestones appropriate for age (Figure 5). The baby underwent repair of cleft lip at the age of 9 months and repair of the cleft palate at 1½ year age (Figure 6a). The anesthetic management during both the surgeries involved anticipation of difficulty in mask holding, laryngoscopy, intubation, post-extubation airway obstruction by tongue fall and difficulty in maintaining a patent airway till the time the infant was fully awake. Sevoflurane-fentanyl anesthesia without a neuromuscular blockade was planned. The child was kept nil orally for 08 hours prior to surgery. No episodes of hypoglycemia were noted as he was maintained on infusion with 0.45% dextrose normal saline overnight. Anticipating difficult intubation and ventilation, difficult airway cart with LMA, I-gel, pediatric intubating fibre-optic bronchoscope (FOB), different sizes of face masks, nasopharyngeal airway, stylet and bougie were kept ready. The child was premedicated with IV glycopyrrolate and fentanyl 02 min before induction, pre-oxygenated and induced with inhalational agent 8% sevoflurane. Giving fentanyl pre-induction was helpful in making the intubation smooth without any pressor response. A well-fitting pediatric facemask was used along with oral airway as the tongue was large and would have surely obstructed the airway and made ventilation difficult. Once adequate depth of anesthesia was confirmed, a size 2 Miller blade was used for laryngoscopy and the child was intubated and airway secured with 4.0 mm ID un-cuffed endotracheal tube. Anesthesia was maintained with only 3% sevoflurane, nitrous oxide and oxygen with spontaneous respiration throughout the surgical procedure. Difficulty in ventilation due to macroglossia was overcome by orophyrangeal airway. Hypothermia was prevented by using fluid warmer set at 39°C and Bear-Hugger patient warmer. The child was extubated fully awake after gentle suctioning and insertion of diclofenac sodium suppository. A smooth fully awake extubation was planned to avoid post-operative complications like laryngospasm, tongue fall and chance of re-intubation. The postoperative period was unremarkable. The child is on regular follow-up and is continuing to show postnatal overgrowth (Figure 6b). On formal testing his developmental quotient (DQ) is within the normal limits. He is enrolled in a speech therapy program and is being periodically screened for embryonal tumors.
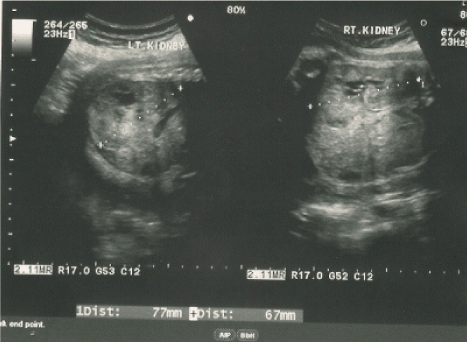
Figure 1. Antenatal Ultrasound at 29 weeks period of gestation showing large bilateral kidneys measuring 77 mm on left side and 67 mm on right side.
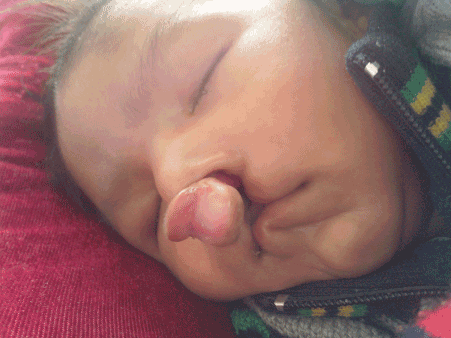
Figure 2. The infant with bilateral cleft lip and palate with floating premaxilla and deficient collumella.
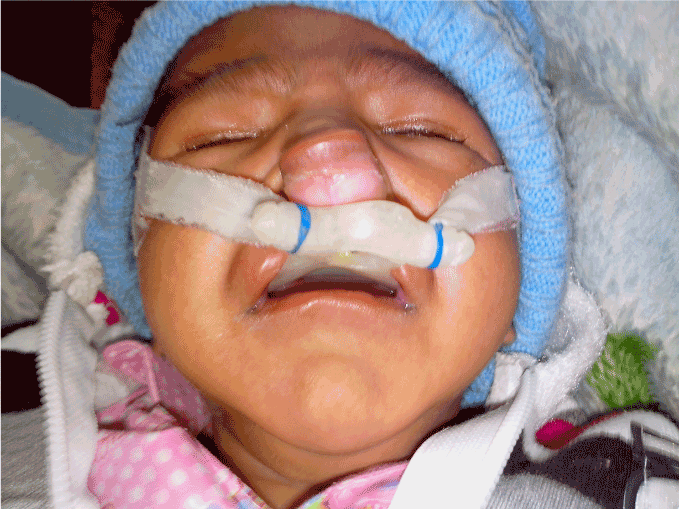
Figure 3. The infant with the palatal obturator fitted to allow oral feeding without the risk of oro-nasal regurgitation of feed or the risk of aspiration pneumonia
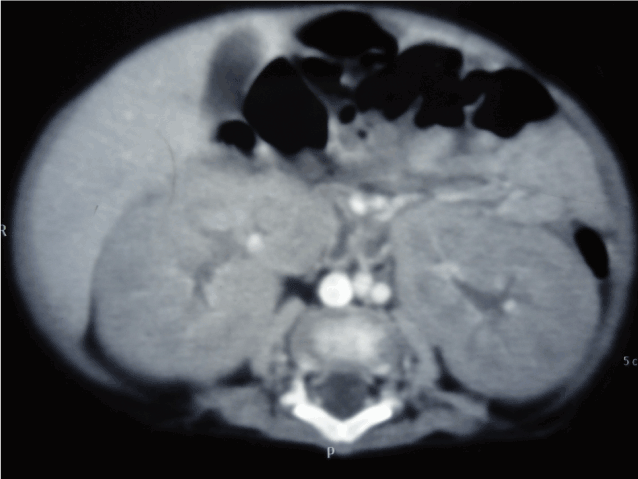
Figure 4. CTScan abdomen at 2 months age showing bilateral nephromegaly with no hepatic space occupying lesions (SOL’s).
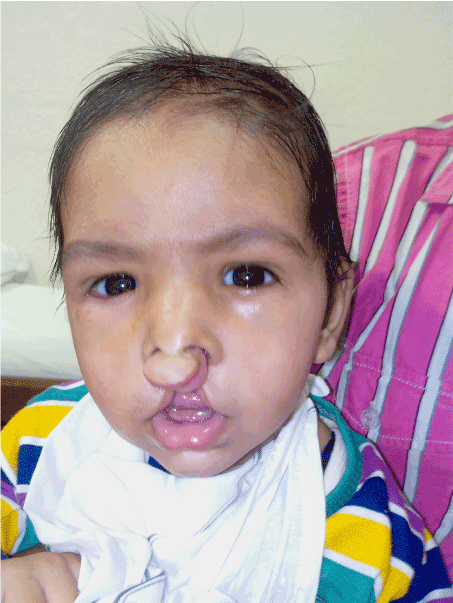
Figure 5. Infant at 9 months of age showing bright look and age appropriate attainment of developmental milestones.

Figure 6a. The child at 1½ year age after surgery for bilateral cleft lip and palate.
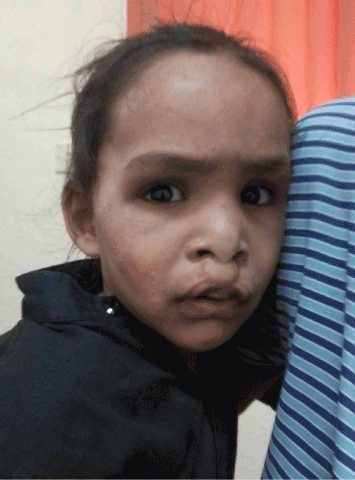
Figure 6b. The child at 2 ½ year age showing postnatal overgrowth.
Discussion
SGBS is a rare true overgrowth syndrome inherited in an X-linked recessive manner. Our case had macrosomia, macrocrania, craniofacial dysmorphism in the form of hypertelerorism, bilateral cleft lip and palate, protruding premaxilla, deficient columella, macroglossia and nephromegaly and hence was identified to have Simpson-Golabi-Behmel fetal overgrowth syndrome. In addition these infants may have generalized hypotonia, congenital heart defects or neoplasms of embryonal origin which include hepatoblastoma, gonadoblastoma, wilms tumour and neuroblastoma [10,11]. An association with laryngeal web has also been described [12]. SGBS needs to be differentiated from other conditions with overgrowth such as Beckwith-Wiedemann Syndrome, Pallister-Killian Syndrome, Sotos Syndrome and Perlman syndrome [13]. Persistent overgrowth including macroglossia in the postnatal period extending into adulthood differentiates SGBS from other overgrowth entities [14,15]. Genetic studies include a karyotype, array comparative genomic hybridization (aCGH), mutation analysis for the GPC3 and GPC4 gene using Next generation sequencing [16]. Genetic tests were not done in our case due to resource limitation. Earlier reports have highlighted airway and anesthetic complication of upper airway obstruction due to macroglossia but not due to cleft palate [17,18]. Features which suggested an impending difficult airway in our case were macroglossia, bilateral cleft lip and palate, protruding premaxilla and retrognathia. These findings along with the history of airway difficulty experienced during the neonatal period prompted deliberate pre-operative planning. Anticipating difficult intubation and ventilation, difficult airway cart was kept in readiness. Due to the high risk of losing airway control, anesthetic induction with an inhalational agent such as sevoflurane, together with maintenance of spontaneous ventilation, was preferred over intravenous induction with propofol and neuromuscular blockade. With this approach if indeed airway obstruction did occur inhalational anesthetic administration could be discontinued and the child woken up [19]. A recent study reported that use of fentanyl allowed a dose-related smooth intubation and calm emergence in children anesthetized with sevoflurane and the same was done in our case [20]. Anticipating difficulty in ventilation, laryngoscopy and intubation an orophyrangeal airway, well-fitting facemask, a size 2 Miller blade and intravenous fentanyl was used along with achieving adequate depth of anesthesia before intubating the baby. Postoperatively the child was extubated fully awake. This case highlights the problems faced in the perinatal period and preoperative and intraoperative anesthetic management. The basic principle of airway management of anticipating and identifying a difficult airway and maintenance of spontaneous ventilation until the airway is secured were followed in this case. Adequate emphasis on macroglossia, avoidance of neuromuscular blockade and excessive use of fentanyl and deep extubation were followed. An important observation in this case was that at the end of cleft palate surgery, the large defect in hard palate which earlier allowed additional space to displace at least some part of the large tongue was closed. Additionally presence of surgical packs, retrognathia and macroglossia reduce the size of the oral cavity practically available for laryngoscopy and suctioning. Under these conditions if the child is not fully awake, tongue fall, clots and secretions can cause severe airway obstruction requiring reintubation which can be challenging. These difficulties are inherent to anesthetic management of a case of SGBS with macroglossia for cleft palate repair. This case report brings to the fore, a rare fetal overgrowth syndrome and the two time points of perinatal and intraoperative period which pose a threat to the infant and where appropriate airway management and intensive care can be rewarding towards the patients clinical outcome. We also would like to bring to attention, the observation of further narrowing of the oral cavity following closure of the cleft palate which can create difficulty in reintubation, thus underlying the necessity of having a well-conceived anesthetic plan. The recommended anesthetic technique should be of anticipating and preparing for a difficult airway, allowing spontaneous ventilation until the airway is secure, avoiding neuromuscular blockade, excessive use of opioids and prevention of need for reintubation.
Consent
Written informed consent was obtained from the patient for publication of this Case report and accompanying images.
References
- Simpson JL, Landey S, New M, German J (1975) A previously unrecognized X-linked syndrome of dysmorphia. Birth Defects Orig Artic Ser 11: 18–24. [Crossref]
- Golabi M, Rosen L (1984) A new X-linked mental retardation overgrowth syndrome. Am J Med Genet 17: 345–358. [Crossref]
- Behmel A, Plochl E, Rosenkranz W (1984) A new X-linked dysplasia gigantism syndrome: identical with the Simpson dysplasia syndrome? Human Genet 67: 409–413. [Crossref]
- Neri G, Marini R, Cappa M, Borrelli P, Opitz JM (1998) Simpson-Golabi-Behmel syndrome: an X-linked encephalo-tropho-schisis syndrome. Am J Med Genet 30:287–299. [Crossref]
- Tenurio J, Arias P, Martinez-Glez V, Santos F, Garcia-Minaur S, et al (2014) Simpson-Golabi-Behmel syndrome types I and II. Orphanet J Rare Dis 9: 138. [Crossref]
- Terespolsky D, Farrell SA, Siegel-Bartelt J, Weksberg R (1995) Infantile lethal variant of Simpson-Golabi-Behmel syndrome associated with hydrops fetalis. Am J Med Genet 59: 329–333. [Crossref]
- Lapunzina P (2005) Risk of tumorigenesis in overgrowth syndromes: A comprehensive review. Am J Med Genet C Semin Med Genet 137C: 53–71. [Crossref]
- Walker RWM, Ellwood J (2009) The management of difficult intubation in children. Pediatr Anesth 19: 77-87. [Crossref]
- Morita Y, Naito K, Kimoto N, Kimoto E, Omata T, et al. (2015) Anesthetic Management of a Child with Simpson-Golabi-Behmel Syndrome-A Case Report. Open Journal of Anesthesiology 5: 23-26.
- Brzustowicz LM, Farrell S, Khan MB, Weksberg R (1999) Mapping of a new SGBS locus to chromosome Xp22 in a family with a severe form of Simpson-Golabi-Behmel syndrome. Am J Hum Genet 65: 779–783. [Crossref]
- Xuan JY, Hughes-Benzie RM, MacKenzie AE (1999) A small interstitial deletion in the GPC3 gene causes Simpson-Golabi-Behmel syndrome in a Dutch-Canadian family. J Med Genet 36: 57-58. [Crossref]
- Agarwal M, Sharma R, Panda A, Gupta A (2009) Laryngeal web associated with Simpson-Golabi-Behmel syndrome in a child. Anaesth Intensive Care 37: 671-672. [Crossref]
- Vora N, Bianchi DW (2009) Genetic considerations in the prenatal diagnosis of overgrowth syndromes. Prenat Diagn 29: 923-929. [Crossref]
- Taniyama2021 Copyright OAT. All rights reservnagi M, et al. (2003) Craniofacial morphology in a patient with Simpson-Golabi-Behmel syndrome. Cleft Palate Craniofac J 40: 550-555. [Crossref]
- Morita Y, Kimoto N, Ogawa H, Omata T, Morita N (2011) Simpson-Golabi-Behmel Syndrome Associated with Cleft Palate. J Craniofac Surg 22: 1917-1918. [Crossref]
- Lapunzina P, Lopez RO, Rodriguez-Laguna L, Garcia-Miguel P, Martinez AR, et al. (2014) Impact of NGS in the medical sciences: Genetic syndromes with an increased risk of developing cancer as an example of the use of new technologies. Genet Mol Biol 37: 241-249. [Crossref]
- Gurkowski MA, Rasch DK (1989) Anesthetic Considerations for Beckwith-Wiedemann Syndrome. Anesthesiology 70: 711-712. [Crossref]
- Filippi G, Mckusick VA (1970) The Beckwith-Wiedemann Syndrome. Medicine 49:279-298.
- Xue FS, He N, Xu YC, Liao X (2009) Anesthesia and Airway Management for Children with Macroglossia. Pediatr Anaesth 19: 275-277. [Crossref]
- Inomata S, Maeda T, Shimizu T, Satsumae T, Tanaka M (2010) Effects of Fentanyl Infusion on Tracheal Intubation and Emergence Agitation in Preschool Children Anaesthetized with Sevoflurane. Br J Anaesth 105: 361-367. [Crossref]







